Received: December 12, 2013
Accepted: April 27, 2014
Meditterr J Hematol Infect Dis 2014, 6(1): e2014036, DOI 10.4084/MJHID.2014.036
This article is available on PDF format at:

Prakas Kumar Mandal1, Supriyo Sarkar2 , Malay Kumar Ghosh3 and Maitreyee Bhattacharyya3
1
Assistant Professor and 3Professor, Department
of Hematology and 2Professor, Department of
Respiratory Medicine, Nilratan Sircar Medical College, Kolkata-700014,
WB, India.

|
This
is an Open Access article distributed
under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
Dear Editor,
We read with interest the article recently published in Mediterranean
journal of Hematology and Infectious Diseases, in whom
Fattorini
et al. pointed out that two billion people
worldwide are
latently infected with Mycobacterium Tuberculosis (Mtb), with a
10% reactivating to active tuberculosis (TB) due to re-growth
of
non-replicating (dormant) Mtb residing in their tissues.[1]
The mechanism of reactivation is complex and can be
attributed to
the treatment with corticosteroids and/or any other condition affecting
T cell function.[2,3] Here we
report a case of
concurrent TB lymphadenitis and diffuse large B- cell lymphoma (DLBCL).
Tuberculous lymphadenitis is one of the common extra-pulmonary
tuberculosis (TB); it is usually diagnosed by fine needle aspiration
cytology (FNAC) which demonstrates caseating epithelioid granulomas and
presence of acid fast bacilli (AFB). Majority of TB lymphadenitis
respond to anti-TB therapy (ATT). However new lymph nodes may appear,
or existing lymph nodes may increase in size during treatment in
immune-suppressed as well as immune-competent patients.[4]
The most important cause of unresponsive lymphadenopathy is infection
caused by atypical or drug resistant Mycobacteria. However rarely
simultaneously double pathology of lymph node may be responsible for
unresponsiveness to ATT extremely diverse pathology.
A 72 years old, non-smoker, non-alcoholic, hypertensive and
non-diabetic female patient attended a private hospital with a history
of low grade fever, cough and multiple swellings in the neck. Her
peripheral blood counts, chest X-ray, were normal. FNAC from the
cervical lymph node showed caseous necrosis and presence of acid fast
bacilli (AFB) in Ziehl Neelsen stain. Her sputum was negative for AFB.
ATT was started as per standard guideline. After one and half month of
treatment, her condition deteriorated existing lymph nodes, appearance
of new lymph nodes with persisting fever, progressively increasing size
of in axillary and inguinal regions. Also, there was significant weight
loss.
At this stage, she was referred to the Haematology department of our
institute by the chest physician from the private hospital. She had
pallor, bilateral pitting oedema, but no organomegaly. Respiratory
examination showed right sided pleural effusion. Complete haemogram
showed hemoglobin, 108 gm/l; total leucocyte count, 8.3x109/l with 65%
neutrophil, 26% lymphocytes, 03% eosinophil, 06% monocyte; Platelets,
1.58x109/l,
ESR–110mm in hour, Corrected reticulocyte count, 2.1% and Direct Coombs
Test was negative. Serum chemistry was normal. Screening for
viral markers HBsAg, Anti-HCV and anti-HIV-I &II were negative.
Contrast enhanced computed tomography (CECT) of thorax and neck
revealed bilateral enlargement (more evident on the right side) of
cervical, mediastinal, axillary lymph nodes and pleural effusion. CECT
of the abdomen including pelvis revealed multiple, enlarged
retroperitoneal and bilateral inguinal lymph nodes. Pleural fluid cell
count was 2500 x 10 /l
with the
presence of atypical mononuclear cells, glucose 39 mg/dl, protein 3.2
g/l and ADA (adenosine deaminase) 28 IU/L. Cervical lymph node
excisional biopsy revealed complete effacement of the lymph node
architecture with presence of diffuse, monotonous large atypical
lymphoid cells having irregular nuclear contour, vesicular nuclei and
prominent multiple nucleoli suggestive of Non Hodgkin’s Lymphoma (NHL),
diffuse, large cell type (Figure
1).
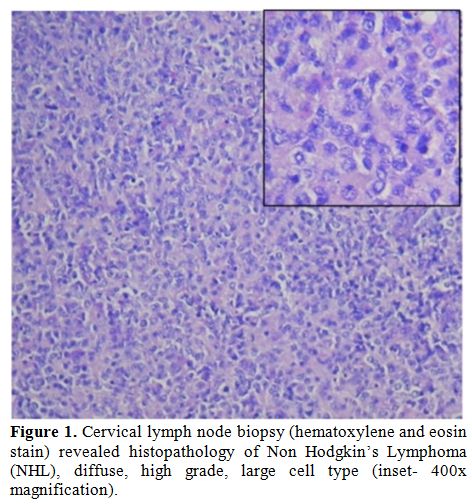 | Figure 1. Cervical lymph node biopsy (hematoxylene and eosin stain) revealed histopathology of Non Hodgkin’s Lymphoma (NHL), diffuse, high grade, large cell type (inset- 400x magnification). |
Immunohistochemistry
(IHC) of the tumor cells expressed CD20, PAX-5, CD30 and negative for
CD3, Alk-1, CD15, Cytokeratins. Trephine biopsy of bone marrow did not
show any granuloma or lymphoid infiltration. Echocardiography revealed
good left ventricular systolic function with mild diastolic
dysfunction. Considering the available clinical, radiological and
histopathological data, she was diagnosed as a case of DLBCL,
Stage-IVB, international prognostic index (IPI) score-4 (high risk)
with concurrent tubercular lymphadenitis. She received six cycles of
R-CHOP (Rituximab, Cyclophosphamide, Doxorubicin, Vincristine,
Prednisolone) along with ATT. She achieved complete remission (CR) of
both the diseases confirmed by negative whole body PET-CT and bone
marrow biopsy, and biopsy from one persistent right axillary lymph node
showed fibro-fatty changes without any evidence of TB or malignancy. TB
lymphadenitis usually presents with multiple matted cervical lymph node
enlargement with variegated consistency and sometimes with sinus
formation; whereas, in lymphomas, lymph nodes are discrete and firm in
consistency. Histopathological demonstration of granuloma is not
necessary for diagnosis of TB lymphadenitis if FNAC demonstrates AFB.
The diagnosis of NHL is confirmed by histopathological examination and
immunohistochemistry.
In a country like India, both TB and NHL are common, but simultaneous
occurrence of the diseases has not been reported before. NHL suppresses
immune system directly or as a result of treatment. As lymphomas
primarily involve the lympho-reticular system, they decrease cell
mediated immunity.[5] Depressed
cell-mediated immunity can activate dormant bacilli in lymph nodes and
thereby might cause active TB lymphadenitis.[3]
Various viruses like Epstein Barr virus, human herpes virus 8, human
immunodeficiency virus has been implicated as possible etiologic
factors of different types of NHL. The hypothesis in favor of these is
i) viruses directly transforming lymphocytes or ii) causing profound
depletion of CD4+ T-lymphocytes. NHL in many conditions has been found
to be preceded by chronic inflammatory diseases.[5]
Association of Helicobacter pylori, campylobacter and Hepatitis C virus
with NHL has already been well established.[6]
But till date, there is no evidence that Mycobacterium tuberculosis
predisposes to NHL.
Conclusion
Randomized study to compare CHOP chemotherapy plus rituximab with CHOP alone in elderly patients with diffuse large-B-cell lymphoma revealed that, addition of rituximab to the CHOP regimen increases the complete-response rate and prolongs event-free and overall survival in elderly patients, without clinically significant increase in toxicity.[7] The present case achieved complete remission with R-CHOP regimen and at the same time the patient was also cured of TB with ATT. Surprisingly, TB did not response till NHL was taken care of, which is difficult to explain. TB is affected by cell-mediated immunity (T-cells) mainly, whereas DLBCL is primarily a B cell neoplasm. Though combination of TB lymphadenitis and Hodgkin’s lymphoma has been reported,[8] the combination of TB lymphadenitis and NHL has not been signaled in the current literature. Our data do not permit to establish if TB precipitated NHL by chronic immune-stimulation or immunodepression present in NHL activated dormant TB bacilli in lymph nodes. A third possibility might be that, both the diseases occurred as a separate entity without any causal relationship. The case will give some impetus to think differently in cases of unresponsive lymphadenitis and raise a question whether histopathological examination of lymph nodes should be done despite the demonstration of AFB in FNAC.
References








[TOP]