The pathobiological rationale for a dynamic, panel-based strategy in managing TTP and differentiating it from sepsis-related thrombotic microangiopathy or disseminated intravascular coagulation lies in the distinct mechanisms and clinical trajectories of these conditions. TTP is primarily characterized by a severe deficiency of ADAMTS13, an enzyme responsible for cleaving ultra-large von Willebrand factor (ULVWF) multimers, which, if left uncleaved, lead to microvascular thrombosis and the clinical manifestations of TTP, such as thrombocytopenia and microangiopathic hemolytic anemia.[11-13] This deficiency is often due to autoimmune inhibition, resulting in persistently low ADAMTS13 activity, which is a hallmark of TTP.[14,15] In contrast, sepsis-related thrombotic microangiopathy or disseminated intravascular coagulation involves transient suppression of ADAMTS13 activity, accompanied by surges in VWF and a consumption coagulopathy, alongside endothelial and inflammatory activation, marked by elevated levels of interleukin-6 (IL-6), heparin-binding protein (HBP), and serum amyloid A (SAA).[16,17] These inflammatory markers help differentiate the trajectory of sepsis-related thrombotic microangiopathy or disseminated intravascular coagulation from TTP.[18] Therefore, a serial measurement of ADAMTS13 activity, along with a thrombo-inflammatory panel, is crucial to capture the kinetics and phenotype of these conditions. This approach allows for the differentiation between the persistent enzymatic deficiency seen in TTP and the transient, inflammation-driven changes observed in sepsis-related thrombotic microangiopathy or disseminated intravascular coagulation, thereby guiding appropriate therapeutic interventions.[19,20]
Our primary objective was to determine whether dynamic ADAMTS-13 profiling, with stepwise addition of a coagulation consumption and a parsimonious inflammation index, improves iTTP risk stratification within the ISTH framework among Sepsis-3 ICU patients relative to a single baseline ADAMTS-13. Secondary objectives were to evaluate a pre treatment rule-out defined at 48 h using an RCV anchored ADAMTS-13 rise (≥35% or ≥10 % points) plus a clinically anchored low-consumption profile, and to quantify associations with ΔSOFA and 28-day mortality, while assessing robustness to heparin and lupus anticoagulant.
Methods
Study design. We conducted a prospective, single center diagnostic-accuracy study at The Fourth Affiliated Hospital of Soochow University (March 2023–February 2024) focused on Sepsis-3 patients with thrombocytopenia (platelets <100×109/L) and either schistocytes ≥1% or LDH >2× ULN, enrolled ≤24 h. All index tests were prespecified and processed blinded to final adjudication. The study addresses differential diagnosis and risk stratification in Sepsis-3 ICU patients with suspected thrombotic microangiopathy, not all comers with TMA. The protocol conformed to the Declaration of Helsinki and was approved by the ethics committee of the Fourth Affiliated Hospital of Soochow University. All participants provided written informed consent prior to any study procedures.Participants. Eligible patients met Sepsis-3 and had thrombocytopenia (platelets <100×109/L) plus either schistocytes ≥1% on an ICSH compliant smear or LDH >2× the upper limit of normal. Patients were excluded if therapeutic plasma exchange (PEx) occurred before baseline sampling, congenital TTP was known or strongly suspected, advanced hepatic failure precluded interpretation of factors, or pre analytical quality criteria were not met.
Index tests and sampling schedule. ADAMTS 13 activity (FRETS VWF73) and VWF:Ag were obtained at 0/24/48/72 h, together with a thrombo inflammatory panel (Prothrombin Time Test and INR [PT/INR], APTT, fibrinogen, thrombin time [TT], antithrombin III [AT III], D dimer, Fibrin degradation products [FDP], anti-factor Xa [anti Xa], and baseline lupus anticoagulant [LA], alongside CRP, PCT, IL 6, HBP, and SAA at all timepoints). Because standard care proceeded in parallel, we prospectively recorded PEx start times. For dynamic indices and the rule out, we defined an evaluable pre treatment window ending at the earliest of 48 h or the time of PEx initiation. Heparin intensity was continuously monitored by anti-Xa assay, and LA testing followed ISTH/ICSH procedures (screen–mix–confirm) with anticoagulant interference mitigation.
Laboratory methods and quality control. Citrate plasma was obtained via venipuncture or new arterial lines, maintained at 2–8 °C, double centrifuged, and assayed within manufacturer stability windows. Residual specimens were stored at −80 °C with ≤1 freeze–thaw cycle for batched cytokines. ADAMTS-13 and VWF assays were run with internal controls and external quality assurance. Inter-assay CVs were recorded across runs. Since unfractionated heparin prolongs TT/APTT, TT was interpreted only when anti-Xa <0.10 IU/mL; if anti-Xa ≥0.10 IU/mL, a reptilase time (or high concentration TT) was obtained to adjudicate fibrinogen function independent of heparin. LA testing used dRVVT and an LA-sensitive APTT in a screen–mix–confirm sequence with heparin neutralization and avoidance or validated removal of DOACs. Schistocytes were quantified per ICSH recommendations by expert morphologists.
Derived indices and operational definitions. We computed the VWF:ADAMTS-13 ratio, ISTH-DIC and SIC scores, and a Coagulation Consumption Index (CCI) (standardized composite of PT/INR, D-dimer/FDP, reverse signed fibrinogen, platelets, antithrombin III, factor V). The Dynamic ADAMTS-13 Index (DAI) combined the patient specific slope (from mixed-effects models) and log-AUC(0–72 h), estimated within the pre treatment window when applicable. The Inflammation Index (II) averaged z-scores of log(IL-6) and log(HBP).
The prespecified main rule out required a ≥ 15% relative rise in ADAMTS-13 by 48 h and a low-consumption profile defined by ISTH DIC < 5, fibrinogen ≥ 2.0 g/L, and antithrombin III ≥ 70%. Because a 15% change can approach analytical/biologic variation, we prespecified an RCV anchored sensitivity analysis requiring ≥ 35% relative rise or ≥ 10 absolute %-points by 48 h plus the same low consumption profile. The reference standard classified iTTP per ISTH definitions (severe ADAMTS-13 deficiency and inhibitor/anti-ADAMTS-13 when available, plus clinical response), acknowledging non independence with index tests.
Outcomes and diagnostic reference standard. The primary outcome was TTP discrimination across a stepwise model ladder: Model 1 (M1) used baseline ADAMTS-13; M2 substituted DAI; M3 added CCI; and M4 added II. The secondary diagnostic outcome was a pragmatic rule-out defined as ≥15% relative rise in ADAMTS-13 by 48 h, together with low CCI, chosen to maximize clinical feasibility; because 15% can fall within analytical/biologic variation, we prespecified a stricter sensitivity analysis requiring ≥35% relative rise or ≥10 absolute %-points by 48 h plus low CCI. Prognostic outcomes included 28-day all-cause mortality and ΔSOFA over 0–72 h. The reference standard for iTTP combined clinical adjudication with severe ADAMTS 13 deficiency (<10%) and, when available, inhibitor/anti ADAMTS-13 IgG and response to PEx. The non TTP group comprised sepsis associated TMA/DIC after exclusion of complement mediated TMA and STEC-HUS.
Statistical analysis. Group contrasts used Wilcoxon and χ²/Fisher tests with Hodges–Lehmann differences/odds ratios and FDR control. Repeated measures were modeled with mixed effects (group, time, interaction; random intercepts), with slopes/AUCs extracted within the pre treatment window when applicable. The model ladder (M1 baseline ADAMTS-13; M2 DAI; M3 DAI + CCI; M4 DAI + CCI + II) was deliberately parsimonious (≤ 3 predictors in M4). AUROC/AUPRC, Brier score, DeLong comparisons, calibration, and decision-curve analysis were reported. Internal validation used a 1,000-sample bootstrap optimism correction.
For the rule-out, performance in the intent-to-diagnose cohort is descriptive/exploratory. The pre-treatment estimand, restricted to patients untreated through 48 h (censoring at PEx start), is the target for decision making. Corresponding analyses are emphasized where feasible.
Results
Of 1,274 patients screened, 330 were enrolled within 24 h of ICU admission and completed the index-test schedule, 34 (10.3%) were adjudicated as iTTP, and 296 as non-TTP sepsis-associated TMA/DIC (Figure 1). Baseline profiles showed the anticipated divergence between iTTP and sepsis-associated coagulopathy (Table 1). iTTP patients were younger (mean difference −11 years, 95% CI −17 to −5, p=0.002, FDR q=0.006) and had more severe thrombocytopenia (18 vs 56×109/L, p<0.001, q<0.001), higher LDH (830 vs 610 U/L, p=0.002, q=0.006), and markedly lower ADAMTS-13 activity (7% vs 28%, p<0.001, q<0.001), resulting in a substantially higher VWF:ADAMTS-13 ratio (33 vs 12, p<0.001, q<0.001). Non-TTP patients exhibited the DIC phenotype with higher PT/INR and APTT, lower fibrinogen and AT-III, higher D-dimer, and higher ISTH-DIC and SIC scores (all p<0.001, q<0.001), consistent with consumption coagulopathy. Heparin exposure by anti-Xa categories was more frequent in non-TTP, while LA prevalence was identical (8.8%, p=0.999).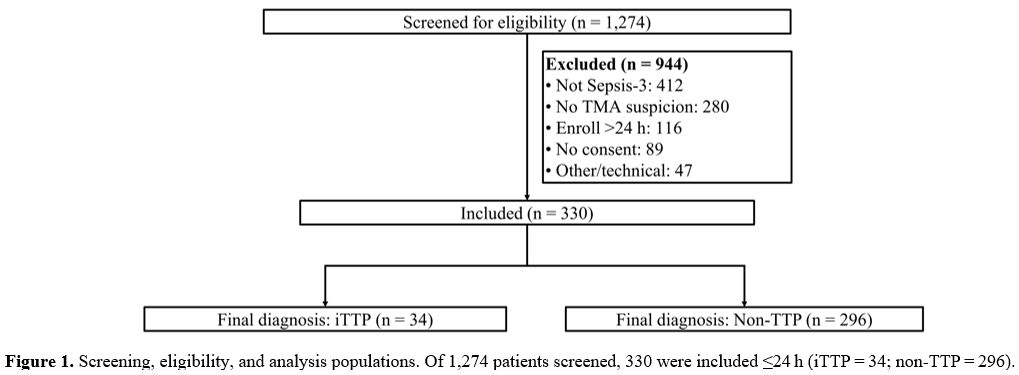 |
Figure 1. Screening,
eligibility, and analysis populations. Of 1,274 patients screened, 330
were included ≤24 h (iTTP = 34; non‑TTP = 296). |
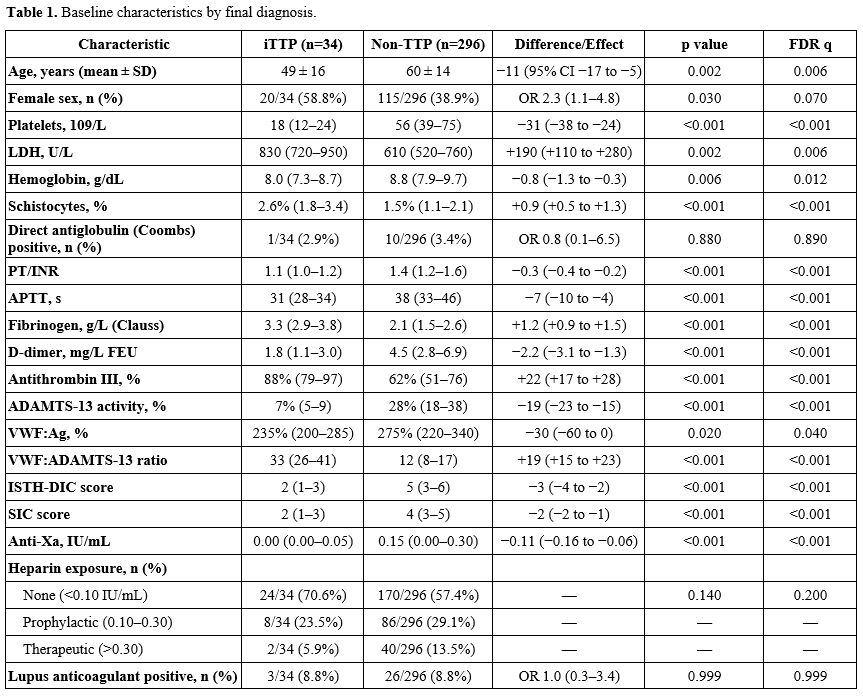 |
Table 1. Baseline characteristics by final diagnosis. |
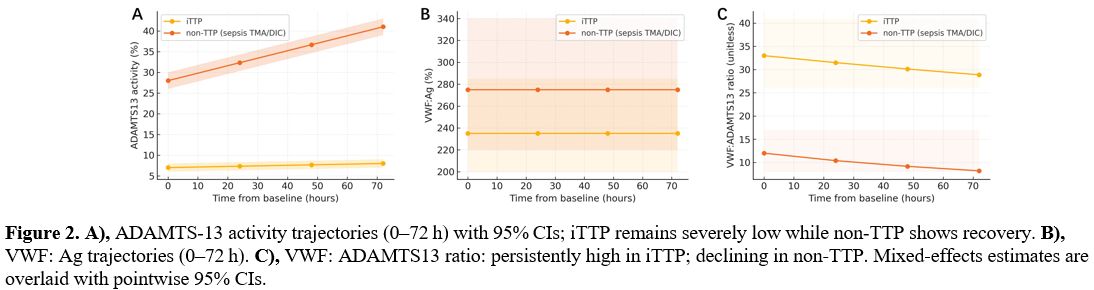
Longitudinal modeling demonstrated distinct kinetics by diagnosis (Figure 2). ADAMTS-13 activity in iTTP remained severely depressed with minimal change from baseline to 72 h (mean 7% [95% CI 6–8] to 8% [7–9]), whereas non TTP recovered from 28% (26–30) to 41% (39–43), yielding a significant group×time interaction (p<0.001). VWF:Ag was high in both groups but modestly higher in non-TTP (group effect p=0.014), and the VWF:ADAMTS-13 ratio declined over time in non-TTP while remaining markedly elevated in iTTP (group×time p<0.001) (Figure 2).
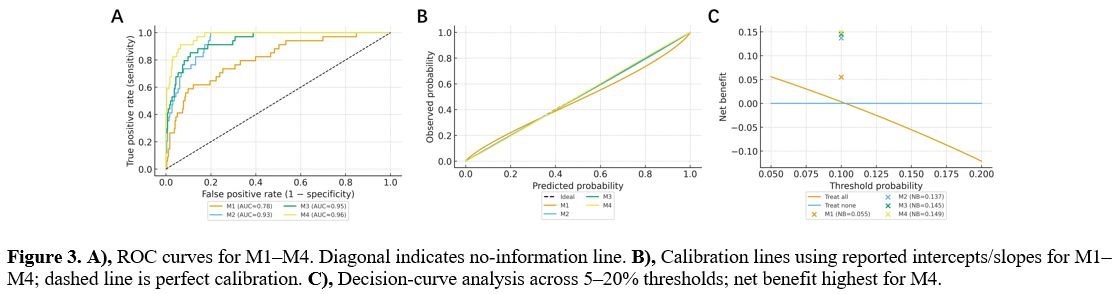
Within Sepsis-3 ICU patients with thrombocytopenia and microangiopathic hemolysis features, discrimination improved stepwise from M1 (baseline ADAMTS-13, AUROC 0.78 [0.70–0.86]) to M2 (DAI, 0.93 [0.88–0.97], Δ=0.15 vs M1, p<0.001), with smaller incremental gains for M3 (0.95 [0.92–0.98], Δ=0.02, p=0.021) and M4 (0.96 [0.93–0.99], Δ=0.01, p=0.048), and acceptable calibration and decision-curve net benefit (Table 2, Figure 3). As DAI includes dynamic information that can be influenced by post-baseline care, we estimated slopes/AUCs within the pre-treatment window where data permitted. Where not, dynamic effects are interpreted as monitoring/prognostic signals rather than pre-treatment diagnostic information. Internal validity was assessed with bootstrap optimism correction (1,000 resamples).
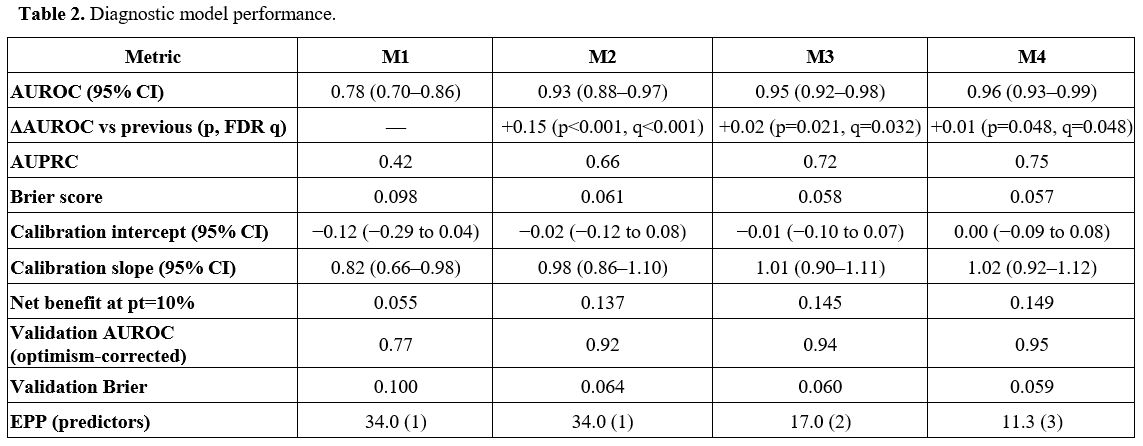 |
Table 2. Diagnostic model performance. |
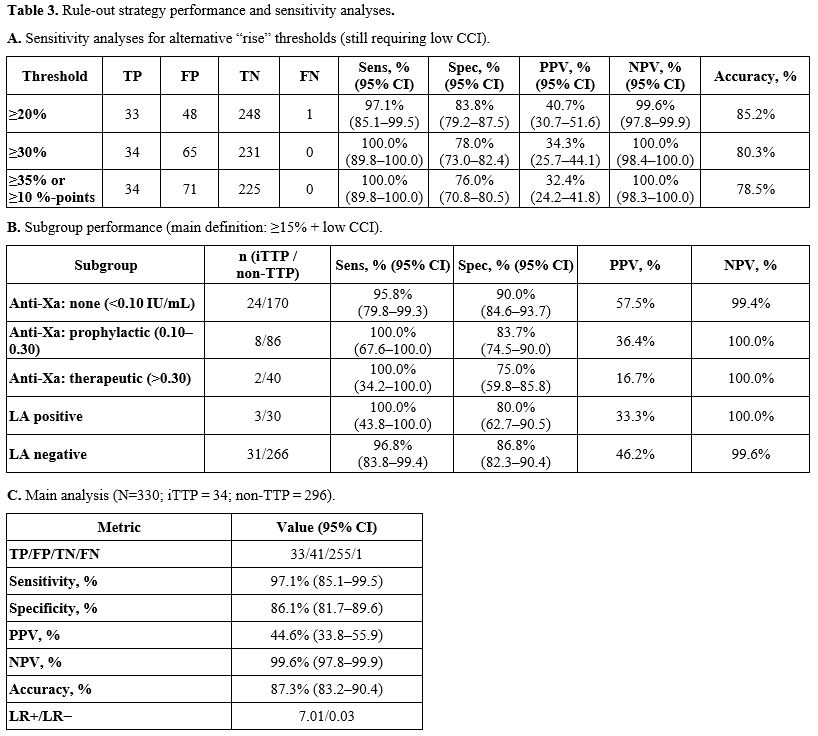
Using the prespecified main rule-out (≥ 15% ADAMTS-13 rise by 48 h plus low CCI), the intent-to-diagnose analysis yielded 97.1% sensitivity, 86.1% specificity, and 99.6% NPV (Table 3). In a prespecified RCV-anchored sensitivity analysis (≥ 35% relative rise or ≥ 10-point rise by 48 h plus low consumption), sensitivity and NPV were 100.0% with 76.0% specificity (Table 3). Because some high-probability iTTP patients initiated PEx before 48 h, these care-embedded estimates cannot be interpreted as pre-treatment diagnostic performance. Pre-treatment analyses are prioritized for decision-making and are presented where data permit. Subgroup patterns were consistent across anti-Xa strata and lupus anticoagulant status (Table 3).
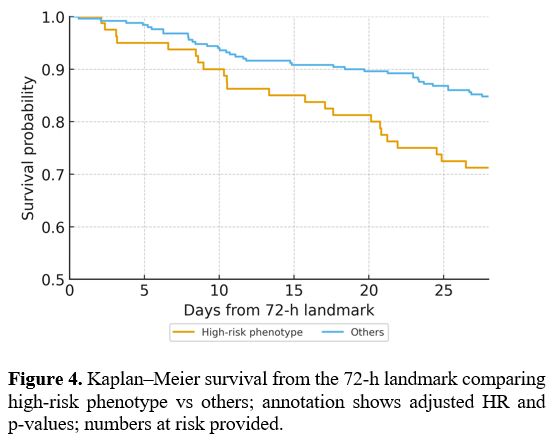
At the 72-h landmark, the high risk phenotype, defined a priori as ADAMTS 13 <10% at 72 h together with IL-6 and/or HBP at 72 h ≥ the 75th percentile of the cohort at 72 h, was associated with worse 28 day survival than all others (29.2% vs 15.2% mortality, log-rank p=0.006) (Figure 4). In Cox models adjusted for age, sex, and baseline SOFA, the high risk phenotype conferred an HR of 2.6 (95% CI 1.3–5.2, p=0.007), proportional hazards assumptions were not violated (global Schoenfeld p=0.22), and mixed-effects models showed greater SOFA worsening from 0–72 h in the high-risk group (p=0.002).
 |
|
Discussion
In this Sepsis-3 ICU cohort with TMA features, moving from a single baseline ADAMTS-13 measure to dynamic profiling produced a substantial gain in iTTP discrimination, with incremental improvements after adding a parsimonious coagulation index and a two-analyte inflammation index, while maintaining calibration and net benefit. The pragmatic rule-out achieved 97.1% sensitivity and 99.6% NPV in the care-embedded analysis, and an RCV-anchored threshold (≥ 35% or ≥ 10%, points) preserved 100% sensitivity/NPV at the expected cost to specificity, supporting a safety first option when the consequences of missed iTTP are high. A 72-h high-risk phenotype was associated with higher 28-day mortality and greater SOFA worsening, underscoring the biological plausibility that early enzymatic and endothelial-inflammatory signals capture clinically meaningful risk.In our ICU cohort, iTTP showed the expected pathognomonic pattern, severe and persistent ADAMTS-13 deficiency with minimal 72-h recovery, whereas sepsis associated thrombotic microangiopathy or disseminated intravascular coagulation exhibited partial suppression with progressive ADAMTS-13 recovery alongside a sustained VWF surge, yielding a markedly higher and more persistent VWF:ADAMTS-13 ratio in iTTP. These trajectories align with guideline definitions that anchor iTTP to ADAMTS-13 activity <10% and inhibitory autoantibodies, whereas sepsis is characterized by endothelial activation and consumptive coagulopathy rather than isolated ADAMTS 13 loss.[21,22] Experimental and clinical studies in sepsis further show that VWF release and relative ADAMTS-13 deficiency track organ dysfunction and mortality, supporting the biologic coherence of our dynamic-index approach and the prognostic salience of the ratio.[23-25] The inflammatory context, IL-6, HBP, and SAA explain why our high risk phenotype was enriched for death and worsening SOFA.[26-28] Finally, our smear threshold aligns with ICSH recommendations across TMAs and supports the initial suspicion of microangiopathy.[29]
The stepwise model gains we observed, moving from baseline ADAMTS-13 (M1) to dynamic ADAMTS-13 (M2), then adding a Coagulation Consumption Index (M3) and a parsimonious Inflammation Index (M4), support embedding serial ADAMTS 13 plus a focused panel early in ICU workflows to sharpen iTTP discrimination and prevent overuse of plasma exchange. Importantly, guideline-concordant care requires not delaying urgent iTTP therapy in high-probability presentations. Dynamic testing primarily refines decisions in sepsis-dominant, low-to-intermediate probability cases.[21,22] Our pragmatic rule out delivered 97.1% sensitivity and 99.6% NPV with robustness across anti-Xa strata and LA status, complementing Sepsis-3 definitions and ISTH DIC/SIC frameworks for coagulopathy phenotyping.[30-32] Implementation should include systematic anti-Xa monitoring to quantify heparin exposure, interpret thrombin time only when anti-Xa is low or confirm with reptilase time, and follow ISTH SSC procedures for LA testing.[33-35] When ADAMTS-13 results are pending, PLASMIC remains useful for pre-test triage but should be contextualized against dynamic enzyme data and the focused panel.[36,37] Serial ADAMTS-13 with a focused coagulation/inflammation panel can be embedded early in ICU workflows to sharpen iTTP discrimination and potentially reduce unnecessary plasma exchange, while flagging a high risk phenotype for intensified care. Broad deployment of a serial ADAMTS-13 strategy depends first on establishing an institutional pathway for rapid ADAMTS-13 activity reporting for Sepsis-3 ICU patients with TMA features. Where this prerequisite is not yet met, the approach we tested can be implemented in stages. 1) Obtain ADAMTS-13 at presentation (0 h) and repeat at 48 h, and apply the prespecified rule out using routine coagulation markers (ISTH DIC <5, fibrinogen ≥2.0 g/L, AT III ≥70%) together with a ≥15% rise in ADAMTS-13 (or the prespecified RCV anchored rise as a safety first option). This tier intentionally avoids complex indices and is designed to be feasible for early adoption. 2) Where serial testing is established, incorporate dynamic ADAMTS-13 information and coagulation consumption signals to refine classification in sepsis-dominant, low-to-intermediate probability presentations, while recognizing that high-probability iTTP remains a “treat-now” diagnosis. 3) Add a parsimonious inflammation signal to support risk phenotyping and prognosis. In settings with informatics support, trajectories/indices can be automatically computed within the laboratory information system or EHR, minimizing additional clinician burden.
Limitations of this study include the single-center setting and a modest iTTP case count, which limited power to detect small incremental gains; potential assay and turnaround constraints that may affect workflow generalizability; and residual confounding despite anti-Xa/LA adjustments. In centers without same-day ADAMTS-13 availability, early decisions will still rely on clinical probability and empiric therapy. Therefore, implementation studies focusing on time to result and laboratory workflows are a prerequisite to generalizability. Although we used a 72-h landmark to avoid immortal-time bias in prognosis analyses, this design can exclude very early deaths and will require confirmation in broader populations. Additionally, a ≥15% rise in ADAMTS-13 may reflect analytical/biologic variation, which we addressed by stricter sensitivity thresholds, but that should be externally validated. Future work should prioritize multicenter external validation with assay specific recalibration where needed, build real-time/near-patient assay pathways and decision-support to operationalize serial ADAMTS-13 plus a focused panel, and conduct health-economic studies quantifying plasma-exchange averted, time-to-treatment, and safety.
Taken together, these findings support embedding dynamic ADAMTS-13, along with a focused coagulation/inflammation panel, into early ICU workflows to enhance diagnosis and risk stratification across sepsis-associated TMAs, enable a pragmatic rule-out pathway that may reduce unnecessary plasma exchange, and inform targeted escalation for high-risk patients, pending multicenter implementation and economic evaluation.
Ethics approval and consent to participate
The protocol conformed to the Declaration of Helsinki and was approved by the ethics committee of the Fourth Affiliated Hospital of Soochow University. All participants provided written informed consent prior to any study procedures.Data availability statement
Data sets generated during the current study are available from the corresponding author on reasonable request.Author Contribution Statement
The authors confirm contribution to the paper as follows: study conception and design: B.Y.; data collection: Y.L., F.L., B.J., W.S., H.S., Y.X.; analysis and interpretation of results: Y.L., F.L., B.J., W.S., H.S., Y.X.; draft manuscript preparation: Y.L., F.L., B.J., W.S., H.S., Y.X., B.Y. All authors reviewed the results and approved the final version of the manuscript.References
- Joly, B. S., Coppo, P., & Veyradier, A. (2017). Thrombotic thrombocytopenic purpura. Blood,129(21), 2836-2846. https://doi.org/10.1182/blood-2016-10-709857 PMid:28416507
- Kremer
Hovinga, J. A., Coppo, P., Lämmle, B., Moake, J. L., Miyata, T., &
Vanhoorelbeke, K. (2017). Thrombotic thrombocytopenic purpura. Nature
Reviews Disease Primers, 3, 17020. https://doi.org/10.1038/nrdp.2017.20 PMid:28382967
- Cohen, C. T. (2018). Background and Presentation of Thrombotic Thrombocytopenic Purpura (pp. 153-169). Springer, Cham. https://doi.org/10.1007/978-3-319-73269-5_8
- Gogia,
P., Gbujie, E., Benge, E., & Bhasin, S. (2020). Thrombotic
Thrombocytopenic Purpura: Revisiting a Miss and an Inevitable
Consequence. Cureus, 12(7). https://doi.org/10.7759/cureus.9283 PMid:32699732 PMCid:PMC7372183
- Bentley, M. J. (2015). Thrombotic Microangiopathies and Their Distinction from TTP (pp. 129-139). Springer, Cham. https://doi.org/10.1007/978-3-319-08717-7_7
- Coppo,
P., Schwarzinger, M., Buffet, M., Wynckel, A., Clabault, K., Presne,
C., Poullin, P., Malot, S., Vanhille, P., Azoulay, E., Galicier, L.,
Lemiale, V., Mira, J. P., Ridel, C., Rondeau, E., Pourrat, J., Girault,
S., Bordessoule, D., Saheb, S., Ramakers, M., French Reference Center
for Thrombotic Microangiopathies (2010). Predictive features of severe
acquired ADAMTS13 deficiency in idiopathic thrombotic
microangiopathies: the French TMA reference center experience. PloS
One, 5(4), e10208. https://doi.org/10.1371/journal.pone.0010208 PMid:20436664 PMCid:PMC2859048
- Mariotte,
E., Zafrani, L., Fadlallah, J., Galicier, L., Ghrenassia, E., Kerhuel,
L., Calvet, L., De Jong, A., Lemiale, V., Valade, S., Joly, B. S.,
Stepanian, A., Azoulay, E., Azoulay, E., Darmon, M., & Darmon, M.
(2021). Performance of Diagnostic Scores in Thrombotic Microangiopathy
Patients in the Intensive Care Unit: A Monocentric Study. Thrombosis
and Haemostasis, 121(11), 1427-1434. https://doi.org/10.1055/a-1378-3804 PMid:33512705
- Kubo,
M., & Matsumoto, M. (2022). Diagnosis of thrombotic
thrombocytopenic purpura (TTP): Current status and challenges. Journal
of the Japanese Society on Thrombosis and Hemostasis, 33(4), 399-407. https://doi.org/10.2491/jjsth.33.399
- Scully,
M. (2017). Thrombocytopenia in hospitalized patients: approach to the
patient with thrombotic microangiopathy. Hematology, 2017(1), 651-659. https://doi.org/10.1182/asheducation-2017.1.651 PMid:29222317 PMCid:PMC6142615
- Scully
M. (2017). Thrombocytopenia in hospitalized patients: approach to the
patient with thrombotic microangiopathy. Hematology. American Society
of Hematology. Education Program, 2017(1), 651-659. https://doi.org/10.1182/asheducation-2017.1.651 PMid:29222317 PMCid:PMC6142615
- Kappers-Klunne,
M. C., van Asten, J. G., & Van Vliet, H. H. D. M. (2009). ADAMTS-13
and Von Willebrand factor in relation to platelet response during
plasma exchange in thrombotic thrombocytopenic purpura: a clue for
disease mechanism? Annals of Hematology, 88(10), 1025-1028. https://doi.org/10.1007/s00277-009-0709-7 PMid:19205653 PMCid:PMC2728906
- Sartain, S. E. (2018). Pathophysiology of Thrombotic Thrombocytopenic Purpura (pp. 171-188). Springer, Cham. https://doi.org/10.1007/978-3-319-73269-5_9
- Smith,
L. (2020). Pathophysiology of thrombotic thrombocytopenia purpura.
Clinical Laboratory Science: Journal of the American Society for
Clinical Laboratory Science, 33(3), 55-59. https://doi.org/10.29074/ascls.2020002261
- Ukleba,
K., & Gvetadze, L. (2019). Thrombotic thrombocytopenic purpura
(TTP) and modern approach to its investigation and treatment. Science
Review, (3), 12-13. https://doi.org/10.31435/rsglobal_sr/31032019/6381
- Papakonstantinou,
A., Kalmoukos, P., Mpalaska, A., Koravou, E. E., & Gavriilaki, E.
(2024). ADAMTS13 in the New Era of TTP. International Journal of
Molecular Sciences, 25(15), 8137. https://doi.org/10.3390/ijms25158137 PMid:39125707 PMCid:PMC11312255
- Kremer
Hovinga, J. A., Coppo, P., Lämmle, B., Moake, J. L., Miyata, T., &
Vanhoorelbeke, K. (2017). Thrombotic thrombocytopenic purpura. Nature
Reviews Disease Primers, 3, 17020. https://doi.org/10.1038/nrdp.2017.20 PMid:28382967
- Lancellotti,
S., Sacco, M., Tardugno, M., Ferretti, A., & De Cristofaro, R.
(2023). Immune and Hereditary Thrombotic Thrombocytopenic Purpura: Can
ADAMTS13 Deficiency Alone Explain the Different Clinical Phenotypes?
Stomatology, 12(9), 3111. https://doi.org/10.3390/jcm12093111 PMid:37176552 PMCid:PMC10179526
- Tsai, H.-M. (2010). Pathophysiology of thrombotic thrombocytopenic purpura. International Journal of Hematology, 91(1), 1-19. https://doi.org/10.1007/s12185-009-0476-1 PMid:20058209 PMCid:PMC3159000
- Kato,
S., & Fujimura, Y. (2015). Thrombotic Thrombocytopenic Purpura
--Pathophysiology and Assays of ADAMTS13 Activity. The Japanese Journal
of Clinical Pathology, 63(10), 1228-1236.
- Valsecchi,
C., Mirabet, M., Mancini, I., Biganzoli, M., Schiavone, L., Faraudo,
S., Mane-Padros, D., Giles, D., Serra-Domenech, J., Blanch, S.,
Trisolini, S. M., Facchini, L., Rinaldi, E., Peyvandi, F., &
Peyvandi, F. (2019). Evaluation of a New, Rapid, Fully Automated Assay
for the Measurement of ADAMTS13 Activity. Thrombosis and Haemostasis,
119(11), 1767-1772. https://doi.org/10.1055/s-0039-1696718 PMid:31587247
- Zheng,
X. L., Vesely, S. K., Cataland, S. R., Coppo, P., Geldziler, B., Iorio,
A., Matsumoto, M., Mustafa, R. A., Pai, M., Rock, G., Russell, L.,
Tarawneh, R., Valdes, J., & Peyvandi, F. (2020). ISTH guidelines
for the diagnosis of thrombotic thrombocytopenic purpura. Journal of
Thrombosis and Haemostasis: JTH,18(10), 2486-2495. https://doi.org/10.1111/jth.15006 PMid:32914582 PMCid:PMC8146131
- Scully,
M., Rayment, R., Clark, A., Westwood, J. P., Cranfield, T., Gooding,
R., Bagot, C. N., Taylor, A., Sankar, V., Gale, D., Dutt, T., McIntyre,
J., Lester, W., & BSH Committee (2023). A British Society for
Haematology Guideline: Diagnosis and management of thrombotic
thrombocytopenic purpura and thrombotic microangiopathies. British
Journal of Haematology, 203(4), 546-563. https://doi.org/10.1111/bjh.19026 PMid:37586700 PMCid:PMC12209848
- Peetermans,
M., Meyers, S., Liesenborghs, L., Vanhoorelbeke, K., De Meyer, S. F.,
Vandenbriele, C., Lox, M., Hoylaerts, M. F., Martinod, K., Jacquemin,
M., Vanassche, T., & Verhamme, P. (2020). Von Willebrand factor and
ADAMTS13 impact on the outcome of Staphylococcus aureus sepsis. Journal
of Thrombosis and Haemostasis : JTH,18(3), 722-731. https://doi.org/10.1111/jth.14686 PMid:31758651
- Schwameis,
M., Schörgenhofer, C., Assinger, A., Steiner, M. M., & Jilma, B.
(2015). VWF excess and ADAMTS13 deficiency: a unifying pathomechanism
linking inflammation to thrombosis in DIC, malaria, and TTP. Thrombosis
and Haemostasis,113(4), 708-718. https://doi.org/10.1160/TH14-09-0731 PMid:25503977 PMCid:PMC4745134
- Bockmeyer,
C. L., Claus, R. A., Budde, U., Kentouche, K., Schneppenheim, R.,
Lösche, W., Reinhart, K., & Brunkhorst, F. M. (2008).
Inflammation-associated ADAMTS13 deficiency promotes formation of
ultra-large von Willebrand factor. Haematologica,93(1), 137-140. https://doi.org/10.3324/haematol.11677 PMid:18166799
- Taha,
A. M., Najah, Q., Omar, M. M., Abouelmagd, K., Ali, M., Hasan, M. T.,
Allam, S. A., Hamam, Y. A., Arian, R., & Abd-ElGawad, M. (2024).
Diagnostic and prognostic value of heparin-binding protein in sepsis: A
systematic review and meta-analysis. Medicine,103(25), e38525. https://doi.org/10.1097/MD.0000000000038525 PMid:38905400 PMCid:PMC11191987
- Bentzer,
P., Fisher, J., Kong, H. J., Mörgelin, M., Boyd, J. H., Walley, K. R.,
Russell, J. A., & Linder, A. (2016). Heparin-binding protein is
important for vascular leak in sepsis. Intensive Care Medicine
Experimental,4(1), 33. https://doi.org/10.1186/s40635-016-0104-3 PMid:27704481 PMCid:PMC5050173
- McMullan,
R. R., McAuley, D. F., O'Kane, C. M., & Silversides, J. A. (2024).
Vascular leak in sepsis: physiological basis and potential therapeutic
advances. Critical Care (London, England),28(1), 97. https://doi.org/10.1186/s13054-024-04875-6 PMid:38521954 PMCid:PMC10961003
- Zini,
G., d'Onofrio, G., Erber, W. N., Lee, S. H., Nagai, Y., Basak, G. W.,
Lesesve, J. F., & International Council for Standardization in
Hematology (ICSH) (2021). 2021 update of the 2012 ICSH Recommendations
for identification, diagnostic value, and quantitation of schistocytes:
Impact and revisions. International Journal of Laboratory
Hematology,43(6), 1264-1271. https://doi.org/10.1111/ijlh.13682 PMid:34431220
- Singer,
M., Deutschman, C. S., Seymour, C. W., Shankar-Hari, M., Annane, D.,
Bauer, M., Bellomo, R., Bernard, G. R., Chiche, J. D., Coopersmith, C.
M., Hotchkiss, R. S., Levy, M. M., Marshall, J. C., Martin, G. S.,
Opal, S. M., Rubenfeld, G. D., van der Poll, T., Vincent, J. L., &
Angus, D. C. (2016). The Third International Consensus Definitions for
Sepsis and Septic Shock (Sepsis-3). JAMA,315(8), 801-810. https://doi.org/10.1001/jama.2016.0287 PMid:26903338 PMCid:PMC4968574
- Iba,
T., Levy, J. H., Maier, C. L., Helms, J., Umemura, Y., Moore, H.,
Othman, M., Thachil, J., Connors, J. M., Levi, M., & Scarlatescu,
E. (2025). Updated definition and scoring of disseminated intravascular
coagulation in 2025: communication from the ISTH SSC Subcommittee on
Disseminated Intravascular Coagulation. Journal of Thrombosis and Haemostasis: JTH,23(7), 2356-2362. https://doi.org/10.1016/j.jtha.2025.03.038 PMid:40216223
- Iba,
T., Nisio, M. D., Levy, J. H., Kitamura, N., & Thachil, J. (2017).
New criteria for sepsis-induced coagulopathy (SIC) following the
revised sepsis definition: a retrospective analysis of a nationwide
survey. BMJ Open,7(9), e017046. https://doi.org/10.1136/bmjopen-2017-017046 PMid:28963294 PMCid:PMC5623518
- Devreese,
K. M. J., de Groot, P. G., de Laat, B., Erkan, D., Favaloro, E. J.,
Mackie, I., Martinuzzo, M., Ortel, T. L., Pengo, V., Rand, J. H.,
Tripodi, A., Wahl, D., & Cohen, H. (2020). Guidance from the
Scientific and Standardization Committee for lupus
anticoagulant/antiphospholipid antibodies of the International Society
on Thrombosis and Haemostasis: Update of the guidelines for lupus
anticoagulant detection and interpretation. Journal of Thrombosis and
Haemostasis: JTH,18(11), 2828-2839. https://doi.org/10.1111/jth.15047 PMid:33462974
- Tripodi,
A., Cohen, H., & Devreese, K. M. J. (2020). Lupus anticoagulant
detection in anticoagulated patients. Guidance from the Scientific and
Standardization Committee for lupus anticoagulant/antiphospholipid
antibodies of the International Society on Thrombosis and
Haemostasis.Journal of Thrombosis and Haemostasis: JTH,18(7),
1569-1575. https://doi.org/10.1111/jth.14846 PMid:32619349
- ARUP Consult. (n.d.). Prolonged clotting time evaluation. ARUP Consult. Retrieved November 1, 2025, from https://arupconsult.com/content/prolonged-clotting-time-evaluation
- Bendapudi,
P. K., Hurwitz, S., Fry, A., Marques, M. B., Waldo, S. W., Li, A., Sun,
L., Upadhyay, V., Hamdan, A., Brunner, A. M., Gansner, J. M.,
Viswanathan, S., Kaufman, R. M., Uhl, L., Stowell, C. P., Dzik, W. H.,
& Makar, R. S. (2017). Derivation and external validation of the
PLASMIC score for rapid assessment of adults with thrombotic
microangiopathies: a cohort study. The Lancet. Haematology,4(4),
e157-e164. https://doi.org/10.1016/S2352-3026(17)30026-1 PMid:28259520
- Li,
A., Khalighi, P. R., Wu, Q., & Garcia, D. A. (2018). External
validation of the PLASMIC score: a clinical prediction tool for
thrombotic thrombocytopenic purpura diagnosis and treatment. Journal of
Thrombosis and Haemostasis: JTH,16(1), 164-169. https://doi.org/10.1111/jth.13882 PMid:29064619 PMCid:PMC5760324