Davide Pata1,2, Danilo Buonsenso1,2,3 and Piero Valentini1.
1 Department of Woman and Child Health and Public Health, Fondazione Policlinico Universitario A. Gemelli, Rome, Italy.
2 Global Health Research Institute, Istituto di Igiene, Università Cattolica del Sacro Cuore, Roma, Italia.
3
Dipartimento di Scienze Biotecnologiche di Base, Cliniche
Intensivologiche e Perioperatorie, Università Cattolica del Sacro
Cuore, Rome, Italy.
Correspondence to: Davide Pata. Largo A. Gemelli 8, 00168, Roma, Italy. Tel: +39 063015 4390. E-mail:
davide.pata01@gmail.com
Published: September 1, 2022
Received: February 11, 2022
Accepted: August 8, 2022
Mediterr J Hematol Infect Dis 2022, 14(1): e2022065 DOI
10.4084/MJHID.2022.065
This is an Open Access article distributed
under the terms of the Creative Commons Attribution License
(https://creativecommons.org/licenses/by-nc/4.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
|
Abstract
Background and Objectives:
Coronavirus disease 2019 (COVID-19) is caused by SARS-CoV-2 and has a
clinical presentation ranging from an asymptomatic course to flu-like
syndrome up to respiratory failure. Seasonal Influenza, due to the
influenza viruses and very common in children, can cause symptoms
similar to COVID-19. In order to identify clinical and laboratory
characteristics that allow healthcare workers to differentiate COVID-19
from Influenza, we performed a systematic review of the existing
literature on the pediatric age. Methods. The research was done via
PubMed for articles published from March 2020 to October 2021,
combining the MeSH words "COVID-19" and "Influenza" and "Children" and
considering the suggestions of the PRISMA Group.
Results:
The most frequently described symptoms were fever and cough in both
groups. In most studies, high fever, cough, nasal congestion or
rhinorrhea, vomiting, and muscle pain were detected more frequently in
the Influenza group. Regarding the value of laboratory tests, the
results were mixed. Almost all studies reported significantly lower
levels of C-reactive protein and procalcitonin in the COVID-19 group
than in the Influenza group. In most manuscripts, COVID-19 had a milder
course than Influenza.
Conclusions:
No symptoms are characteristic of a single infectious agent, with
flu-like disorders being the most common. In addition, laboratory tests
do not help in the differential diagnosis; however, they show a limited
inflammatory response in COVID-19, which could explain the fewer
complications compared to adulthood, with a less severe clinical course.
|
Introduction
Coronavirus
disease 2019 (COVID-19) was first reported in Wuhan in December 2019,
spreading to the rest of the world and causing a major pandemic.[1] Actually, about 290 million cases and nearly 5 million deaths have been reported to date.[2]
The disease is caused by SARS-CoV-2 and has a clinical presentation
ranging from an asymptomatic course to flu-like syndrome up to
respiratory failure even at pediatric age.[3,4]
However,
although SARS-CoV-2 was found in children of all ages, including
newborns, the infection is generally milder, and the outcome is
favorable compared to adults.[5,6]
Adult patients
with COVID-19 are characterized by the presence of cough, fever,
dyspnoea, and lymphopenia. Men, especially if elderly and with
comorbidities, are at higher risk for severe acute respiratory syndrome
and death.[7]
The symptoms and mortality from
COVID-19 increase proportionally with age in a U-shaped pattern, with
the lowest rate described at 3-10 years of age.[6]
Seasonal
Influenza is a respiratory infection caused by the influenza viruses.
It can cause symptoms similar to COVID-19 and spreads in occasional
outbreaks.[8] It can have a serious course, even up to death, in elderly subjects or with chronic diseases.
Given
the health, social and economic consequences that COVID-19 is causing
in the world, to contain its spread, it would be essential to identify
clinical and laboratory characteristics that allow healthcare workers
to differentiate it from seasonal Influenza. This differential
diagnosis becomes especially important in the pediatric age, a period
in which, due to the characteristics of the behavior (with fewer
hygiene measures) and the immune system of children, Influenza is very
frequent.
To our knowledge, no studies performed a systematic
review to compare the clinical and laboratory characteristics between
COVID-19 and Influenza in the pediatric age. Consequently, we conducted
this research to evaluate children's differences between the two
infectious diseases.
Methods
We performed a systematic review of the existing literature considering the suggestions of the PRISMA Group. [9]
Search Strategy. Our
literature search strategy was aimed at comparing the clinical and
laboratory features of COVID-19 and Influenza in pediatric age (<18
years old).
The systematic search was conducted according to the
following PICOS approach: Population, pediatric patients with COVID-19
or Influenza infection; Intervention, study of the clinical features of
patients, and execution of blood samples for tests such as blood count,
blood chemistry tests, coagulation tests, inflammation indices, heart
damage indices; Comparison of clinical features and laboratory tests of
children with COVID-19 or Influenza; Outcomes, identify clinical
manifestations or laboratory tests allowing to diagnose/suspect
COVID-19 or Influenza without waiting for the microbiological
confirmation. Study design, all types of studies such as case reports,
cohort studies, and retrospective studies.
A systematic search
of PubMed was performed from March 2020 to October 2021, combining the
MeSH words "COVID-19" and "Influenza" and "Children".
Eligibility criteria and identification of studies. We
included in our review only studies aimed at comparing clinical and
laboratory characteristics in children with COVID-19 or Influenza.
Observational
cohort studies - prospective or retrospective - and case reports were
selected, including children under 18 and evaluating the presence of
elements useful for a differential diagnosis.
We excluded
manuscripts without full free text that were not in English, with
studies with different focuses and a population over 18 years of age.
Study selection. All
studies published between March 2020 and October 2021 were considered
(n= 414). To increase consistency among reviewers, all of them (three
seniors) screened the same publications, discussed results, and amended
the screening and data extraction before beginning screening for this
review. The same reviewers working in pairs then assessed the titles,
abstracts, and full text of all publications identified by our
searches, and we used an online platform to define which paper should
have been included (covidence.org). Finally, we resolved disagreements
on study selection and data extraction by consensus and discussion with
other reviewers if needed.
Data extraction. Two
reviewers independently extracted data from each included study
relating to clinical features, laboratory results, and reported
outcomes. Results were then checked again across the original
manuscript by a third researcher.
Data synthesis. Characteristics
of the included (and excluded) studies were presented in a tabulated
form on an excel sheet. The study data were collected in columns:
citation, year; country; type, retrospective or prospective; number of
patients involved; demographic findings; clinical features; laboratory
results, and outcome.
Quality assessment in individual studies.
The quality of included studies with comments about study limitations,
including the age groups which will limit the translation of results to
that specific age group, have been assessed by two reviewers.
Possible
biases are the inclusion of individual studies and the exclusion from
the analysis of manuscripts published in non-indexed journals.
Statistical analyses. A
direct comparison was not possible due to few observational and
retrospective studies. Therefore, study results were synthesized
between the two groups by comparing the symptoms, and the values
reported. A p-value <0.05 was considered significant.
Results
We initially imported 414 studies (Figure 1).
Three hundred forty-one were excluded after evaluation of title and
abstract. After assessment of the full text, a further 62 manuscripts
were eliminated: 45 were not relevant to the aim of our study, 15
concerned the adult population, and 2 because the full free text was
unavailable. Finally, 1 manuscript was added after the editor's
revision. A total of 12 articles were included:[10-21] 8 retrospective studies, 2 cohort studies, 1 cross-sectional study, and 1 case-control study (Table 1).
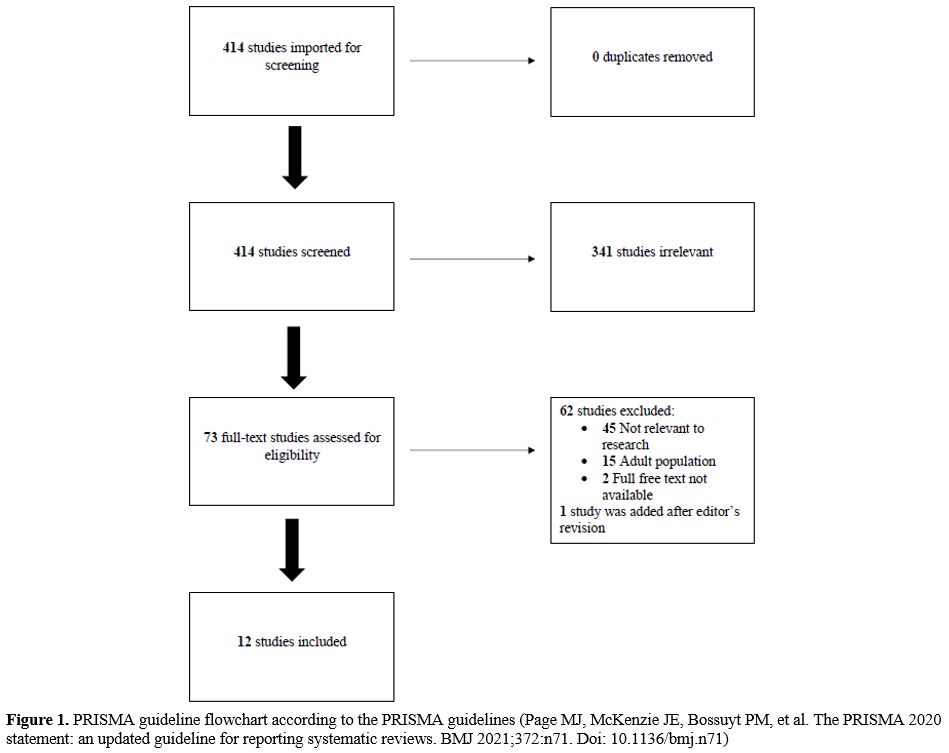 |
Figure 1. PRISMA guideline
flowchart according to the PRISMA guidelines (Page MJ, McKenzie JE,
Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for
reporting systematic reviews. BMJ 2021;372:n71. Doi: 10.1136/bmj.n71) |
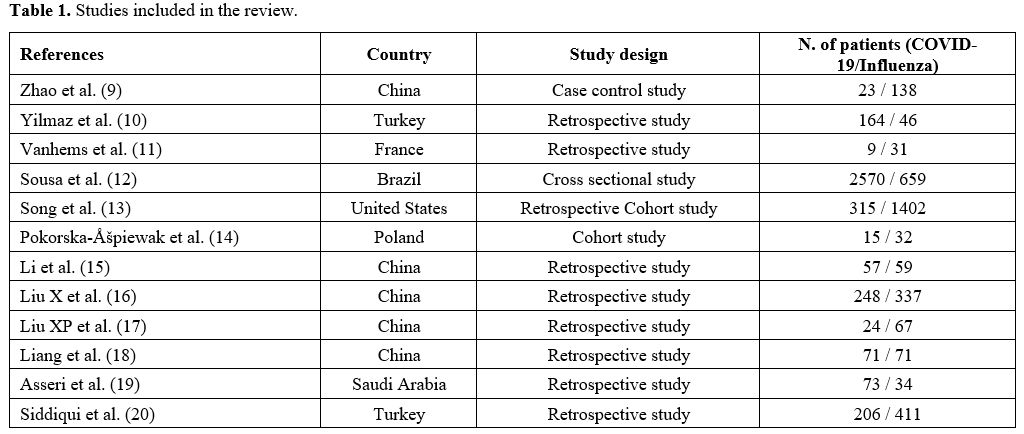 |
Table 1. Studies included in the review.
|
Clinical features in COVID-19 and Influenza (Table 2). A comparison of clinical features between COVID-19 and Influenza was reported in 11 of 12 studies.[10-17,19-21]
The
most frequently described symptoms were fever and cough in both groups.
However, unlike patients with COVID-19, no cases of asymptomatic
children were reported in the Influenza group.[15] In
most studies, high fever, cough, nasal congestion or rhinorrhea,
vomiting, and muscle pain were detected more frequently in the
Influenza group than in the COVID-19 group;[10-12,16,17,19] however, the results of the studies were not always statistically significant.
However,
Song et al. reported more symptoms (such as fever, cough, dyspnea,
chest pain, diarrhea or vomiting, myalgia, and headache) in children
with COVID-19 than in those with Influenza.[14] The remaining studies confirmed, although not statistically significant (Table 2),
that fever, dyspnea, and cough were higher in the Influenza group;
however, they found that the frequency of diarrhea was higher in
children with COVID-19.[13,15,20] Instead, Yilmaz et al. reported similar rates of diarrhea in the two groups.[11]
Siddiqui
et al. statistically confirmed that symptoms such as fever, cough,
rhinorrhea, vomiting, and abdominal pain were greater in children with
Influenza than in those with COVID-19.[21] Also, in
this study, diarrhea was more frequent (but non-statistically
significant) in the Influenza group than in the one with COVID-19,
while headache was a symptom more frequently reported by children with
COVID-19.[21]
Interestingly, Sousa et al.
described that most patients with severe COVID-19 had no fever or cough
at onset, unlike children with Influenza.[13]
Ageusia and anosmia are characteristic symptoms in adult COVID-19 patients.[7]
However, almost none of the studies included in the manuscript reported
these symptoms in the group of children with COVID-19. Only the study
by Siddiqui et al. pointed out that ageusia and anosmia were present
respectively in 4.9% and 3.4% of patients with COVID-19, while they
were not reported in the Influenza group. Probably this is due to the
age of the patients, not yet able to speak or report the presence of
symptoms, especially anosmia and ageusia, that are relatively complex
to explain for an individual who is not yet autonomous.
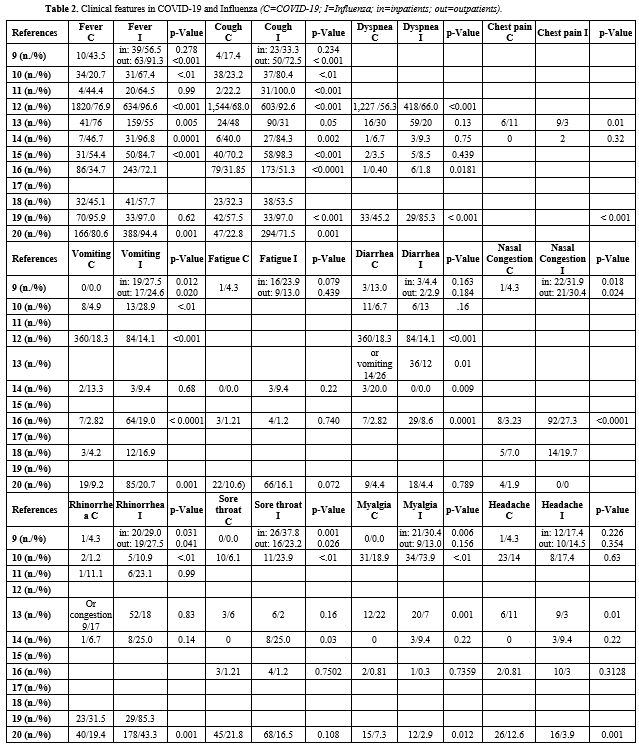 |
Table 2. Clinical features in COVID-19 and Influenza (C=COVID-19; I=Influenza; in=inpatients; out=outpatients). |
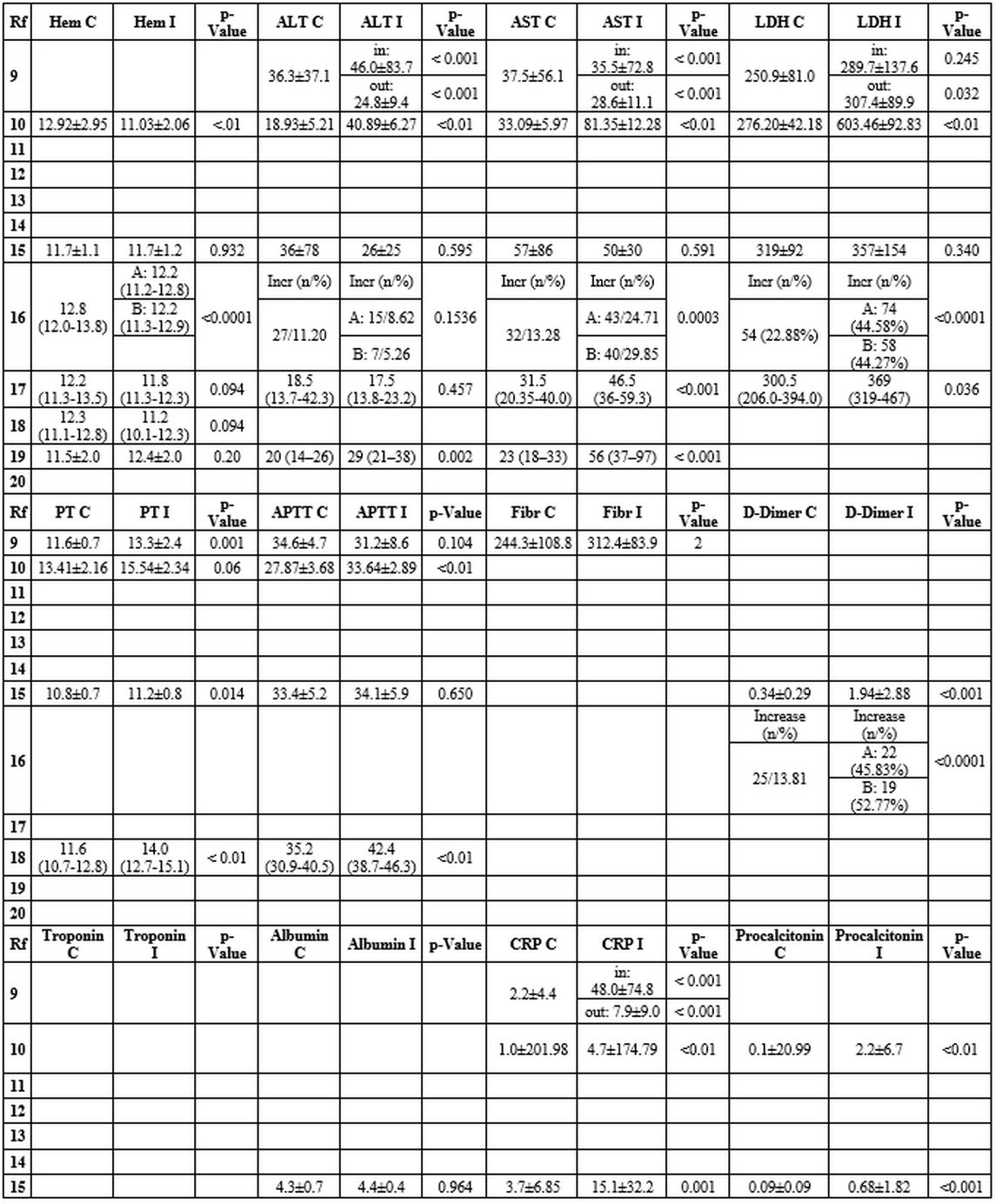
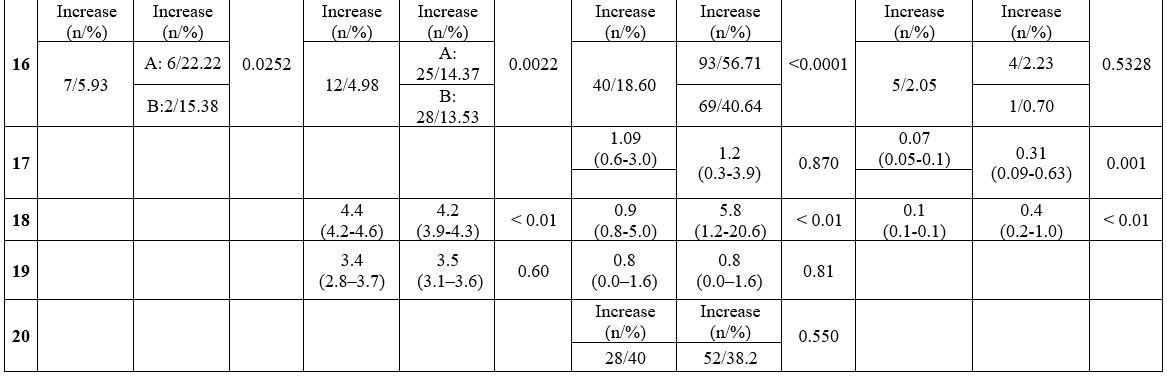
Laboratory findings in COVID-19 and Influenza (Table 3). A total of 8 out of 12 studies analyzed the differences in laboratory test results between COVID-19 and Influenza.
The
results were mixed regarding the value of the white blood cells count
(WBC) and the leukocyte formula. Most manuscripts reported a lower WBC
in children with COVID-19 than in those with Influenza.[11,16,20]
Other studies reported a higher percentage of leukocytopenia in the
Influenza group and a greater number of children with leukocytosis than
those with COVID-19.[17,21] Some studies described lower neutrophil count in the COVID-19 group than in the Influenza group.[11,16,19] Zhao et al. showed higher lymphocyte count in children with COVID-19,[10] while lymphocytopenia was more frequent in children with Influenza.[17,19,21]
Almost
all studies reported significantly lower levels of C-reactive protein
and procalcitonin in the COVID-19 group than in the Influenza group.[10,11,16,17,19]
No studies found an alteration of the coagulation tests. However, significantly lower values of prothrombin time,[10,11,16,19] aPTT,[11,19] and d-Dimer[16,17] were described in children with COVID-19 matched to those with Influenza.
In
the COVID-19 group, compared to the Influenza group, most manuscripts
reported significantly lower levels of alanine aminotransferase,[10,11,20] aspartate aminotransferase,[11,18,20] and lactate dehydrogenase.[11,18] However, Zhao et al. described higher levels of aspartate aminotransferase and lactic acid.[10]
Liu
et al. reported acute cardiac injury in approximately 7% of patients
with COVID-19, 20% with influenza A and 18% with influenza B.[17]
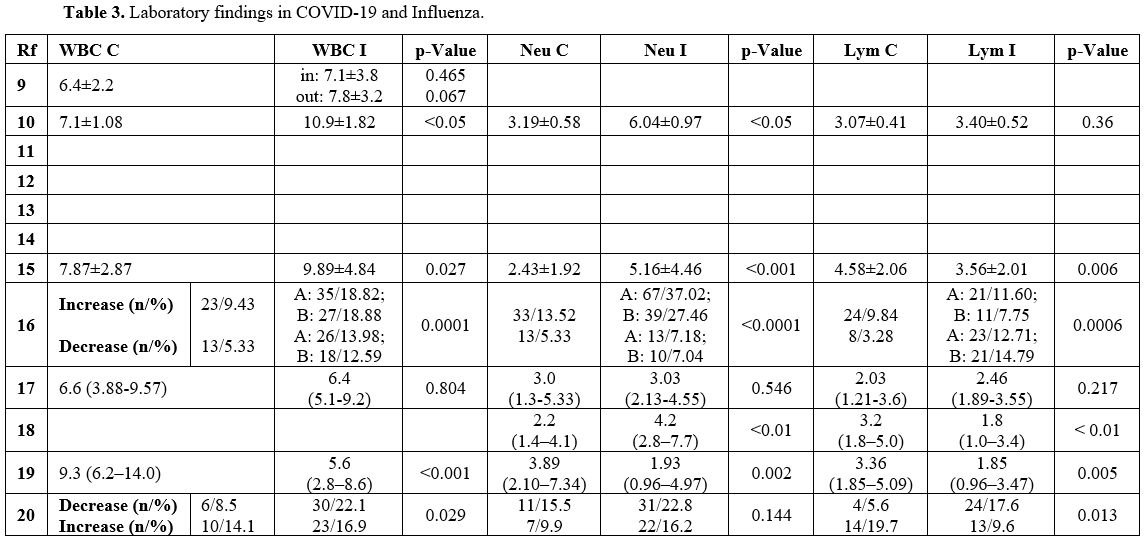 |
Table 3. Laboratory findings in COVID-19 and Influenza. |
 |
|
 |
|
Outcome (Table 4). 11 out of 12 studies analyzed the complications and consequences of infections.
In most manuscripts, COVID-19 had a milder course than Influenza.[12,19]
Indeed, children with Influenza were characterized by a higher rate of
hospitalization duration, need for intensive care, oxygen therapy,
ventilatory support, and mortality than those with COVID-19.[10,11,16,17]
Asseri
et al. reported that 11% of children in the COVID-19 group were
diagnosed with Inflammatory Syndrome in Children (MIS-C), which was not
observed in the Influenza group. Furthermore, they noted that children
with Influenza had a longer length of stay in the ICU than those with
COVID-19, which needed a longer period of oxygen therapy.[20]
Song
et al. described that the 2 groups had a similar rate of ICU admission;
however, 65% of children with COVID-19 had at least one comorbidity,
significantly higher than 42% with Influenza.[14]
Instead,
Pokorska-Åšpiewak et al. noted that patients hospitalized with
Influenza had a higher number of comorbidities, although not
statistically significant.[15]
Contrary to other
studies, Sousa et al. reported a worse outcome in the COVID-19 group.
Although these children had a lower need for non-invasive ventilatory
support, their mortality rate was higher than in the influenza group.[13]
Siddiqui
et al. reported no significant differences in oxygen therapy,
hospitalization, and mortality between the 2 groups. However, ICU
hospitalization was higher in children with COVID-19.[21]
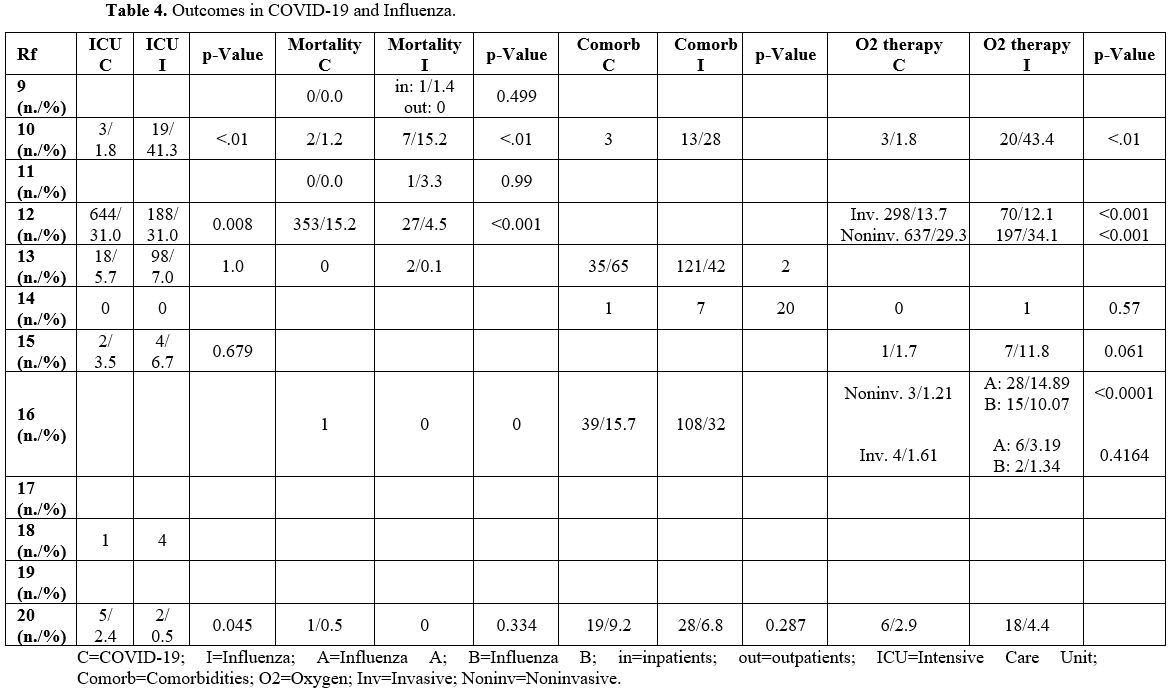 |
Table 4. Outcomes in COVID-19 and Influenza. |
Discussion
Our study compared the clinical characteristics, laboratory findings, and outcomes among children with COVID-19 or Influenza.
A
previous unsystematic review reported that SARS-CoV-2 infection is
generally mild in children, being asymptomatic or with symptoms of a
common viral upper respiratory infection, such as fever, cough, runny
nose, diarrhea, and vomiting, not allowing to distinguish COVID-19 from
any other community-acquired respiratory virus.[22]
In
our study, symptoms such as fever, cough, rhinorrhea, myalgia, and
vomiting were described more frequently in the Influenza group, while
some studies showed that diarrhea was more frequent in the COVID-19
group. Although these differences have not always been statistically
significant and have not been confirmed by all the studies analyzed,
they could help to differentiate the two etiological agents (perhaps by
combining them with other elements) and could be explained by the
different distribution of the receptors of the 2 viruses.
In fact, the Influenza virus uses sialic acid to penetrate inside human cells.[23] This receptor is mainly expressed in the upper respiratory tract,[24] explaining the greater frequency of flu symptoms and the rarity of neurological and gastrointestinal symptoms.
Instead,
the SARS-CoV-2 receptor is represented by angiotensin-converting enzyme
2 (ACE2). This metallopeptidase has a ubiquitous distribution,
including intestinal and alveolar epithelial cells and central nervous
system endothelial cells.[25] This datum would
justify the higher incidence of pneumonia, gastrointestinal and
neurological symptoms (including anosmia and ageusia not described in
Influenza) in patients with SARS-CoV-2 infection.
As described in adulthood,[26]
the presence in a patient of fever, cough, rhinorrhea, myalgia, and
arthralgia does not allow to distinguish between the 2 infectious
diseases, but the addition of diarrhea, perhaps associated with anosmia
or ageusia, should lead to a preponderance towards COVID-19.
An extensive systematic review conducted in adulthood[27]
showed that rhinorrhea, sore throat, dyspnoea, nausea, and vomiting
were less common in COVID-19 patients than in those with Influenza,
confirming our data. Still, they found no differences in
gastrointestinal symptoms between the 2 groups. Therefore, an analysis
of more data is needed to ensure the importance of diarrhea in the
diagnosis of pediatric COVID-19.
Laboratory findings did not show specific changes in a single infectious agent.
Some studies reported lower neutrophils in the COVID-19 pediatric groups,[11,16,19] like that observed in adulthood.[26]
Lymphocytopenia, described in the general population affected by severe COVID-19,[26] was not a diagnostic marker either:[28]
it does not appear to be a peculiar feature of this infection and does
not allow to distinguish SARS-CoV-2 pneumonia from influenza pneumonia.
However, lymphopenia seems to be associated with disease severity for
both conditions in adulthood.
Lymphocytopenia seems to be
generated by the direct infection of the lymphocytes and by the damage
consequent to the "cytokine storm",[26] a characteristic generally not present in the pediatric age.[25]
All this could explain the different values found in the leukocyte
formula and the absence of elevated inflammatory markers. Our review,
in fact, found lower levels of CRP and Procalcitonin in the COVID-19
group compared to Influenza. They are acute phase proteins produced
after inflammatory stimuli following an infection. While severe
COVID-19 in adulthood is associated with high levels of proinflammatory
cytokines, acute phase proteins, and consequent tissue damage,[26]
the low values of pediatric age testify to a less exuberant immune
response and a milder clinical course. Therefore, the alteration of
hemostasis and the hypercoagulability characteristic of severe COVID-19
are not present, as confirmed by the finding of normal coagulation
tests. The lack of the cytokine storm would also justify the lower
levels of transaminases, lactate dehydrogenase, and indices of
myocardial damage.
These data are also confirmed by numerous
pediatric COVID-19 studies, where the white blood cell count and
inflammation indices are normal in most cases. Instead, although not
specific to SARS-CoV-2 infection, lymphocytopenia and an increase in
C-reactive protein, procalcitonin, D-dimer, muscle enzymes, and liver
enzymes were described in the most severe rare cases,[29,30]
infrequently included in our study. Therefore, although not useful in
diagnosis, these altered laboratory tests could be useful in defining
the prognosis of the infection.
Regarding the outcome, both
infectious agents can lead to ARDS and death. Although affected by the
timing and by the region (the quality of the care offered varies
according to the phase of the pandemic, the "preparedness" of the
health system and the economic level of the country), COVID-19 showed
higher mortality or worse outcomes than Influenza in adulthood.[26,28,31-33]
During the pediatric age, the SARS-CoV-2 infection is rarely fatal.[34] In 2020, the mortality rate was two cases per million in England.[35] A systematic review reported a good prognosis in children under five years, with less than 10% of cases of severe COVID-19.[36]
Nearly
all of the studies included in our review reported lower severity of
SARS-CoV-2 infection compared to Influenza, both for ICU admission and
mortality. However, only Sousa et al. described, albeit rare, a higher
death rate in COVID-19. This contradictory finding could be caused by a
large number of cases in Brazil, associated with disparities in the
socio-economic level and provision of health care in different regions.[13]
Instead,
in our study, we did not find a greater number of complications
secondary to an infection, as evidenced in adulthood, in the COVID-19
group compared to that with Influenza, such as thrombosis[37] and acute kidney injury.[38]
The
lower severity of the infection in the pediatric population could be
explained by the presence of a more "plastic" immune system, able to
offer better performance against a new infectious agent, in contrast to
the predominantly "memory" response of the adult immune system.
Additionally, children have an increased ability to control the immune
response and repair tissue damage.[39]
Oualha et
al. highlighted the existence of severe COVID-19 even at pediatric age,
with a non-negligible death rate (18%) in an observational study
conducted in a pediatric intensive and high-dependency care unit (PICU)
in an urban hospital in Paris, mostly in children with comorbidities
but even in children without comorbidities. Furthermore, they suggested
the presence of a cytokine storm with subsequent prothrombotic events,
as described in adult patients.[40]
A greater
number of complications from COVID-19 compared to Influenza, such as
hospitalization and the need for oxygen therapy, were also reported by
a large international study.[41] Although this
manuscript included approximately 250,000 children with COVID-19 and 2
million with Influenza, due to the presence of electronic data
collected for administrative purposes, not completely available to our
literature research and with unclear statistical analysis, it was
excluded from our review.
Wei described a case fatality rate of
SARS-CoV-2 pneumonia in children aged <5 years lower than that of
respiratory syncytial virus (RSV) pneumonia but higher than those of
Influenza pneumonia.[42]
Piroth et al. reported
a 3-times greater mortality from COVID-19 in the general population
than from Influenza, with twice the likelihood of being hospitalized in
ICU and receiving invasive mechanical ventilation. In addition, a
subgroup analysis found that in-hospital mortality of children with
COVID-19 was more than four times higher than those with Influenza.[43]
Consequently,
studies with a large number of children are needed to evaluate the
outcome of SARS-CoV-2 infection in pediatric age compared to Influenza
infection. The initial estimates that predicted a better outcome in the
COVID-19 group would probably be revised with the increase in cases in
the pediatric population initially spared from infection in the early
stages of the pandemic.
Several studies described typical radiological features of COVID-19, both via chest CT and ultrasound.[44,45]
However, we decided not to include this topic in our review given the
limited data comparing chest CT (limited use due to minor disease
severity and to save exposure to ionizing radiation in pediatric age)
and chest ultrasound (limited use in pre-pandemic years) in SARS-CoV-2
and Influenza infections.
In fact, although chest CT was used for
COVID-19 patients in the early stages of the pandemic, radiological
exams are not routine tests for children with respiratory symptoms.[34]
They do not modify the first level assistance or therapy, and, to avoid
the potential damage caused by ionizing radiation, they are not used
unless clinical worsening and the onset of complications. Therefore, it
is rare to find its use in data on children with Influenza. Pulmonary
ultrasound was employed in diagnosing and monitoring pneumonia; this
new method was used for the advantage of being carried out bedside
during the pandemic,[46] but there are few data from previous years in the Influenza group.
Our
systematic review has several limitations. The number of studies
included and the sample size are limited due to the lower incidence of
pediatric infection recorded in the early stages of the pandemic.
Furthermore, the presence of retrospective studies, with data on
Influenza reported from previous-search databases, could potentially
influence the results. Due to the greater survival of chronic diseases
and syndromes, the number of comorbidities and risk factors has
increased, requiring a comparison between epidemic seasons of the same
year. The lack of information on SARS-CoV-2 and Influenza infections in
out-of-hospital settings and in primary care may have generated bias in
the comparison between the two groups. The included studies did not
analyze the different variants of the two viruses. The spread of more
infectious variants could increase the number of pediatric cases and a
greater significance of previously considered rare events.
Finally,
the use of only articles with data available and open-access and the
initial selection through abstracts may have led to the omission of
some manuscripts.
Conclusions
Our
study compares clinical and laboratory characteristics between COVID-19
and Influenza in children. To our knowledge, it is the only systematic
review carried out at the pediatric age. No symptoms are characteristic
of a single infectious agent, with flu-like disorders being the most
common. However, the presence of diarrhea could be a discriminating
factor. Laboratory tests do not help in the differential diagnosis but
show a limited inflammatory response in COVID-19. This is reflected in
complications, with a less severe clinical course and rare fatal events.
Prospective
studies are needed, with a larger sample size and comparison from the
hospital and non-hospital cases to confirm our observations.
References
- Zhu N, Zhang D, Wang W, et al.; China Novel
Coronavirus Investigating and Research Team. A Novel Coronavirus from
Patients with Pneumonia in China, 2019. N Engl J Med. 2020 Feb
20;382(8):727-733. doi: 10.1056/NEJMoa2001017. Epub 2020 Jan 24. https://doi.org/10.1056/NEJMoa2001017 PMid:31978945 PMCid:PMC7092803
- Available at https://covid19.who.int/; accessed 06/01/2022
- Li
Y, Guo F, Cao Y, Li L, Guo Y. Insight into COVID-2019 for
pediatricians. Pediatr Pulmonol. 2020 May;55(5):E1-E4. doi:
10.1002/ppul.24734. Epub 2020 Mar 18. https://doi.org/10.1002/ppul.24734 PMID: 32187887 PMCID: PMC7167677
- Buonsenso
D, Sali M, Pata D, et al. Children and COVID-19: Microbiological and
immunological insights. Pediatr Pulmonol. 2020 Oct;55(10):2547-2555.
doi: 10.1002/ppul.24978. Epub 2020 Jul 30. https://doi.org/10.1002/ppul.24978 PMid:32710652
- Flerlage
T, Boyd DF, Meliopoulos V, Thomas PG, Schultz-Cherry S. Influenza virus
and SARS-CoV-2: pathogenesis and host responses in the respiratory
tract. Nat Rev Microbiol. 2021 Jul;19(7):425-441. doi:
10.1038/s41579-021-00542-7. Epub 2021 Apr 6. ttps://doi.org/10.1038/s41579-021-00542-7 PMid:33824495 PMCid:PMC8023351
- Khera
N, Santesmasses D, Kerepesi C, Gladyshev VN. COVID-19 mortality rate in
children is U-shaped. Aging (Albany NY). 2021 Aug
18;13(16):19954-19962. doi: 10.18632/aging.203442. Epub 2021 Aug
18. https://doi.org/10.18632/aging.203442 PMid:34411000 PMCid:PMC8436910
- Sun
D, Li H, Lu XX, et al. Clinical features of severe pediatric patients
with coronavirus disease 2019 in Wuhan: a single center's observational
study. World J Pediatr. 2020 Jun;16(3):251-259. doi:
10.1007/s12519-020-00354-4. Epub 2020 Mar 19. https://doi.org/10.1007/s12519-020-00354-4 PMid:32193831 PMCid:PMC7091225
- Available at https://www.who.int/health-topics/influenza-seasonal#tab=tab_1; accessed 06/01/2021
- Page
MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an
updated guideline for reporting systematic reviews. BMJ. 2021 Mar
29;372:n71. doi: 10.1136/bmj.n71. https://doi.org/10.1136/bmj.n71 PMid:33782057 PMCid:PMC8005924
- Zhao
Y, Sun L, Bouchard HC, et al. Coronavirus Disease 2019 versus Influenza
A in Children: An Observational Control Study in China. Biomed Environ
Sci. 2020 Aug 20;33(8):614-619. doi: 0.3967/bes2020.080. PMID: 32933613
PMCID: PMC7505108.
- Yılmaz K, Şen
V, Aktar F, Onder C, Yılmaz ED, Yılmaz Z. Does Covid-19 in children
have a milder course than Influenza? Int J Clin Pract. 2021
Sep;75(9):e14466. doi: 10.1111/ijcp.14466. Epub 2021 Jun 27. https://doi.org/10.1111/ijcp.14466 PMID: 34107134 PMCid:PMC8237020
- Vanhems
P, Endtz H, Dananché C, Komurian-Pradel F, Sanchez Picot V; Pneumonia
Study GABRIEL members*. Comparison of the Clinical Features of
SARS-CoV-2, Other Coronavirus and Influenza Infections in Infants Less
Than 1-Year-Old. Pediatr Infect Dis J. 2020 Jul;39(7):e157-e158. doi:
10.1097/INF.0000000000002705. https://doi.org/10.1097/INF.0000000000002705 PMid:32525645 PMCid:PMC7279055
- Sousa
BLA, Sampaio-Carneiro M, de Carvalho WB, Silva CA, Ferraro AA.
Differences among Severe Cases of Sars-CoV-2, Influenza, and Other
Respiratory Viral Infections in Pediatric Patients: Symptoms, Outcomes
and Preexisting Comorbidities. Clinics (Sao Paulo). 2020 Nov
30;75:e2273. doi: 10.6061/clinics/2020/e2273. https://doi.org/10.6061/clinics/2020/e2273 PMid:33263633 PMCid:PMC7688070
- Song
X, Delaney M, Shah RK, Campos JM, Wessel DL, DeBiasi RL. Comparison of
Clinical Features of COVID-19 vs Seasonal Influenza A and B in US
Children. JAMA Netw Open. 2020 Sep 1;3(9):e2020495. doi:
10.1001/jamanetworkopen.2020.20495. https://doi.org/10.1001/jamanetworkopen.2020.20495 PMid:32897374 PMCid:PMC7489826
- Pokorska-Śpiewak
M, Talarek E, Popielska J, et al. comparison of clinical severity and
epidemiological spectrum between coronavirus disease 2019 and Influenza
in children. Sci Rep. 2021 Mar 11;11(1):5760. doi:
10.1038/s41598-021-85340-0. https://doi.org/10.1038/s41598-021-85340-0 PMid:33707568 PMCid:PMC7952543
- Li
Y, Wang H, Wang F, et al. comparison of hospitalized patients with
pneumonia caused by COVID-19 and Influenza A in children under 5 years.
Int J Infect Dis. 2020 Sep;98:80-83. doi: 10.1016/j.ijid.2020.06.026.
Epub 2020 Jun 12. https://doi.org/10.1016/j.ijid.2020.06.026 PMid:32535301 PMCid:PMC7289729
- Liu
X, Li W, Zhang B, et al. Comparative study of hospitalized children
with acute respiratory distress syndrome caused by SARS-CoV-2 and
influenza virus. BMC Infect Dis. 2021 May 4;21(1):412. doi:
10.1186/s12879-021-06068-w. https://doi.org/10.1186/s12879 -021-06068-w PMid:33947345 PMCid:PMC8093914
- Liu
XP, Guo MM, Liu SF, Kuo HC. Comparison of laboratory data between
children with COVID-19 and Influenza. Kaohsiung J Med Sci. 2021
Feb;37(2):158-159. doi: 10.1002/kjm2.12353. Epub 2021 Jan 15. https://doi.org/10.1002/kjm2.12353 PMid:33452732 PMCid:PMC8014750
- Liang
F, Wang X, Shao J, et al. comparison of clinical features on admission
between coronavirus disease 2019 and influenza a among children: a
retrospective study in China. BMC Infect Dis. 2021 Apr 17;21(1):365.
doi: 10.1186/s12879-021-06037-3. https://doi.org/10.1186/s12879-021-06037-3 PMid:33865314 PMCid:PMC8052833
- Asseri
AA, Shati AA, Al-Qahtani SM, et al. Distinctive clinical and laboratory
features of COVID-19 and H1N1 influenza infections among hospitalized
pediatric patients. World J Pediatr. 2021 Jun;17(3):272-279. doi:
10.1007/s12519-021-00432-1. Epub 2021 May 10. https://doi.org/10.1007/s12519-021-00432-1 PMid:33970449 PMCid:PMC8108014
- Siddiqui
M, Gültekingil A, Bakırcı O, Uslu N, Baskın E. Comparison of clinical
features and laboratory findings of coronavirus disease 2019 and
influenza A and B infections in children: a single-center study. Clin
Exp Pediatr. 2021 Jul;64(7):364-369. doi: 10.3345/cep.2021.00066. Epub
2021 May 18. https://doi.org/10.3345/cep.2021.00066 PMid:34015895 PMCid:PMC8255514
- Wald
ER, Schmit KM, Gusland DY. A Pediatric Infectious Disease Perspective
on COVID-19. Clin Infect Dis. 2021 May 4;72(9):1660-1666. doi:
10.1093/cid/ciaa1095. https://doi.org/10.1093/cid/ciaa1095 PMid:32766824 PMCid:PMC7454399
- Kuiken
T, Holmes EC, McCauley J, Rimmelzwaan GF, Williams CS, Grenfell BT.
Host species barriers to influenza virus infections. Science. 2006 Apr
21;312(5772):394-7. doi: 10.1126/science.1122818. https://doi.org/10.1126/science.1122818 PMid:16627737
- Kumlin
U, Olofsson S, Dimock K, Arnberg N. Sialic acid tissue distribution and
influenza virus tropism. Influenza Other Respir Viruses. 2008
Sep;2(5):147-54. doi: 10.1111/j.1750-2659.2008.00051.x. https://doi.org/10.1111/j.1750-2659.2008.00051.x PMid:19453419 PMCid:PMC4941897
- Buonsenso
D, Sali M, Pata D, et al. Children and COVID-19: Microbiological and
immunological insights. Pediatr Pulmonol. 2020 Oct;55(10):2547-2555.
doi: 10.1002/ppul.24978. Epub 2020 Jul 30. https://doi.org/10.1002/ppul.24978 PMid:32710652
- Osman
M, Klopfenstein T, Belfeki N, Gendrin V, Zayet S. A Comparative
Systematic Review of COVID-19 and Influenza. Viruses. 2021 Mar
10;13(3):452. doi: 10.3390/v13030452. https://doi.org/10.3390/v13030452 PMid:33802155 PMCid:PMC8001286
- Pormohammad
A, Ghorbani S, Khatami A, et al. Comparison of influenza type A and B
with COVID-19: A global systematic review and meta-analysis on
clinical, laboratory and radiographic findings. Rev Med Virol. 2021
May;31(3):e2179. doi: 10.1002/rmv.2179.
https://doi.org/10.1002/rmv.2179
- Cobb
NL, Sathe NA, Duan KI, et al. Comparison of Clinical Features and
Outcomes in Critically Ill Patients Hospitalized with COVID-19 versus
Influenza. Ann Am Thorac Soc. 2021 Apr;18(4):632-640. doi:
10.1513/AnnalsATS.202007-805OC. https://doi.org/10.1513/AnnalsATS.202007-805OC PMid:33183067 PMCid:PMC8009008
- Alsohime
F, Temsah MH, Al-Nemri AM, Somily AM, Al-Subaie S. COVID-19 infection
prevalence in pediatric population: Etiology, clinical presentation,
and outcome. J Infect Public Health. 2020 Dec;13(12):1791-1796. doi:
10.1016/j.jiph.2020.10.008. Epub 2020 Oct 20. https://doi.org/10.1016/j.jiph.2020.10.008 PMid:33127335 PMCid:PMC7574780
- Badal
S, Thapa Bajgain K, Badal S, et al. Prevalence, clinical
characteristics, and outcomes of pediatric COVID-19: A systematic
review and meta-analysis. J Clin Virol. 2021 Feb;135:104715. doi:
10.1016/j.jcv.2020.104715. Epub 2020 Dec 8. https://doi.org/10.1016/j.jcv.2020.104715 PMid:33348220 PMCid:PMC7723460
- Ludwig
M, Jacob J, Basedow F, Andersohn F, Walker J. Clinical outcomes and
characteristics of patients hospitalized for Influenza or COVID-19 in
Germany. Int J Infect Dis. 2021 Feb;103:316-322. doi:
10.1016/j.ijid.2020.11.204. Epub 2020 Dec 31. https://doi.org/10.1016/j.ijid.2020.11.204 PMid:33279652 PMCid:PMC7774009
- Fröhlich
GM, De Kraker MEA, Abbas M, Keiser O, et al. Hospital outcomes of
community-acquired COVID-19 versus Influenza: Insights from the Swiss
hospital-based surveillance of Influenza and COVID-19. Euro Surveill.
2022 Jan;27(1):2001848. doi: 10.2807/1560-7917.ES.2022.27.1.2001848. https://doi.org/10.2807/1560-7917.ES.2022.27.1.2001848 PMid:34991775 PMCid:PMC8739338
- Talbot
HK, Martin ET, Gaglani M, et al. Coronavirus disease 2019 (COVID-19)
Versus Influenza in Hospitalized Adult Patients in the United States:
Differences in Demographic and Severity Indicators. Clin Infect Dis.
2021 Dec 16;73(12):2240-2247. doi: 10.1093/cid/ciab123. https://doi.org/10.1093/cid/ciab123 PMid:34050659 PMCid:PMC8195096
- Jia
Z, Yan X, Gao L, et al. Comparison of Clinical Characteristics Among
COVID-19 and Non-COVID-19 Pediatric Pneumonias: A Multicenter
Cross-Sectional Study. Front Cell Infect Microbiol. 2021 Jul
1;11:663884. doi: 10.3389/fcimb.2021.663884.. https://doi.org/10.3389/fcimb.2021.663884 PMid:34277466 PMCid:PMC8281119
- Smith
C, Odd D, Harwood R, et al. Deaths in children and young people in
England after SARS-CoV-2 infection during the first pandemic year. Nat
Med. 2021 Nov 11. doi: 10.1038/s41591-021-01578-1. Epub ahead of print.
https://doi.org/10.1038/s41591-021-01578-1 PMid:34764489
- Bhuiyan
MU, Stiboy E, Hassan MZ, et al. Epidemiology of COVID-19 infection in
young children under five years: A systematic review and meta-analysis.
Vaccine. 2021 Jan 22;39(4):667-677. doi: 10.1016/j.vaccine.2020.11.078.
Epub 2020 Dec 5. https://doi.org/10.1016/j.vaccine.2020.11.078 PMid:33342635 PMCid:PMC7833125
- Stals
MAM, Grootenboers MJJH, van Guldener C, et al. risk of thrombotic
complications in Influenza versus COVID-19 hospitalized patients. Res
Pract Thromb Haemost. 2021 Apr 8;5(3):412-420. doi: 10.1002/rth2.12496.
https://doi.org/10.1002/rth2.12496 PMid:33821230 PMCid:PMC8014477
- Birkelo
BC, Parr SK, Perkins AM, et al. Comparison of COVID-19 versus Influenza
on the incidence, features, and recovery from acute kidney injury in
hospitalized United States Veterans. Kidney Int. 2021
Oct;100(4):894-905. doi: 10.1016/j.kint.2021.05.029. Epub 2021 Jun 7. https://doi.org/10.1016/j.kint.2021.05.029 PMid:34111501 PMCid:PMC8183091
- Bogunovic
D, Merad M. Children and SARS-CoV-2. Cell Host Microbe. 2021 Jul
14;29(7):1040-1042. doi: 10.1016/j.chom.2021.06.015. https://doi.org/10.1016/j.chom.2021.06.015 PMid:34265242 PMCid:PMC8279573
- Oualha
M, Bendavid M, Berteloot L, et al. Severe and fatal forms of COVID-19
in children. Arch Pediatr. 2020 Jul;27(5):235-238. doi:
10.1016/j.arcped.2020.05.010. Epub 2020 Jun 4. https://doi.org/10.1016/j.arcped.2020.05.010 PMid:32518045 PMCid:PMC7269941
- Duarte-Salles
T, Vizcaya D, Pistillo A, et al. Thirty-Day Outcomes of Children and
Adolescents With COVID-19: An International Experience. Pediatrics.
2021 Sep;148(3):e2020042929. doi: 10.1542/peds.2020-042929. Epub 2021
May 28. https://doi.org/10.1542/peds.2020-042929 PMid:34049958
- Wei
JS. How lethal is SARS-CoV-2 pneumonia when compared with respiratory
syncytial virus and Influenza in young children? Aust J Gen Pract. 2020
Oct;49(10):683-686. doi: 10.31128/AJGP-04-20-5357. https://doi.org/10.31128/AJGP-04-20-5357 PMid:33015684
- Piroth
L, Cottenet J, Mariet AS, et al. Comparison of the characteristics,
morbidity, and mortality of COVID-19 and seasonal Influenza: a
nationwide, population-based retrospective cohort study. Lancet Respir
Med. 2021 Mar;9(3):251-259. doi: 10.1016/S2213-2600(20)30527-0. Epub
2020 Dec 17. https://doi.org/10.1016/S2213-2600(20)30527-0 PMID:33341155; PMCID:PMC7832247
- Pata
D, Valentini P, De Rose C, De Santis R, Morello R, Buonsenso D. Chest
Computed Tomography and Lung Ultrasound Findings in COVID-19 Pneumonia:
A Pocket Review for Non-radiologists. Front Med (Lausanne). 2020 Jun
26;7:375. doi:10.3389/fmed.2020.00375. https://doi.org/10.3389/fmed.2020.00375 PMid:32671086 PMCid:PMC7332745
- Buonsenso
D, Pata D, De Rose C, et al. Clinical application of Lung Ultrasound
for the management of pregnant women with suspicion of COVID-19: a
review of literature. J.Gynecol. Obstet. 2020, 32, N.3. doi:
10.36129/jog.32.03.04 https://doi.org/10.36129/jog.32.03.04
- Buonsenso
D, Pata D, Chiaretti A. COVID-19 outbreak: less stethoscope, more
ultrasound. Lancet Respir Med. 2020 May;8(5):e27. doi:
10.1016/S2213-2600(20)30120-X. Epub 2020 Mar 20. https://doi.org/10.1016/S2213-2600(20)30120-X PMID: 32203708 PMCID: PMC7104316.
[TOP]






