Viral hepatitis reactivation in CML after TKI treatment is not frequent, and the mechanism is uncertain due to the limited number of case reports.[2-9] Kong et al reported an analysis of 2278 CML patients, and 143 (6.3%) patients were HBV carriers or had previous HBV infection, and HBV reactivation occurred in 23.1% of the patients.[4] Other studies reported HBV reactivation rates ranging from 0% to 26.3%.[7,10] Viral reactivation is associated with an immunosuppressive and cytotoxic condition induced by TKI, which affects the production, modulation, and proliferation of immune system cells.[2,3,11] In some cases, hepatitis flares occurred after the patients achieved a complete molecular or cytogenetic response, suggesting that the flare may be due to restoration of the immune response after TKI treatment.[2]
We aimed to investigate the prevalence of viral hepatitis in CML patients at diagnosis and reactivation during treatment. This study was conducted at Centro de Hematologia e Hemoterapia, Universidade Estadual de Campinas (Hemocentro-Unicamp) and was approved by the local institutional review board (CAAE: 18742919.7.0000.5404). Informed consent was obtained from all subjects currently in treatment. Clinical and laboratory data were collected from medical records, including age, gender, disease phase (chronic, accelerated, or blastic), serological status of hepatitis B and C at the time of CML diagnosis, date of initiation of TKI treatment, type of TKI used, occurrence of hepatotoxicity, and viral hepatitis B and C reactivation during the treatment. Prospectively, we evaluated the serological status of HBV and HCV collected at study entry from patients in follow-up who had been treated with TKI for more than 5 years. Serologies were performed using enzyme-linked immunosorbent assay (ELISA). We also collected data about hepatic toxicity during TKI treatment. Patients with a diagnosis of viral hepatitis were referred to the Infectious Diseases outpatient clinics for treatment and follow-up.
Hepatitis B reactivation was defined as the development of hepatitis with serum alanine transaminase (ALT) levels greater than three times the upper limit of normal or an absolute increase of 100 IU/L, associated with a demonstrable increase in HBV DNA of at least 10 times, in patients whose infection was previously inactive or resolved.[9] Hepatic toxicity was graded according to CTCAE version 5.0.
We analyzed clinical and laboratory data from 254 CML patients diagnosed between 1988 and 2022, with a median follow-up of 6 years; 150 (59.1%) were male, and the median age at CML diagnosis was 48 years (range 4-86 years). In this cohort, 189 were on treatment, and 59 have died. Six patients lost follow-up. The cut-off date for this analysis was 2022. Patients' characteristics are presented in Table 1. As a risk factor for viral hepatitis, prior blood transfusion was identified in 8 (3.1%) patients. Hepatitis B vaccination history was unknown. Patients were initially treated with imatinib (n=239; 94.1%), nilotinib (n=8; 3.1%), bosutinib (n=6; 2.4%), and dasatinib (n=1; 0.4%). 89 pts (35%) switched to second-line therapy, using dasatinib (n=59; 66.3%), nilotinib (n=22; 24.7%), imatinib (n=7; 7.9%), and bosutinib (n=1; 1.1%). Twenty-five (9.8%) patients received a third-line TKI: nilotinib (n=11; 44%), dasatinib (n=8; 32%), imatinib (n=5; 20%), and ponatinib (n=1; 4%). Six patients (2.4%) received a fourth-line TKI: 3 (50%) received dasatinib, 2 (33.3%) received imatinib, and 1 (16.7%) received nilotinib.
At CML diagnosis, hepatitis B and C serology results were available in 190 (74.8%) and 177 (69.7%) patients, respectively. In the follow-up, 41 (16.1%) additional patients were tested for hepatitis B and 43 (16.9%) for hepatitis C. Therefore, the serological status of hepatitis B during TKI treatment was determined in 231 (90.9%) patients, and hepatitis C status in 220 (86.6%) patients. No cases of HCV were detected at diagnosis or follow-up. Among the 190 patients with hepatitis B serology available at the time of CML diagnosis, 102 remained in follow-up, and 50/102 (49%) underwent a new test. Among the 177 patients tested for hepatitis C at diagnosis, 92 remained in follow-up, and 42/92 (45.6%) collected a new test.
Hepatotoxicity during TKI treatment occurred in 69 (27.2%) patients, with grade 1 in 40 (58%), grade 2 in 15 (21.7%), grade 3 in 7 (10.1%), and grade 4 in 7 (10.1%). The median time between CML diagnosis and hepatotoxicity was one year. Elevations in hepatic transaminases have been reported frequently with TKIs.[12] Forty-three (62.3%) cases occurred during imatinib therapy, including one associated with concomitant use of tibolone and another with isoniazid. Two of the imatinib-related cases recurred during dasatinib treatment as a second-line therapy. Eleven (15.9%) cases were observed during nilotinib therapy, 10 (14.5%) during dasatinib, and 5 (7.2%) during bosutinib. In the BFORE trial, the frequency of increased alanine aminotransferase and aspartate aminotransferase was 30.6% and 22.8%, respectively, in newly diagnosed patients treated with bosutinib.[13] Fifty-one (73.9%) cases emerged during first-line therapy, 13 (18.8%) during second-line, 3 (4.3%) during third-line, and 2 (2.9%) during both first- and second-line treatments. Among all cases, seven experienced a new episode, with two presenting toxicity in both first- and second-line therapies, initially with imatinib followed by dasatinib, as previously exposed.
A total of 17 (6.7%) CML cases were anti-HBsAg positive and anti-HBcAg positive, indicating natural immunity acquisition after HBV infection, and 4 (1.6%) cases were anti-HBsAg positive and anti-HBcAg and HBsAg negative.
At diagnosis, there were three cases (1.2%) of active chronic hepatitis B. The first was a 52-year-old male patient with HbsAg, anti-HBcAg, and anti-HBeAg positive. This patient underwent treatment with lamivudine 150mg/day for 3 years and had HbsAg seroreversion, but died months later due to complications of an allogeneic hematopoietic stem cell transplant (HSCT). The second case was a 41-year-old male patient who was HBsAg, HBeAg, anti-HBcAg, and anti-HbeAg positive, with a positive PCR test for HBV. He initiated treatment with tenofovir 300 mg/day and is currently using entecavir 0.5 mg/day. The patient has experienced anti-HbeAg seroreversion, with HBV PCR still detectable but not quantifiable. The third case was a 52-year-old female patient with positive HBsAg, anti-HBcAg, and anti-HbeAg, and an undetectable HBV PCR. She is under treatment with entecavir 0.5 mg/day and did not have seroreversion. None had hepatitis B reactivation during TKI treatment.
We also identified three cases (1.2%) with anti-HBcAg-positive and anti-HBsAg/HBsAg-negative at CML diagnosis. The first case was a 79-year-old male patient who underwent treatment with imatinib, switched to dasatinib after 2 years due to resistance and died 5 years after diagnosis from metastatic lung cancer. Despite not receiving antiviral prophylaxis, this patient had no hepatitis reactivation. The second case was a 67-year-old male patient with CML diagnosis in 2012. He was previously treated with imatinib and dasatinib, and is currently using nilotinib. This patient has been followed by the infectology department with serology and liver enzyme tests every 6 months. The last case was a 56-year-old female with positive anti-HBcAg and anti-HBeAg, and negative anti-HBsAg (titer < 10 mIU/ml)/HBsAg/HBeAg at CML diagnosis in June 2022. She was treated with preemptive treatment with entecavir 0.5 mg/day before starting imatinib.
There is limited evidence that antibodies against hepatitis B surface antigen (anti-HBsAg) are protective against hepatitis B virus (HBV) reactivation.[14] Immunosuppression caused by other treatments, such as rituximab, may induce hepatitis B reactivation in these cases.[15,16] However, data are limited to make recommendations on management and prophylaxis in this situation.
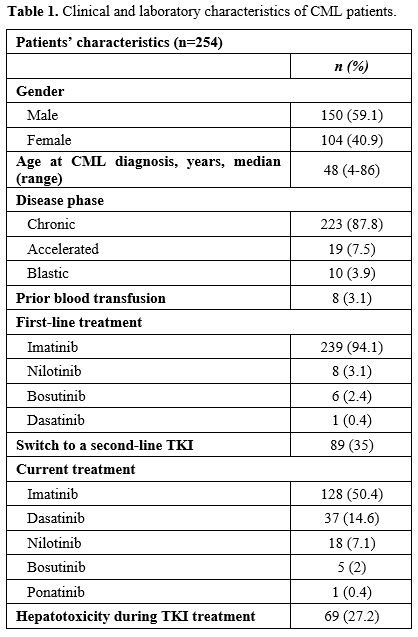
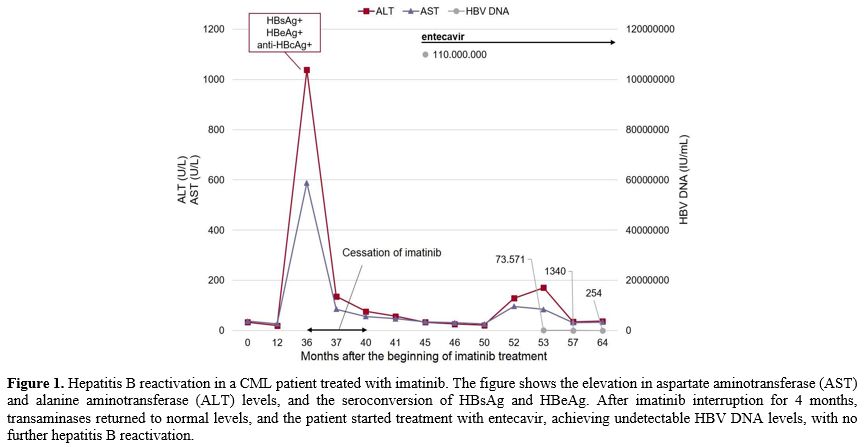
There was one case of HBV reactivation in a patient treated with imatinib for 3 years. This patient was previously treated at another hospital, with an unknown serological status at the time of CML diagnosis. Imatinib was interrupted for 4 months, and the patient was treated with entecavir. During imatinib interruption, the patient maintained a major molecular response (Figure 1). When liver enzymes returned to normal levels, imatinib was reintroduced at a lower dose (300mg/day), with no further toxicity.
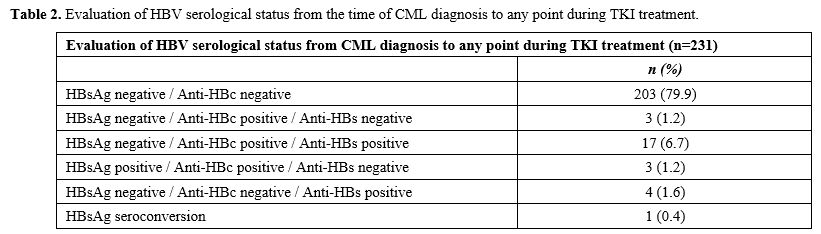 |
Table 2. Evaluation of HBV serological status from the time of CML diagnosis to any point during TKI treatment. |
Although the medical literature on TKI-induced hepatitis B reactivation remains limited to case reports and case series, it is estimated that the risk of HBV reactivation is moderate in HBsAg-positive patients (1%-10%) and in HBsAg-negative/anti-HBcAg-positive patients (1%).[14] Ikeda et al. reported a fatal case of hepatitis B virus reactivation in a CML patient using imatinib in a 54-year-old man with no prior liver dysfunction.[8] A recent search in the literature, from 2001 to 2022, identified 15 cases of hepatitis B reactivation in CML patients treated with TKI.[9]
Data about hepatitis C reactivation induced by TKI are even more limited.[17] In the present study, we did not detect any cases of HCV among CML patients, although HCV incidence in the Southeast region in Brazil is 8.7 new cases/100,000 inhabitants.[18]
The European Association for the Study of the Liver (EASL) recommends that all candidates for chemotherapy and immunosuppressive therapy should be tested for HBV markers prior to immunosuppression, and that preventive treatment with nucleoside/nucleotide analogs should be given to patients undergoing chemotherapy or immunosuppressive therapy.[19] All HBsAg-positive patients should receive entecavir or tenofovir disoproxil fumarate or tenofovir alafenamide as treatment or prophylaxis, and HBsAg-negative/anti-HBc-positive subjects should receive anti-HBV prophylaxis if they are at high risk of HBV reactivation.[19] Based on our findings and the current recommendations, the serological status of hepatitis B and C should be screened in all CML patients at diagnosis to identify active chronic infection or previous exposure. Patients with HBV infection at diagnosis should receive entecavir and TKI as soon as hepatic enzymes return to grade 1. All HBsAg-positive patients should receive antiviral treatment. For reactivation during TKI therapy, hepatitis should be treated, and TKI should be interrupted, resuming when liver enzymes are grade 1.
References
- Delord M, Foulonb S, Cayuelad JM, Rousselotf P,
Bonastreb J. The rising prevalence of chronic myeloid leukemia in
France. Leukemia Research June 2018; vol. 69. https://doi.org/10.1016/j.leukres.2018.04.008 PMid:29734071
- Lai
GM, Yan SL, Chang CS, Tsai CY. Hepatitis B reactivation in chronic
myeloid leukemia patients receiving tyrosine kinase inhibitors. World J
Gastroenterol 2013; 19(8): 1318-1321. https://doi.org/10.3748/wjg.v19.i8.1318 PMid:23483799 PMCid:PMC3587491
- Inayat
F, Song F, Ali NS, et al. Hepatitis B virus reactivation following
imatinib therapy: A comparative review of 9 cases. Journal of Oncology
Pharmacy Practice. 2019;25(6):1500-1508. https://doi.org/10.1177/1078155218790337 PMid:30079802
- Kong
JH, Jang JY, Ko TH, Kang SH, Kim Y. High rate of hepatitis B
reactivation during tyrosine kinase inhibitor treatment among patients
with chronic myeloid leukemia in Korea. Blood Res. 2022 Dec
31;57(4):290-293. https://doi.org/10.5045/br.2022.2022099 PMid:36419239 PMCid:PMC9812728
- Benjamini
O, Zlotnick M, Ribakovsky E, et al. Evaluation of the risk of hepatitis
B reactivation among patients with chronic myeloid leukemia treated
with tyrosine kinase inhibitors. Blood. 2016;128(22):5429. https://doi.org/10.1182/blood.V128.22.5429.5429
- Sora
F, Ponziani FR, Laurenti L, et al. Low risk of hepatitis B virus
reactivation in patients with resolved infection and chronic B myeloid
leukemia treated with tyrosine kinase inhibitors. Leuk Lymphoma.
2017;58(4):993-995. https://doi.org/10.1080/10428194.2016.1219906 PMid:27546591
- Orlandi
EM, Elena C, Bono E. Risk of hepatitis B reactivation under treatment
with tyrosine-kinase inhibitors for chronic myeloid leukemia. Leuk
Lymphoma. 2017;58(7):1764-1766 https://doi.org/10.1080/10428194.2016.1260127 PMid:27892750
- Ikeda
K, Shiga Y, Takahashi A, Kai T, Kimura H, Takeyama K, Noji H, Ogawa K,
Nakamura A, Ohira H, Sato Y, Maruyama Y. Fatal hepatitis B virus
reactivation in a chronic myeloid leukemia patient during imatinib
mesylate treatment. Leuk Lymphoma. 2006 Jan;47(1):155-7.
https://doi.org/10.1080/14639230500236818 PMid:16321842
- Aldapt
MB, Al-Mashdali AF, Obeidat K, Chandra P, Yassin M. Viral infections
and incidence of reactivations in chronic myeloid leukemia patients.
Oncology. 2023 Oct 17. https://doi.org/10.1159/000534266 PMid:37848004 PMCid:PMC10994575
- Wang
YH, Liang JD, Sheng WH, Tien FM, Chen CY, Tien HF. Hepatitis B
reactivation during treatment of tyrosine kinase inhibitors-Experience
in 142 adult patients with chronic myeloid leukemia. Leuk Res. 2019
Jun;81:95-97 https://doi.org/10.1016/j.leukres.2019.05.001 PMid:31075669
- de
Lavallade H, Khoder A, Hart M, Sarvaria A, Sekine T, Alsuliman A, et
al. Tyrosine kinase inhibitors impair B-cell immune responses in CML
through off-target inhibition of kinases important for cell signaling.
Blood. 2013 Jul;122(2):227-38. https://doi.org/10.1182/blood-2012-11-465039 PMid:23719297 PMCid:PMC5726329
- Steegmann,
J., Baccarani, M., Breccia, M. et al. European LeukemiaNet
recommendations for the management and avoidance of adverse events of
treatment in chronic myeloid leukaemia. Leukemia 30, 1648-1671 (2016). https://doi.org/10.1038/leu.2016.104 PMid:27121688 PMCid:PMC4991363
- Cortes
JE, Gambacorti-Passerini C, Deininger MW, Mauro MJ, Chuah C, Kim DW, et
al. Bosutinib versus imatinib for newly diagnosed chronic myeloid
leukemia: results from the randomized BFORE trial. J Clin Oncol.
2018;36:231-7. https://doi.org/10.1200/JCO.2017.74.7162 PMid:29091516 PMCid:PMC5966023
- Pattullo
V. Hepatitis B reactivation in the setting of chemotherapy and
immunosuppression - prevention is better than cure. World J Hepatol.
2015;7(7):954-967. https://doi.org/10.4254/wjh.v7.i7.954 PMid:25954478 PMCid:PMC4419099
- Niitsu
N, Hagiwara Y, Tanae K, Kohri M, Takahashi N (2010) Prospective
analysis of hepatitis B virus reactivation in patients with diffuse
large B-cell lymphoma after rituximab combination chemotherapy. J Clin
Oncol 28(34):5097-5100. https://doi.org/10.1200/JCO.2010.29.7531 PMid:20837949
- Pei,
SN., Ma, MC., Wang, MC. et al. Analysis of hepatitis B surface antibody
titers in B cell lymphoma patients after rituximab therapy. Ann Hematol
91, 1007-1012 (2012). https://doi.org/10.1007/s00277-012-1405-6 PMid:22273839
- Yazici
O, Şendur MAN, Aksoy S. Hepatitis C virus reactivation in cancer
patients in the era of targeted therapies. World J Gastroenterol 2014;
20(22): 6716-6724. https://doi.org/10.3748/wjg.v20.i22.6716
- Hanus
JS, Ceretta LB, Simões PW, Tuon L. Incidence of hepatitis C in Brazil.
Rev Soc Bras Med Trop [Internet]. 2015Nov;48(6):665-73. https://doi.org/10.1590/0037-8682-0230-2015 PMid:26676490
- EASL
2017 Clinical Practice Guidelines on the management of hepatitis B
virus infection. Journal of Hepatology 2017; vol. 67 j 370-398. https://doi.org/10.1016/j.jhep.2017.03.021 PMid:28427875