We describe a rare case of community-acquired bloodstream infection caused by carbapenem-resistant Escherichia coli (CRECO), which led to an aplastic crisis.
A 75-year-old woman, weighing 40 kg and with no prior medical history, was admitted on September 10, 2025, after experiencing one week of fever, chills, rigors, nausea, and vomiting. On admission, her vital signs were: temperature 38.2 °C, pulse 59 beats/min, respiratory rate 20 breaths/min, blood pressure 100/55 mmHg, and SpO₂ 96%. Physical examination revealed petechiae, ecchymoses, pallor, and an anemic appearance. Laboratory tests showed severe pancytopenia: white blood cell count 0.25 × 10⁹/L, red blood cell count 2.5 × 10¹²/L, hemoglobin 76.0 g/L, platelet count 9.0 × 10⁹/L, absolute neutrophil count 0.02 × 10⁹/L, lymphocyte count 0.21 × 10⁹/L, and reticulocyte count 0.0028 × 10¹²/L. C-reactive protein was 276.4 mg/L, and procalcitonin was 0.754 ng/mL. Blood culture (anaerobic bottle) yielded Gram-negative bacilli. Empirical therapy with imipenem/cilastatin was started, along with intravenous fluconazole for antifungal prophylaxis, oral trimethoprim-sulfamethoxazole for Pneumocystis jirovecii prophylaxis, and oral acyclovir for herpes zoster prevention. Platelet transfusion was also administered. Bone marrow cytology showed markedly reduced cellularity with relative lymphocytosis. A bone marrow biopsy performed on September 11, 2025, revealed extensive fatty replacement, with hematopoietic tissue comprising about 20% and adipose tissue about 80% of the marrow. Both granulocytic and erythroid precursors were markedly decreased, consistent with very severe aplastic anemia. Further diagnostic evaluation was performed to investigate the cause of the severe pancytopenia and suspected aplastic crisis. In routine practice, the diagnostic workup of aplastic crisis includes viral studies such as parvovirus B19 PCR and serologies; however, these assays were not available at our institution. Other clinically relevant viral markers, including Epstein–Barr virus, cytomegalovirus, and hepatitis viruses, were assessed according to local laboratory capability and showed no evidence of acute infection. Bone marrow cytology and biopsy demonstrated markedly reduced cellularity, supporting the diagnosis of transient infection-related marrow suppression rather than primary aplastic anemia, particularly given the subsequent hematologic recovery following targeted antimicrobial therapy. Laboratory results are summarized in Table 1, and the temperature trend is shown in Figure 1.
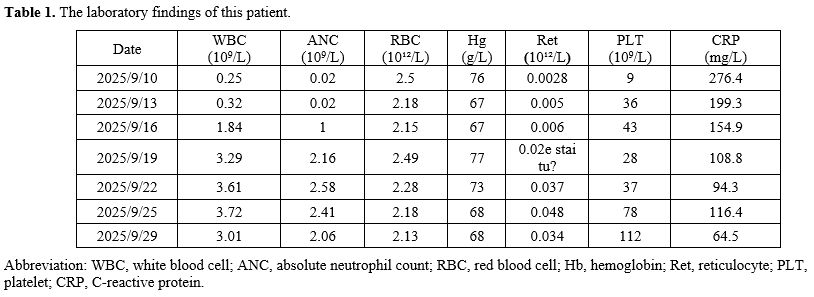 |
Table 1. The laboratory findings of this patient. |
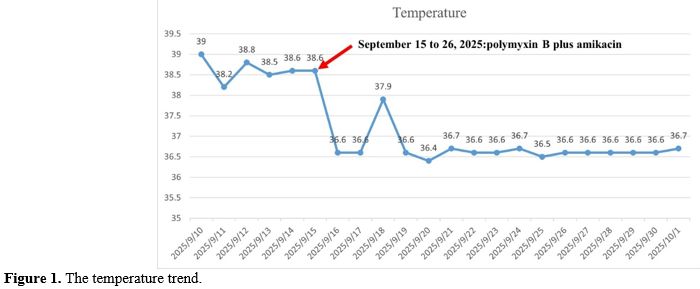 |
Figure 1. The temperature trend. |
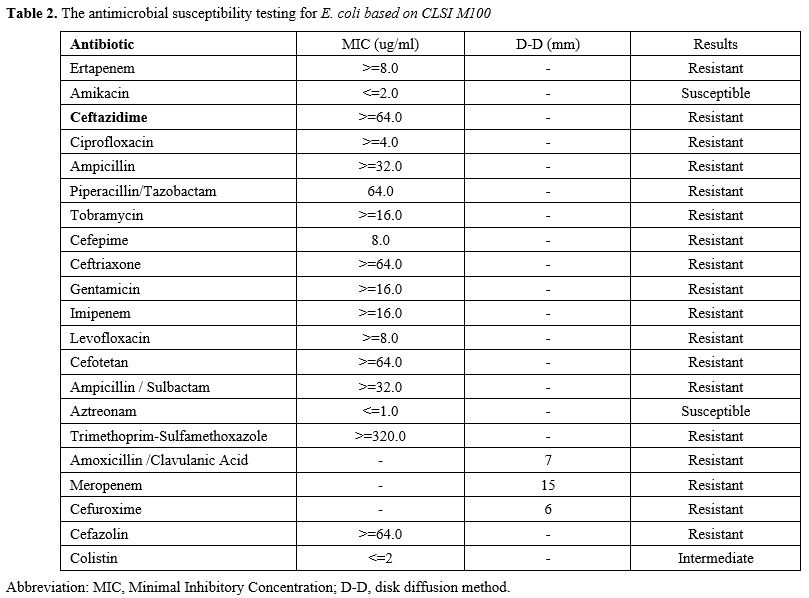
On September 13, 2025, antimicrobial susceptibility testing identified carbapenem-resistant E. coli producing NDM metallo-lactamase (Table 2). In accordance with treatment guidelines,[3] therapy was changed to polymyxin B plus amikacin. The patient’s temperature normalized, hematologic indices gradually improved, and follow-up bone marrow examination showed recovery of hematopoiesis. (Table 1, Figure 1)
Infections are defined as community-acquired when they are made within 48 hours of admission in the absence of healthcare-associated risk factors, such as prior hospitalization, surgery, dialysis, residence in a long-term care facility, or use of invasive devices within the previous 12 months. This patient met all criteria for community-acquired infection, as blood cultures obtained on the first hospital day confirmed the presence of carbapenem-resistant E. coli.
Epidemiologic data on community-acquired CRE bloodstream infections are limited because such cases are rare. Reported rates of community-associated or community-onset CRE infections range from 0.04% to 29.5%.[2] Potential sources include environmental reservoirs and animal-to-human transmission, which can lead to colonization. Previous studies suggest that community-onset CRE bacteremia may have a better prognosis than hospital-acquired infection.[5]
In this case, the infection also triggered an aplastic crisis. Transient bone-marrow suppression during severe bacterial sepsis is thought to result mainly from overwhelming systemic inflammation. High levels of cytokines such as TNF-α and IFN-γ can directly inhibit hematopoietic stem and progenitor cells, induce apoptosis, and impair erythroid and myeloid proliferation. In addition, bacterial components such as lipopolysaccharide can activate Toll-like receptor pathways within the marrow niche, further suppressing hematopoiesis.[6-9] These effects are functional and reversible, and marrow recovery usually occurs after infection control measures are implemented. Published reports of bacterial sepsis presenting as aplastic-appearing marrow are uncommon, and most involve hospital-acquired infections in critically ill or immunocompromised patients.[10,11] Unlike those previous cases, our patient developed profound but reversible marrow hypoplasia following community-acquired CRE bacteremia, highlighting that even community infections can cause transient bone-marrow failure when systemic inflammation is severe. This case reinforces the importance of early pathogen identification and targeted therapy to promptly reverse marrow suppression and prevent progression to irreversible marrow failure.
This case has two notable limitations. First, molecular genotyping of the resistance determinants was not performed, preventing detailed characterization of the genetic context of NDM production and limiting epidemiologic interpretation. Second, parvovirus B19 molecular testing and serology were not available at our institution, limiting our ability to exclude B19-associated aplastic crisis definitively. However, the negative results from other viral evaluations and the rapid recovery of hematopoiesis after targeted antibacterial therapy support the interpretation of transient sepsis-related marrow suppression.
The study protocol was approved by the Medical Ethics Committee of the First People’s Hospital of Zigong. The patient gave informed consent for the publication of this case report.
References
- WHO bacterial priority pathogens list, 2024
(2024-05-17): Bacterial pathogens of public health importance to guide
research, development and strategies to prevent and control
antimicrobial resistance. https://www.who.int/publications/i/item/9789240093461
- Kelly
AM, Mathema B, Larson EL. Carbapenem-resistant Enterobacteriaceae in
the community: a scoping review. Int J Antimicrob Agents. 2017
Aug;50(2):127-134. https://doi.org/10.1016/j.ijantimicag.2017.03.012 PMid:28647532 PMCid:PMC5726257
- Zeng,
M., et al., Guidelines for the diagnosis, treatment, prevention and
control of infections caused by carbapenem-resistant gram-negative
bacilli. Journal of microbiology, immunology, and infection = Wei mian
yu gan ran za zhi 2023, 56 (4), 653-671. https://doi.org/10.1016/j.jmii.2023.01.017 PMid:36868960
- Thaden,
J. T., et al., Rising rates of carbapenem-resistant enterobacteriaceae
in community hospitals: a mixed-methods review of epidemiology and
microbiology practices in a network of community hospitals in the
southeastern United States. Infection control and hospital epidemiology
2014, 35 (8), 978-83. https://doi.org/10.1086/677157 PMid:25026612 PMCid:PMC4217156
- Boutzoukas,
A. E., et al., Increased mortality in hospital- compared to
community-onset carbapenem-resistant enterobacterales infections. The
Journal of antimicrobial chemotherapy 2024, 79 (11), 2916-2922. https://doi.org/10.1093/jac/dkae306 PMid:39236214 PMCid:PMC11531819
- Selleri,
C., et al., Interferon-gamma and tumor necrosis factor-alpha suppress
both early and late stages of hematopoiesis and induce programmed cell
death. Journal of cellular physiology 1995, 165 (3), 538-46. https://doi.org/10.1002/jcp.1041650312 PMid:7593233
- Espinoza,
J. L., et al., Microbe-Induced Inflammatory Signals Triggering Acquired
Bone Marrow Failure Syndromes. Frontiers in immunology 2017, 8, 186. https://doi.org/10.3389/fimmu.2017.00186
- Nagaraju, N., et al., Bone Marrow Changes in Septic Shock: A Comprehensive Review. Cureus 2023, 15 (7), e42517. https://doi.org/10.7759/cureus.42517
- Davis,
F. M., et al., Sepsis Induces Prolonged Epigenetic Modifications in
Bone Marrow and Peripheral Macrophages Impairing Inflammation and Wound
Healing. Arteriosclerosis, thrombosis, and vascular biology 2019, 39
(11), 2353-2366. https://doi.org/10.1161/ATVBAHA.119.312754 PMid:31644352 PMCid:PMC6818743
- Wang,
H.-W., et al., Acquired Aplastic Anemia After Nosocomial Serratia
marcescens Bone Marrow Infection in an Elderly Patient. International
Journal of Gerontology 2011, 5 (4), 222-224. https://doi.org/10.1016/j.ijge.2011.12.002
- Rajendran,
A., et al., Severe systemic infection masking underlying childhood
leukemia. Indian J Hematol Blood Transfus 2013, 29 (3), 167-70. https://doi.org/10.1007/s12288-012-0166-7 PMid:24426366 PMCid:PMC3710556
- Giordano,
P., et al., An Epidemic of Parvovirus B19-Induced Aplastic Crises in
Pediatric Patients with Hereditary Spherocytosis Following the COVID-19
Pandemic: A Single-Center Retrospective Study. Children (Basel,
Switzerland) 2025, 12 (6). https://doi.org/10.3390/children12060772