Received: December 9, 2013
Accepted: February 7, 2014
Meditter J Hematol Infect Dis 2014, 6(1): e2014013, DOI 10.4084/MJHID.2014.013
This article is available on PDF format at:

Arben Pilaca1, Gentian Vyshka2, Arben Pepa3, Kastriot Shytaj4, Valentin Shtjefni5, Arben Boçari6, Arben Beqiri7 and Dhimitër Kraja1
1Service
of
Infective Diseases, University Hospital Centre “Mother Theresa”,
Tirana, Albania
2Biomedical and Experimental Department, Faculty
of
Medicine, University of Tirana, Albania
3Obstetrical and Gynecological Hospital, Tirana,
Albania
4Faculty of Medical and Technical Sciences,
University
of Medicine in Tirana, Albania
5Institute of Veterinary Care, Tirana, Albania
6Faculty of Veterinary Medicine, Agricultural
University of Tirana, Albania
7Service of Surgery, University Hospital Centre
“Mother Theresa”, Tirana, Albania

|
This
is an Open Access article distributed
under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
|
Abstract Echinococcosis is an endemic zoonosis in the Mediterranean area, with Albania interested actually to a level that is becoming a public health concern. Authors describe preliminary data from the only tertiary (university) medical facility of Albania, positioned in the capital of the country (Tirana), with 333 new cases diagnosed and treated during the period 2005 – 2011. Out of all these 333 new cases an impressive majority of 91% had a surgical treatment right from the first admission, rendering the disease almost a surgical exclusivity. Even more, 80% of all patients from the study group were hospitalized straightforwardly in surgical wards, with options of surgical intervention’s percentages outrunning figures from other sources and authors of the same geographical area. Such a situation, together with a very important level of patients’ origin from highly urbanized areas such as those of the capital, suggest the necessity of well-organized interventions, among which might be the mandatory notification of all human cases with Echinococcus infection. |
Introduction
Echinococcosis
is a chronic and zoonotic infection, actually representing one of the
three major helminth diseases that cause a major public health concern
especially in developed countries, together with cysticercosis and
fascioliasis.[1]
Echinococcosis is synonymously known as the hydatid disease, with the
term ‘hydatid’ deriving from the Greek ‘hydōr’,
meaning water, and thus reflecting the cystic character of the
infective process. Controversial data regarding the epidemiology and
the severity of the infection among humans are available, with some
authors offering an optimistic perspective of a dramatic fall in the
incidence and prevalence of the most common form of the disease, namely
the cystic echinococcosis.[2]
However, and in spite of large preventive
and therapeutic interventions, cystic echinococcosis remains a frequent
condition in developed and undeveloped countries.[3,4]
Classic cystic echinococcosis (CE) is caused by E. granulosus, one
of the two major
species of the genus Echinococcus able to infect humans. E. multilocularis
is the second most important pathological species of this genus. These
species are respectively responsible for CE, and for the alveolar
echinococcosis (AE). AE is caused from E. multilocularis
and represents the other clinical picture related to these tapeworms,
able to infest humans as well as other mammalian intermediate hosts.
Wildlife animals and domesticated pets might be infested in a variety
of ways or forms, and from different species of Echinococcus.[5,6]
Thus, E. granulosus,
E. multilocularis,
E. vogeli and
E. oligarthus
have shown to have different but intrinsic pathological potential
regarding the ability to infest humans, with other species of the same
genus having only un unclear infective potential, if at all.[7,8]
Multiple studies have scrutinized risk factors and environmental
changes that influence on the global spread of the infection. Host
characteristics as well as host population dynamics and density are
among the most studied in a variety of animals. Obviously canids, dogs
in first line, serve as definitive host; foxes, jackals, dingo, hyena,
wolves and raccoon-dogs might be definitive hosts as well.[8,9] The
list of intermediate hosts might be even longer, with rodents on the
top, and with a variety of domesticated animals (sheep, pigs, goats)
serving that role, but without excluding even exotic or at risk of
extinction wildlife organisms, such as hippopotamus, giraffes,
antelopes (kudu) and so on.[8,10-12]
Echinococcosis is considered as an endemic zoonotic disease in the
Mediterranean area.[2] Some authors
suggest, with obvious and sound
reasons, that this disease with variegate clinical features, is posing
a severe threat even to the public health level.[13]
Both major
clinical forms, CE and AE, have been found present in different
Mediterranean countries, although CE and E. granulosus,
having the latter an intrinsic affinity for warmer climates, represent
the overwhelming form. Much more disturbing is the fact that E. multilocularis,
the causative factor of AE, generally considered as a species occurring
only sporadically in this geographical area, is however recently found
to expand his habitat continuously.[13,14]
Although representing a major public health problem and a considerable
burden of disease for several Mediterranean countries, epidemiological
data are scarce, and the precise incidence among humans is unknown for
CE as well as for AE in Albania and in countries neighboring it.[15]
With a worldwide prevalence of approximately six million, CE needs
global approaches and strategies to prevent further progression.[3]
Among the factors explaining this persistence of the disease, sources
quote the climate changes and the warmer temperatures.[16,17]
Host
density is another major factor; if there are no epidemiological data
regarding human incidence of echinococcosis in Albania, no data either
are offered regarding stray dog populations in our country.
The aim of the present study was to systematize preliminary data
gathered from our University Hospital Facility in Tirana, regarding
cases of echinococcosis diagnosed and treated during recent years
(2005-2011). An insight to epidemiological characteristics of this
zoonosis in Albania is given as well, and a geographical distribution
of cases was made, aiming at localizing districts of the country being
at “high risk” for possible outbreaks, due to the actual high
expression of the disease.
Materials and Methods
This retrospective and single-centre study was performed in the only
University Hospital Centre of Tirana, capital of Albania. Due to
panoply of reasons, district hospitals almost constantly refer cases to
this facility; however the data are not meant to represent the overall
country presence of the disease, since many cases of Echinococcosis
might go untreated.
UHC of Tirana is organized in several services that function as
clinical wards; all data and medical files are deposited in a central
statistical office, with medical doctors and authorized personnel able
to access their content decades after the discharge of a patient.
This is a descriptive study of the hospitalizations at the University
Hospital Centre “Mother Theresa” of Tirana (UHC of Tirana), during the
period 2005 – 2011, regarding all cases whose admission diagnosis was ‘echinococcosis’.
A study of the cases was made after carefully controlling the central
statistical office of UHC of Tirana, taking note of following details:
Results
During the period 1st
January 2005 –
31st December 2011 we had a total of 385 hospitalizations with the
admission diagnosis of ‘Echinococcosis’, for a total of 333 patients
(de novo and recurrent admissions), with 303 cases operated.
Only one case
had a fatal outcome, and is not included in the database of the Table 2.
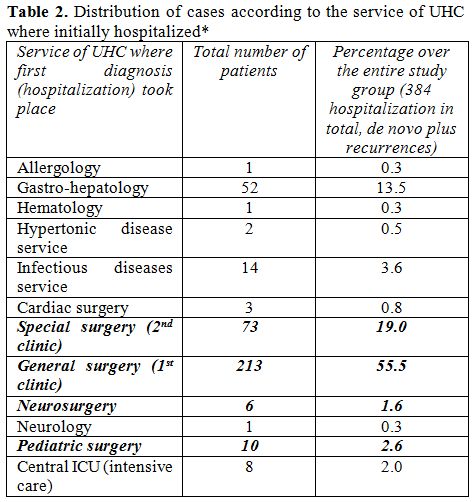
According to our data, 91% of the cases of echinococcosis admitted in
the UHC of Tirana during a seven years period, was initially admitted
or referred during the same hospitalization at the UHC, in a surgical
facility, and underwent a surgical intervention.
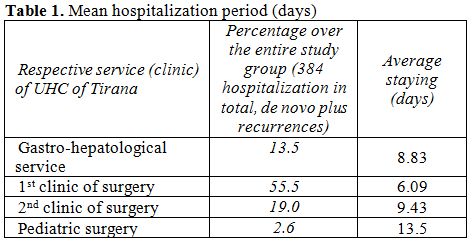
The mean period of hospitalization
is given in the Table 1,
where
are considered the services of UHC accounting for the majority of all
cases admitted during this period.
The mean age
of patients
forming the study group (a total of 333 patients) resulted 36 years old
(from a minimum of six month, to a maximum age of 77 years).
Overall distribution of cases in the different
clinical services of UHC of Tirana is described at the Table 2.
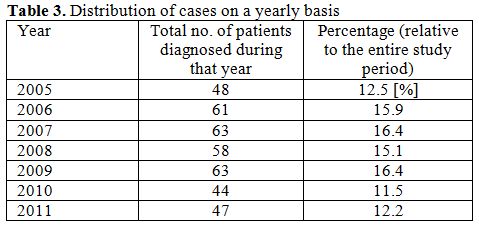
Time distribution (total yearly figures from 2005 to 2011) is described
in the Table 3.
Worthwhile is stating that it has been an almost constant figure year
after year (2005 – 2011) with a minimum of 44 cases during the year
2010 and a maximum of 63 cases (2007, 2009), thus the yearly level of
newly diagnosed cases did not changed substantially.
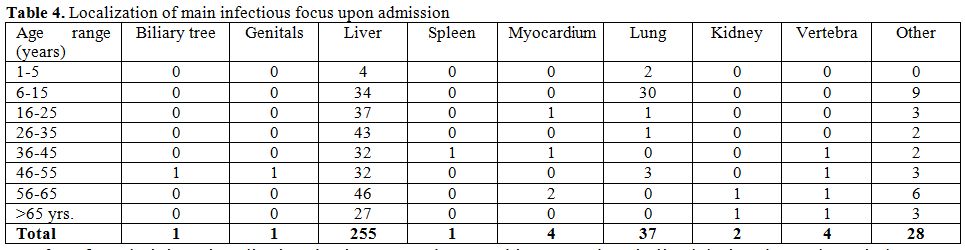
The clinical localization of the disease (visceral distribution; in
terms of the main focus upon admission) is described in the Table 4. The
distribution of cases
is made in separate age ranges (decennia).
 | Table 1. Mean hospitalization period (days) |
 | Table 2. Distribution of cases according to the service of UHC where initially hospitalized* |
 | Table 3. Distribution of cases on a yearly basis |
 | Table 4. Localization of main infectious focus upon admission |
The geographical
distribution of the cases was made accordingly with
the administrative map of Albania, shown in the Figure 1.
In order to simplify the exposition of data, we have separated
graphically the districts of the country in the map below through
illustrating the number of cases with a different color.
Thus, in the map below (Figure
1)
we have separated different district of the country through using
following illustrative colors:
Blue;
for administrative
districts having more than thirty cases hospitalized during the study
period;
Yellow-orange;
for
districts having 10-30 cases hospitalized during the same period;
Green;
for districts
having 5-9 hospitalizations in total (2005-2011);
Grey;
for districts
having less than five cases in total for the study period.
According to the data we gathered, we had a total of 99 patients
hospitalized during the study period, originating from the district of
Tirana (capital of the country, and surrounding areas). Thus, this city
and the respective district are shown in the map with blue color.
In fact, the absolute number of cases coming from this district was
overwhelmingly higher when compared with other districts of the
country.
With yellow-orange
color
there
are shown a total of eight districts of Albania, including four
northern administrative districts (namely Kukës [with 30 patients],
Dibra [17 patients], Shkodra [13 patients] and Tropoja [10 patients]).
With the same color we illustrated four other districts almost
symmetrically positioned in the lower half of the country’s map,
including a central Albanian district (Elbasan with 10 patients); a
south-west district close to the Adriatic seashore (Fier with 13
patients); a south-east district neighboring Greece (Korça with 21
patients) and a southern district almost at the lower extreme of the
map (Gjirokastra with 14 patients).
Other administrative districts of the country having less than 10
hospitalizations during the entire study period were depicted in green and grey
(see map). The distribution of the cases in these districts followed a
disparate order as well, although districts close to the Adriatic
seashore had somehow higher absolute figures (port cities of Durrës and
Vlora [shown in green] with 9 patients each); when compared to southern
– southeastern districts neighboring Greece, with lower absolute
figures (Devoll, Delvina and Skrapar [shown in grey] each of them with
1 patient for the entire study period).
The size of cysts was registered only in a minority of our cases (114 patients), and the available data were collected from echography findings. Due to this gap we cannot refer exhaustive values of cyst sizes in our study group; however the size of the cysts’ diameter varied from a minimum of 2 centimeters to a maximum of 15 centimeters, in this subgroup of patients where the cyst size was registered.
Discussion
Echinococcosis (hydatidosis) is an endemic disease for the
Mediterranean area, and Albania will obviously be interested to the
same extent as other countries of the region. Since no exhaustive or
thorough statistical and epidemiological study is available, comparing
the epidemiological situation with that of neighboring countries still
remains a challenge.
As far as it regards a non-reportable infection, collecting statistics
for an entire country might be virtually impossible. Nevertheless,
attempts to profile and to analyze echinococcosis in endemic regions
have been made.[18] Another non
trivia0l difficulty is related to the
panoply of clinical features through which echinococcosis might
present, becoming in certain situation a diagnostic conundrum. Our
study confirms as well an almost universal finding, that the majority
of cases of echinococcosis are confined to the liver and to the
lungs.
Hydatid cyst will show a clear mass effect and therefore will be a
surgical exclusivity in many settings. Highly peculiar clinical forms
are reported, such as Budd-Chiari syndrome, left ventricle hydatid cyst
mimicking heart attack, genital localization with scrotal extension and
so on.[19-21] Such a variety of
localizations is related to the fact
that hydatidosis affects almost all body regions.
In the present paper we have encountered two worth mentioning phenomena:
A very high proportion of patients (99 from the total study group of 333) originating from the district of Tirana, comprising the capital and its suburban areas;
An excessively high proportion of surgical approach as well, with 91% of all patients forming our study group that were operated during the initial admission at the UHC of Tirana, and even with almost 80% of all patients admitted straightforward in a surgical facility (here including general, special, pediatric and neurological surgery services).
The fact that we had the majority of cases originating from Tirana
might be artifactual, since UHC itself is situated in this capital
city, and inhabitants of the district will have an easier access to the
facility. However, UHC of Tirana is the single tertiary (university)
hospital in Albania; therefore all difficult and complicated cases will
be referred herein. This is much true nowadays when almost all
specialized medical help, staff and equipment, is focused inside this
centre, with periphery and remote districts suffering from inexistent
or incompetent medical services.
The high proportion of cases originating from Tirana might be also
related to other reasons. High and aggressive urbanization of the
capital and its suburban areas, with the sanitary problems that such a
process with entail, here including even the garbage disposal and
landfills (Sharra landfill among other) in the vicinity of the city,
have been at the focus of media reports.[22]
The presence of
domesticated animals that are part of the transmitting chain of this
infection to humans, such as sheep and dogs, inside landfills and in
the city itself has been a constant concern. Serious attempts have been
made to study the intestinal parasite fauna of dogs of the suburban
area of Tirana, with Echinococcus being part of the list.[23]
The other major suggestion of the present study was the fact that
actually in Albania, and in the only tertiary medical facility of the
country (UHC), echinococcosis has practically become a surgical
exclusivity. Approximately 90% of the entire study group admitted with
this diagnosis during 2005-2011 ended up with a surgical intervention.
But probably the most important fact is that 80% of all patients were
right from the start hospitalized in a surgical ward, thus the
condition has (almost) never been consulted from infectious disease
specialists, with treatment focused alone in the surgery. In fact, from
all patients treated surgically, only 23 of them had later (within six
months from the intervention) an infectionist consultancy, and none of
these patients were receiving an ad hoc pharmacological therapy prior
to surgery.
This very high level of surgical interventions contrasts with findings
from other tertiary centers, with authors from countries neighboring
Albania referring surgery as a single option only in 10% of the study
group.[4]
Under these circumstances, and in view of the endemic situation in the
Mediterranean area, mandatory notification of all human cases of
Echinococcus infection, through taking necessary steps toward rendering
the condition obligatorily a reportable one, seems more than logical.
References






















[TOP]