Introduction
Chronic lymphocytic leukemia (CLL) is characterized by the presence of >5.000/mmc monoclonal B lymphocytes expressing CD5, CD23, CD19, CD20 and kappa or lambda immunoglobulin light chains in the peripheral blood, while small lymphocytic lymphoma (SLL) diagnosis is made if these B lymphocytes in peripheral blood are <5.000/mmc and confirmed by a lymph node biopsy.[1] Only patients who develop symptoms or signs of active disease need to start treatment with Bruton tyrosine kinase inhibitors (BTKis), the B-cell lymphoma 2 inhibitor venetoclax, or anti-CD20 monoclonal antibodies.[1] BTKis bind to a kinase involved in the signaling cascade of the B-cell receptor, reducing cell proliferation and survival. The main side effects of BTKis are cardiotoxicity, bleeding, myelosuppression, infections, diarrhoea, and fatigue.[2] The aim of this paper is to describe a case of macular edema in a patient treated with ibrutinib and to explore possible pathogenetic mechanisms linking BTKi therapy to this rare ocular complication.Clinical Presentation
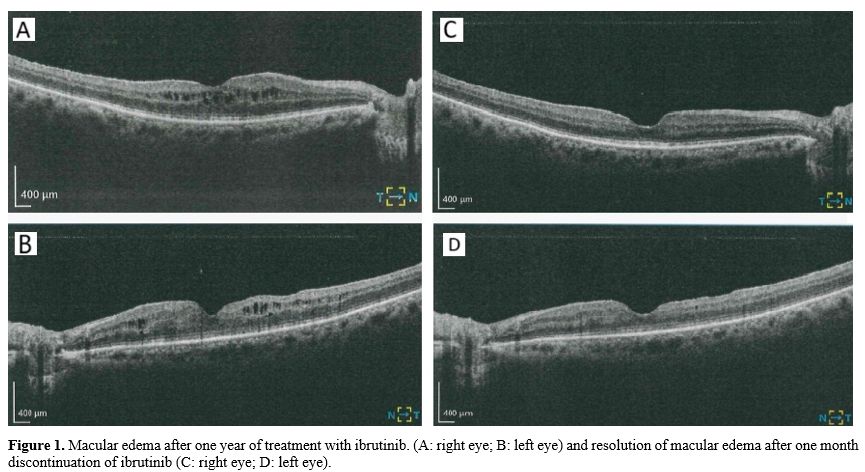
Discussion
Ophthalmic adverse events are very rare during treatment with ibrutinib. In a study of ibrutinib's safety profile, Allouchery et al. found that 0.64% of patients experienced serious ocular adverse events.[3] In contrast, the RESONATE study reported that 10% of patients taking ibrutinib experienced blurred vision.[4] Mirgh et al. described a case of a CLL patient who underwent third-line ibrutinib treatment and developed cystoid macular edema (CME). Despite topical and systemic anti-inflammatory drugs and a dose reduction of ibrutinib, the macular edema did not resolve until ibrutinib was discontinued.[5] On the other hand, Saenz-de-Viteri et al. reported a patient treated with ibrutinib for CLL who was diagnosed with CME by an OCT scan and improved with topical steroids without discontinuing the BTKi.[6] The peculiarity of our patient’s case is that he developed these side effects together with cardiac adverse events, while the other patients developed them as the only side effect. Furthermore, his symptoms resolved without any topical or systemic ophthalmologic therapy.We reviewed the off-target kinase-inhibition profile of ibrutinib to identify potential mechanisms underlying macular edema. Ibrutinib crosses the blood–brain barrier and can likely reach retinal tissues.[7] Lipsky et al. and Xiao et al.’s studies demonstrated ibrutinib off-target effects on other kinases, such as interleukin-2–inducible T-cell kinase (ITK), tyrosine-protein kinase (TEC), endothelial growth factor receptor (EGFR), FYN tyrosine kinase (FYN), mitogen-activated protein kinase 5 (MEK5), C-terminal c-Src kinase (CSK), and Receptor-interacting serine/threonine-protein kinase 3 (RIPK3).[8,9] They do not seem likely to cause or predispose macular edema, except for FYN tyrosine kinase.[10,11,12,13] Chavez-Solano et al. investigated the functional role of FYN tyrosine kinase in the postnatal neural retina, focusing on Müller cells, a specialized glial cell type that maintains retinal homeostasis and integrity through cytokine and chemokine secretion. They found that Fyn-deficient retinas exhibit distinct alterations, including zones devoid of cells in the inner and outer nuclear layers, increased immunoreactivity, reduced cell proliferation, and impaired focal adhesion formation.[11] This may be related to our research because inadequate retinal architecture and increased immunoreactivity, with higher levels of cytokines and chemokines, could lead to edema; however, this hypothesis needs to be tested in experimental studies.
Conclusions
We report a rare case of reversible macular edema associated with ibrutinib therapy in a patient with SLL. The temporal relationship between drug exposure and symptom resolution after discontinuation, without any ophthalmologic-specific therapy, strongly supports a causal link. Only a few reports have described BTKis as causing macular edema, and none have provided a pathogenic mechanism. Therefore, the pathogenesis of this side effect remains uncertain and may be linked to ibrutinib's off-target effects on other tyrosine kinases. The one we identify is Fyn tyrosine kinase, whose deficiency alters the retina’s structure and induces the production of cytokines. Surely, this hypothesis is speculative, and further reports and clinical experimental studies are required to confirm the correlation between ibrutinib and macular edema.Author contributions
C.M., A.N. and L.C. wrote the main manuscript text; C.M. e M.L. prepared figure 1; F.E., E.B., L.P., M.L., M.C. and M.P. made the principal corrections; A.V. and MI.DP. supervised and made the final corrections.Ethical Approval
Ethical approval was obtained from the Territorial Ethic Committee (R.S.3.25CS).Consent to publish
Informed consent was obtained from the patient at the patient's first hospital admission in accordance with the protocols approved by the Institutional Review Board and the ethical principles set forth by the Declaration of Helsinki.References
- Eichhorst B, Robak T,
Montserrat E, Ghia P, Niemann CU, Kater AP, Gregor M, Cymbalista F,
Buske C, Hillmen P, Hallek M, Mey U. Chronic lymphocytic leukaemia:
ESMO Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol;32(1):23-33. https://doi.org/10.1016/j.annonc.2020.09.019
- Byrd
JC, Furman RR, Coutre SE, Flinn IW, Burger JA, Blum KA, Grant B,
Sharman JP, Coleman M, Wierda WG, Jones JA, Zhao W, Heerema NA, Johnson
AJ, Sukbuntherng J, Chang BY, Clow F, Hedrick E, Buggy JJ, James DF,
O'Brien S. Targeting BTK with Ibrutinib in Relapsed Chronic Lymphocytic
Leukemia. New Engl J Med. 2013 Jul 4 ;369(1):32-42. https://doi.org/10.1056/NEJMoa1215637
PMid:23782158 PMCid:PMC3772525
- Allouchery
M, Tomowiak C, Lombard T, Pérault-Pochat MC, Salvo F. Safety Profile of
Ibrutinib: An Analysis of the WHO Pharmacovigilance Database. Front
Pharmacol. 2021 Oct 28 https://doi.org/10.3389/fphar.2021.769315
PMid:34776981 PMCid:PMC8580940
- Byrd
JC, Brown JR, O'Brien S, Barrientos JC, Kay NE, Reddy NM, Coutre S, Tam
CS, Mulligan SP, Jaeger U, Devereux S, Barr PM, Furman RR, Kipps TJ,
Cymbalista F, Pocock C, Thornton P, Caligaris-Cappio F, Robak T,
Delgado J, Schuster SJ, Montillo M, Schuh A, de Vos S, Gill D, Bloor A,
Dearden C, Moreno C, Jones JJ, Chu AD, Fardis M, McGreivy J, Clow F,
James DF, Hillmen P. Ibrutinib versus Ofatumumab in Previously Treated
Chronic Lymphoid Leukemia. New Engl J Med. 2014 Jul 17;371(3):213-23. https://doi.org/10.1056/NEJMoa1400376
PMid:24881631 PMCid:PMC4134521
- Mirgh
SP, Ahmed R, Agrawal N, Bothra S, Mohan B, Garg A, Thekkudan SF,
Khushoo V, Bhurani D. Knowing the Flip Side of the Coin: Ibrutinib
Associated Cystoid Macular Edema. Indian J Hematol Blood Transfus. 2019
Sep 13; 36(1):208-10. https://doi.org/10.1007/s12288-019-01181-y
PMid:32158110 PMCid:PMC7042422
- Saenz-de-Viteri
M, Cudrnak T. Bilateral cystoid macular edema in a patient with chronic
lymphocytic leukemia treated with ibrutinib. Leuk Amp Lymphoma. 2018
Sep 6; 60(3):842-4. https://doi.org/10.1080/10428194.2018.1508673
PMid:30188238
- Bernard
S, Goldwirt L, Amorim S, Brice P, Brière J, de Kerviler E, Mourah S,
Sauvageon H, Thieblemont C. Activity of ibrutinib in mantle cell
lymphoma patients with central nervous system relapse. Blood. 2015 Oct
1; 126(14):1695-8. https://doi.org/10.1182/blood-2015-05-647834
PMid:26239089 PMCid:PMC4591793
- Lipsky
A, Lamanna N. Managing toxicities of Bruton tyrosine kinase inhibitors.
Hematology. 2020 Dec 4 ;2020(1):336-45. https://doi.org/10.1182/hematology.2020000118
PMid:33275698 PMCid:PMC7727553
- Xiao
L, Salem JE, Clauss S, Hanley A, Bapat A, Hulsmans M, Iwamoto Y,
Wojtkiewicz G, Cetinbas M, Schloss MJ, Tedeschi J, Lebrun-Vignes B,
Lundby A, Sadreyev RI, Moslehi J, Nahrendorf M, Ellinor PT, Milan DJ.
Ibrutinib-Mediated Atrial Fibrillation Attributable to Inhibition of
C-Terminal Src Kinase. Circulation. 2020 Dec 22;142(25):2443-55. https://doi.org/10.1161/CIRCULATIONAHA.120.049210
PMid:33092403 PMCid:PMC9661397
- Fernandes
AF, Bian Q, Jiang JK, Thomas CJ, Taylor A, Pereira P, Shang F.
Proteasome Inactivation Promotes p38 Mitogen-activated Protein
Kinase-dependent Phosphatidylinositol 3-kinase Activation and Increases
Interleukin-8 Production in Retinal Pigment Epithelial Cells. Mol Biol
Cell. 2009 Aug 15; 20(16):3690-9. https://doi.org/10.1091/mbc.e08-10-1068
PMid:19570915 PMCid:PMC2777929
- Wu
Y, Feng B, Chen S, Zuo Y, Chakrabarti S. Glucose-induced endothelin-1
expression is regulated by ERK5 in the endothelial cells and retina of
diabetic ratsThis article is one of a selection of papers published in
the two-part special issue entitled 20 Years of Endothelin Research.
Can J Physiol Pharmacol. 2010 Jun;88(6):607-15. https://doi.org/10.1139/Y10-033
PMid:20628425
- Adasme
MF, Parisi D, Van Belle K, Salentin S, Haupt VJ, Jennings GS, Heinrich
JC, Herman J, Sprangers B, Louat T, Moreau Y, Schroeder M.
Structure-based drug repositioning explains ibrutinib as VEGFR2
inhibitor. PLOS ONE. 2020 May 27;15(5):e0233089. https://doi.org/10.1371/journal.pone.0233089
PMid:32459810 PMCid:PMC7252619
- Chavez-Solano
M, Ibarra-Sanchez A, Treviño M, Gonzalez-Espinosa C, Lamas M. Fyn
kinase genetic ablation causes structural abnormalities in mature
retina and defective Müller cell function. Mol Cell Neurosci. 2016 Apr;
72:91-100. https://doi.org/10.1016/j.mcn.2016.01.008
PMid:26808221