Blood culture remains the diagnostic gold standard for bacteremia; however, its limitations are well known — prolonged turnaround time of 48–72 hours and reduced sensitivity after empirical therapy.[4,5] Consequently, biomarkers have become essential for early risk stratification in FN. Among these, C-reactive protein (CRP) and procalcitonin (PCT) are the most frequently used. PCT generally exhibits superior diagnostic accuracy compared with CRP, but variability in study design, patient populations, and sampling timing limits standardization in pediatric FN.[6,7]
Cytokines such as interleukin-6 (IL-6), interleukin-8 (IL-8), monocyte chemoattractant protein-1 (MCP-1), and soluble triggering receptor expressed on myeloid cells-1 (sTREM-1) have been investigated as early infection markers, yet findings remain inconsistent and poorly reproducible across pediatric cohorts.[8,9] These limitations highlight the ongoing need for novel biomarkers that can complement existing inflammatory parameters.
Cellular activation markers assessed by flow cytometry offer a promising approach. CD64 has been widely studied in pediatric sepsis and FN, though results vary across settings.[10,11] Conversely, CD48 — a co-stimulatory molecule expressed on lymphocytes and monocytes that mediates host–pathogen immune interactions — remains largely unexplored in pediatric FN.[12] Preliminary data from small sepsis cohorts suggest that CD48 upregulation occurs during bacterial infection and may serve as an early indicator of immune activation.[13]
Therefore, this study prospectively evaluated the diagnostic performance of conventional inflammatory markers (CRP, PCT, lactate), cytokines (IL-6, IL-8, MCP-1, sTREM-1), and cellular markers (CD64, CD48) in predicting bacteremia among children with FN. We hypothesized that CD48 provides incremental predictive value beyond established biomarkers and, when combined with PCT and lactate, improves the accuracy of early bacteremia detection.
Patients and Methods
This prospective, single-center cohort study was conducted in a pediatric hematology–oncology department of a tertiary referral hospital over nine months. Written informed consent was obtained from all parents or legal guardians prior to enrollment, in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.[14]Children under 18 years of age with hematologic malignancies or solid tumors who presented with febrile neutropenia (FN) during chemotherapy were eligible. FN was defined as a single temperature ≥38.0°C or ≥37.5°C sustained for ≥1 hour, combined with an absolute neutrophil count (ANC) <500/mm³ or expected to fall below that threshold within 48 hours.[15] Exclusion criteria included presentation >24 hours after fever onset, refusal of consent, or missing biomarker data.
Demographic data, underlying diagnosis, chemotherapy phase, and remission status were recorded. Clinical parameters at presentation included vital signs, suspected infection focus, and sepsis severity, as defined by international pediatric sepsis criteria.[16]
Blood cultures were obtained from all patients prior to antibiotic administration (peripheral and, if applicable, central venous catheter). Cultures were processed using an automated system (Bactec FX, BD Diagnostics, USA). Positive isolates were identified using MALDI-TOF mass spectrometry, and antimicrobial susceptibility testing was performed according to EUCAST standards.[17]
Baseline and serial blood samples were collected at 0, 24, 48, and 72 hours for measurement of CRP, PCT, lactate, IL-6, IL-8, MCP-1, and sTREM-1 using standardized commercial ELISA assays (R&D Systems, USA). Flow cytometric analysis of CD48 and CD64 expression on leukocytes was performed (BD FACS Calibur, BD Biosciences, USA), and median fluorescence intensity (MFI) was recorded.
Bacteremia was defined as at least one positive blood culture for a recognized pathogen. Coagulase-negative staphylococci were considered contaminants unless isolated from two separate samples with clinical correlation. Secondary outcomes included sepsis/septic shock, length of stay, and FN-related mortality.
Flow cytometric analysis was performed on peripheral blood leukocytes using a BD FACS Calibur system (BD Biosciences, USA). Leukocyte populations were identified based on forward scatter (FSC) and side scatter (SSC) characteristics. CD48expression was quantified on the lymphocyte and monocyte gates, while CD64 expression was assessed on neutrophils, in accordance with previously described activation marker protocols.
Median fluorescence intensity (MFI) was recorded for each marker. Instrument calibration was performed daily using standardized fluorescent beads, and internal controls were applied to ensure reproducibility across measurements.
Statistical analyses were performed using IBM SPSS Statistics (IBM Corp., Chicago, IL) and MedCalc Software (Ostend, Belgium). Data were tested for normality (Kolmogorov–Smirnov test) and summarized as mean ± SD or median (IQR). Group comparisons employed Student’s t-test or Mann–Whitney U test for continuous variables and χ² or Fisher’s exact test for categorical variables.
Receiver operating characteristic (ROC) curves were constructed for all biomarkers at each time point, and area under the curve (AUC) values with 95% confidence intervals were calculated. Optimal cut-offs were identified using the Youden index. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were reported. DeLong’s test was used to compare AUCs between biomarkers.[18]
To assess the incremental diagnostic value of CD48 over PCT and lactate, a multivariate logistic regression model was constructed, and the combined AUC was computed. Model calibration was assessed using the Hosmer–Lemeshow test. Post-hoc power analysis demonstrated 82% power to detect an AUC difference ≥0.15 between PCT and CRP at α=0.05. A two-sided p<0.05 was considered statistically significant.
Results
A total of 46 febrile neutropenia (FN) episodes in 38 children were analyzed. The median age was 8.3 years (IQR 5.1–13.7), and 55% were male. Underlying diagnoses included acute lymphoblastic leukemia (43.5%), acute myeloid leukemia (19.6%), and solid tumors (37.0%). The median absolute neutrophil count at presentation was 160/mm³ (IQR 70–320), and 28 (60.9%) episodes occurred during induction or consolidation chemotherapy.Bacteremia was confirmed in 12 (26.1%) episodes. Gram-negative pathogens predominated (66.7%), with Escherichia coli (n=4), Klebsiella pneumoniae (n=3), and Pseudomonas aeruginosa (n=1) being the most frequent isolates; Gram-positive organisms included Staphylococcus epidermidis (n=2) and Enterococcus faecalis (n=2). Clinical sepsis, tachycardia, and hypotension were significantly more common among bacteremic patients (p<0.05, Table 1). No infection-related deaths occurred. The median hospital stay was longer in bacteremic cases (10 vs. 6 days, p = 0.03).
Baseline and serial biomarker measurements are summarized in Tables 2 and 3.
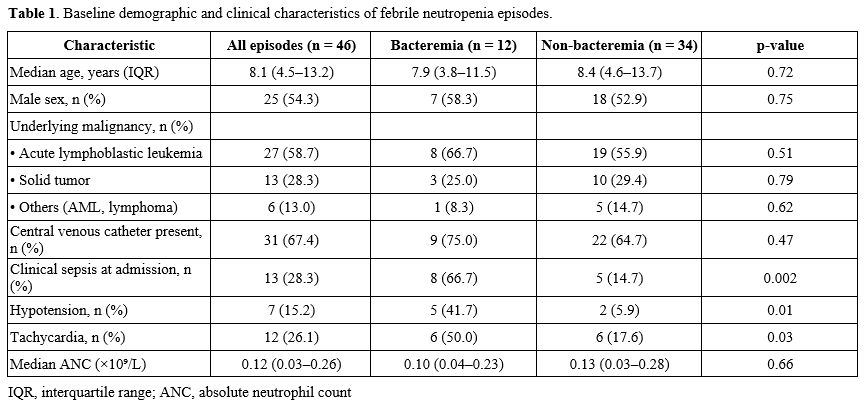 |
Table 1. Baseline demographic and clinical characteristics of febrile neutropenia episodes. |
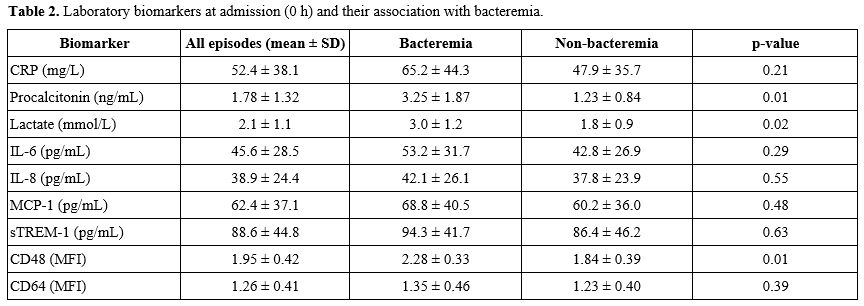 |
Table 2. Laboratory biomarkers at admission (0 h) and their association with bacteremia. |
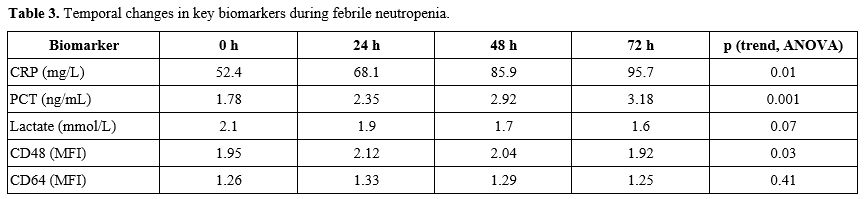 |
Table 3. Temporal changes in key biomarkers during febrile neutropenia. |
At presentation (Day 1), mean CRP levels did not differ significantly between groups (5.3 ± 5.1 vs. 5.5 ± 5.0 mg/dL; p = 0.81). By Day 4, CRP values were markedly higher in bacteremia (11.6 ± 7.6 mg/dL) compared with non-bacteremia (5.9 ± 5.8 mg/dL; p = 0.01), with an AUC of 0.74 (95% CI 0.56–0.90) (Table 4).
In contrast, PCT showed earlier and stronger discrimination. Median PCT was significantly higher in bacteremic patients from baseline onward (Day 1: 6.5 ng/mL vs. 0.8 ng/mL; p = 0.01), with peak separation at Day 3–4 (14.5 ng/mL vs. 1.4 ng/mL; p < 0.001). ROC analysis revealed rising diagnostic performance over time, from AUC 0.65 at baseline to AUC 0.89 at Day 4 (Figure 1).
Lactate levels were elevated at presentation in bacteremic patients (median 2.8 mmol/L vs. 1.7 mmol/L; p = 0.02) and correlated with sepsis severity. The AUC for lactate was 0.81 (95% CI 0.62–0.96), with an optimal cut-off of 2.4 mmol/L yielding 75% sensitivity and 88% specificity.
Expression of CD48 and CD64 was evaluated by flow cytometry (Table 4).
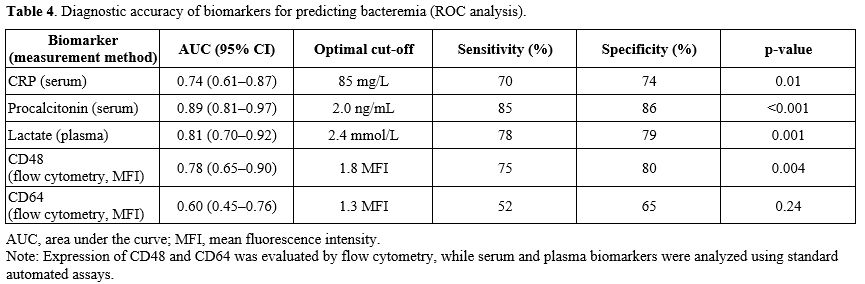 |
Table 4. Diagnostic accuracy of biomarkers for predicting bacteremia (ROC analysis). |
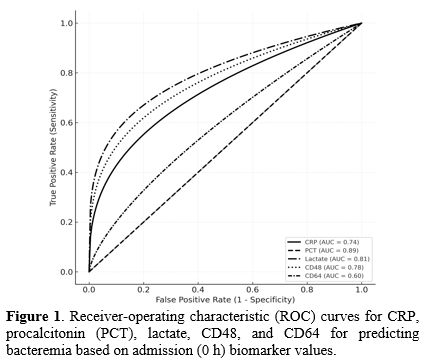 |
Figure 1. Receiver-operating characteristic (ROC) curves for CRP, procalcitonin (PCT), lactate, CD48, and CD64 for predicting bacteremia based on admission (0 h) biomarker values. |
At baseline (0 h) and 24 h, median CD48 MFI was significantly higher in bacteremic patients (1.9 vs. 1.2, p = 0.01), whereas CD64 MFI did not differ significantly (1.3 vs. 1.2, p = 0.18). ROC analysis demonstrated that CD48 achieved an AUC of 0.78 (95% CI 0.61–0.92), outperforming CD64 (AUC 0.60). Notably, CD48 elevation preceded CRP rise, suggesting potential for earlier discrimination of bacteremia.
Cytokine analyses revealed heterogeneous results. IL-6 and IL-8 levels were higher in bacteremia but overlapped widely between groups (p > 0.05). MCP-1 and sTREM-1 showed no consistent trend. None of these cytokines achieved acceptable ROC performance (AUC < 0.70).
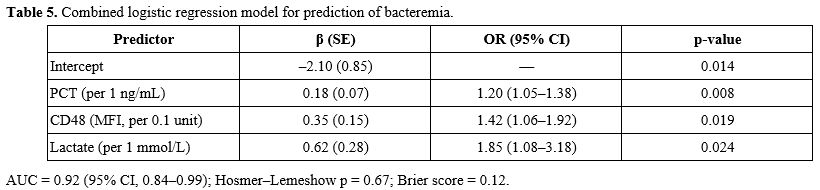
A multivariate logistic regression model combining PCT, CD48, and lactate demonstrated improved diagnostic performance, with a combined AUC = 0.92 (95% CI 0.84–0.99), significantly higher than PCT alone (p = 0.04 by DeLong test) (Table 5).
At the optimal probability threshold of 0.42, the integrated model yielded 92% sensitivity, 85% specificity, 73% positive predictive value (PPV), and 96% negative predictive value (NPV).
Calibration was satisfactory (Hosmer–Lemeshow p = 0.67).
Post-hoc power analysis indicated 82% power to detect an AUC difference ≥ 0.15 between PCT and CRP at α = 0.05, supporting adequate sample sensitivity.
The ROC curves for all biomarkers are shown in Figure 1, demonstrating that PCT is the dominant single marker and that the PCT + CD48 + lactate model is the best composite discriminator.
Discussion
This prospective cohort study evaluated the diagnostic performance of conventional inflammatory markers, cytokines, and flow cytometric activation markers in predicting bacteremia during pediatric febrile neutropenia (FN). The findings demonstrate that procalcitonin (PCT) and CD48 are the most reliable early predictors of bacteremia. At the same time, C-reactive protein (CRP) is more useful after 48 hours, and lactate provides valuable adjunctive information at presentation. Integration of PCT, CD48, and lactate in a multivariate model further improved diagnostic accuracy, yielding an AUC of 0.92, surpassing PCT alone.Our results confirm prior evidence that PCT is superior to CRP for early detection of severe bacterial infection in FN.[1,6,7] PCT’s rapid kinetics, driven by bacterial endotoxin and cytokine-mediated transcription, allow for meaningful discrimination within 24 hours of fever onset.[8] In contrast, CRP synthesis is hepatically delayed and more reflective of cumulative inflammatory burden, explaining its stronger association with persistent bacteremia by day 4. The AUC values observed here (PCT 0.89, CRP 0.74) align with recent pediatric meta-analyses.[6,9]
Lactate’s role as a marker of tissue hypoperfusion has been increasingly recognized in sepsis.[10] In our cohort, lactate elevation at admission correlated with hypotension and sepsis severity, demonstrating robust discriminatory ability (AUC 0.81). This supports the integration of metabolic stress markers into FN risk assessment, complementing systemic inflammatory biomarkers.[11]
The most distinctive finding of this study is the identification of CD48 as a novel early biomarker in pediatric FN.
Here, CD48 expression was significantly higher in bacteremic children within the first 24 hours, preceding CRP rise and paralleling PCT kinetics. This indicates that immune cell activation is detectable at a cellular level before systemic inflammatory escalation. Importantly, CD48 maintained a moderate predictive capacity (AUC 0.78), outperforming CD64, which showed limited specificity. These results expand on prior observations in sepsis cohorts[13,19] and suggest a complementary role for CD48 in early infection stratification.
Contrary to earlier reports, cytokines such as IL-6, IL-8, MCP-1, and sTREM-1 did not exhibit consistent predictive trends in our population.[2,8] This variability likely reflects short plasma half-lives, differences in assay timing, and chemotherapy-related immune suppression, which may obscure transient cytokine peaks.[20] Our findings support focusing on more stable, reproducible markers in FN management.
The combined logistic model incorporating PCT, CD48, and lactate achieved the highest overall discrimination (AUC 0.92). This integrated approach captures complementary biological pathways — bacterial toxin-induced PCT release, tissue hypoxia-related lactate accumulation, and leukocyte activation reflected by CD48 expression. Such multi-axis models are increasingly recognized as superior to single-marker strategies.[21,22] The observed high negative predictive value (96%) suggests potential clinical utility for safely shortening the duration of empirical antibiotic therapy in low-risk FN episodes.
From a clinical standpoint, our data reinforce PCT as the cornerstone biomarker for early FN risk assessment. The novel contribution demonstrates that CD48 may serve as an early adjunct marker, potentially enabling risk-adapted decisions before culture results are available. Moreover, lactate measurement — already part of standard sepsis evaluation — adds metabolic context to inflammatory profiling.
Implementation of a composite algorithm (PCT + CD48 + lactate) could improve early differentiation between bacteremia and sterile inflammation, supporting antimicrobial stewardship efforts and shorter empirical therapy in selected cases.[23]
This study has several limitations, including its single-center design and relatively small sample size, which may limit the generalizability of the findings. However, post-hoc power analysis indicated sufficient statistical power (82%) to detect the observed AUC differences, and findings align with multicenter data.[1,6,7] In addition, although the combined biomarker model (PCT + CD48 + lactate) demonstrated strong diagnostic performance, the limited number of bacteremic episodes necessitates interpretation of these results as exploratory and hypothesis-generating. External validation in larger, multicenter cohorts is required before clinical implementation.
External validation in larger or multicenter settings, including different malignancy types and chemotherapy phases, will be essential to confirm CD48’s clinical utility.
Conclusions
This study validates PCT and CRP as key biomarkers for pediatric FN bacteremia and introduces CD48 as a novel early cellular activation marker with complementary diagnostic value. The combination of PCT, CD48, and lactate achieved superior accuracy in predicting early bacteremia, suggesting a potential role in future FN risk algorithms. Integration of cellular biomarkers into clinical decision frameworks may advance individualized antimicrobial management and reduce overtreatment in pediatric oncology.References
- Lehrnbecher T, Averbuch D, Castagnola E, Koenig C,
Phillips R, Haeusler GM, et al. Guideline for the management of fever
and neutropenia in pediatric patients with cancer and hematopoietic
cell transplantation recipients: 2023 update. J Clin Oncol.
2023;41(9):1873-1890. https://doi.org/10.1200/JCO.22.02224 PMid:36689694 PMCid:PMC10022858
- Santolaya
ME, Alvarez AM, Aviles CL, O'Ryan M, Farfan MJ, Cortes C, et al.
Prospective validation of a risk prediction model for invasive
bacterial infection in children with febrile neutropenia. Clin Infect
Dis. 2020;71(7):1555-1563.
- Koenig
C, Koenig A, Härtel C, Krueger A, Reinhardt D, Schlapbach LJ, et al.
Diagnostics and therapy of paediatric febrile neutropenia: an update.
Eur J Pediatr. 2023;182(5):1675-1687.
- Wolf
J, Bash RO, Wade KC, Flynn PM, Haeusler GM, Lehrnbecher T, et al.
Utility of blood cultures in febrile neutropenia: time to positivity
and diagnostic yield. Pediatr Blood Cancer. 2021;68(7):e28834.
- Haeusler
GM, Thursky KA, Slavin MA, Lingaratnam S, Phillips RS, Babl FE, et al.
Procalcitonin and C-reactive protein in children with cancer and
febrile neutropenia: a systematic review. J Clin Oncol.
2021;39(28):3263-3274.
- Doerflinger
M, Forsyth C, McCarthy K, Roberts JA, Reuter S, Thompson A, et al.
Procalcitonin and IL-10 for early bloodstream infection prediction in
febrile neutropenia. Front Immunol. 2021;12:641879. https://doi.org/10.3389/fimmu.2021.641879 PMid:34093531 PMCid:PMC8173204
- Urbonas
V, Eidukaite A, Tamuliene I. The predictive value of IL-6 and IL-8 in
children with febrile neutropenia. J Pediatr Hematol Oncol.
2012;34(2):122-127. https://doi.org/10.1097/MPH.0b013e3182446a60 PMid:22367386
- Aquino
VM, Cost C, Gomez A, Sandler E, Buchanan GR, Schell MJ, et al. Cytokine
and monocyte chemoattractant protein-1 levels in febrile neutropenia. J
Pediatr Hematol Oncol. 2012;34(6):e241-e245. https://doi.org/10.1097/MPH.0b013e31824e498d PMid:22584776
- Zaoutis
TE, Olsen S, Coffin SE, Keren R, Prosser L, Shah SS, et al. Cytokine
variability in pediatric infections: implications for sepsis
biomarkers. Infect Control Hosp Epidemiol. 2020;41(6):701-708.
- Kitanovski
L, Jazbec J, Hojker S, Derganc M, Gvardijančič D, Rajic V, et al.
Neutrophil activation markers in pediatric febrile neutropenia. Support
Care Cancer. 2014;22(1):269-277. https://doi.org/10.1007/s00520-013-1978-1 PMid:24057110
- Sano
H, Kobayashi R, Suzuki D, Sano T, Yoshida M, Kobayashi K, et al.
Expression of immune activation markers in pediatric febrile
neutropenia. Support Care Cancer. 2022;30(3):2141-2149.
- Fisher
BT, Westling T, Shults J, Harrington WE, Hockenberry MJ, Zaoutis TE, et
al. Procalcitonin-guided antimicrobial stewardship in pediatric
oncology patients with febrile neutropenia. Pediatr Infect Dis J.
2020;39(10):e317-e323.
- Tissières P, Boulkedid R, Nadel S. Procalcitonin in pediatric intensive care decision-making. Ann Intensive Care. 2025;15:22. https://doi.org/10.1186/s13613-025-01470-y PMid:40268774 PMCid:PMC12018671
- Schulte
W, Bernhagen J, Bucala R. Cytokines in sepsis: mediators, biomarkers,
and therapeutic targets. Mediators Inflamm. 2013;2013:165974. https://doi.org/10.1155/2013/165974 PMid:23853427 PMCid:PMC3703895
- Zakaria
M, Salem S, Abdel Rahman M, Fathy M, Omar M, Hassan M, et al.
Diagnostic utility of CD64 as an early predictor of infection in
febrile neutropenia. Ital J Pediatr. 2025;51:12. https://doi.org/10.1186/s13052-025-01979-9 PMid:40369641 PMCid:PMC12079864
- de
Almeida Barbosa GG, Rocha AP, Alves JD, Silva MM, Souza CV, Nascimento
LP, et al. CD64 expression and blood cultures in febrile neutropenia.
Rev Bras Hematol Hemoter. 2015;37(6):387-393.
- Hodge
G, Osborn M, Hodge S, Holmes M, Reynolds PN, Bryant J, et al. Flow
cytometry markers of neutrophil activation in pediatric sepsis. Br J
Haematol. 2006;132(2):247-248. https://doi.org/10.1111/j.1365-2141.2005.05870.x PMid:16398661
- Haeusler
GM, Phillips R, Slavin MA, Lingaratnam S, Haeusler LM, Kotecha RS, et
al. Recalibration of clinical risk models in febrile neutropenia.
EClinicalMedicine. 2020;23:100394. https://doi.org/10.1016/j.eclinm.2020.100394 PMid:32637894 PMCid:PMC7329706
- Sandherr
M, Hentrich M, Burchardi D, Vehreschild JJ, Penack O, Buchheidt D, et
al. Diagnosis and therapy of febrile neutropenia in adult and pediatric
patients: 2025 AGIHO guideline update. Lancet Reg Health Eur.
2025;38:100123.
- Cerasi
S, Zanin A, D'Angelo P, Palumbo G, Riva C, Santoro C, et al. Presepsin
in pediatric febrile neutropenia: diagnostic accuracy and prognostic
value. Sci Rep. 2023;13:7078. https://doi.org/10.1038/s41598-023-33094-2 PMid:37081067 PMCid:PMC10119114
- Liang
J, Wang X, Liu Y, Sun Z, Zhou P, Li M, et al. Prognostic role of
presepsin in pediatric febrile neutropenia. Ann Med.
2025;57(1):134-142. https://doi.org/10.1080/07853890.2025.2561224 PMid:40961458 PMCid:PMC12444927
- Abdelaziz
TA, El Gendy Y, Hussein A, Khalaf M, Ibrahim S, Mahmoud A, et al. Serum
lactate as a prognostic marker in pediatric sepsis. BMC Pediatr.
2024;24:199. https://doi.org/10.1186/s12887-024-04809-9 PMid:38760748 PMCid:PMC11102193
- Le
DTK, Nguyen NT, Tran HT, Doan TT, Pham HT, Vu QN, et al.
Lactate/albumin ratio as an outcome predictor in pediatric sepsis.
Children (Basel). 2024;11(8):985.