The pathogenesis of SICM remains incompletely understood. Existing evidence suggests that inflammatory mediator release, oxidative stress, mitochondrial dysfunction, and metabolic disorders are central mechanisms.[5,6] During the progression of sepsis, dysregulated immune responses trigger the massive release of inflammatory mediators (e.g., TNF-α and IL-6), directly damaging cardiomyocytes and impairing contractile function.[7] Oxidative stress not only exacerbates inflammation but also disrupts mitochondrial energy metabolism, leading to impaired myocardial contraction and relaxation.[8] In addition, hemodynamic disturbances and inadequate microcirculatory perfusion may induce myocardial ischemia and further aggravate cardiac dysfunction.[9] Therefore, SICM is closely linked to disruption of the inflammatory microenvironment, and inflammatory markers may help reflect inflammatory severity and the risk of myocardial injury.[10]
In clinical research, hs-cTnT is a commonly used marker of myocardial injury and has been applied in the prediction of several infection-related cardiac conditions.[11-14] At the same time, peripheral blood inflammatory indices (PBIIs) derived from routine blood parameters have received increasing attention in recent years.[15,16] These indices are simple, low-cost, and widely available in routine practice. Commonly used indicators include the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), systemic immune-inflammation index (SII), systemic inflammation response index (SIRI), and pan-immune-inflammation value (PIV). Existing studies suggest that these markers have predictive value in multiple diseases, but evidence supporting their use in sepsis, especially SICM, remains limited.[17-20]
Given the adverse impact of SICM on patient outcomes and the limitations of existing single inflammatory markers or myocardial injury indicators in clinical prediction, this study aimed to explore the early predictive value of hs-cTnT combined with PBIIs (SII, NLR, PLR, SIRI, and PIV) for identifying SICM in patients with sepsis, with the goal of providing a simple, accessible, and clinically practical predictive approach.
Methods
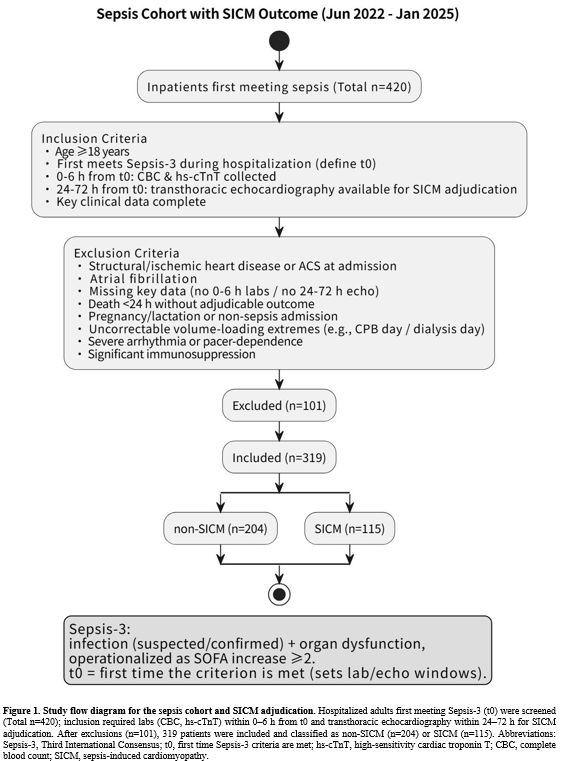
Study population and inclusion and exclusion criteria. Adult patients (aged ≥18 years) who first met the definition of sepsis during hospitalization between June 2022 and January 2025 were included. The time point at which the criteria were first met was defined as t0. Participants were required to undergo a complete blood count and hs-cTnT testing within 0–6 hours after t0, and to have an interpretable transthoracic echocardiogram within 24–72 hours after t0. Key clinical data, including demographics, comorbidities, vital signs, therapeutic interventions, and organ function scores, were also required. Exclusion criteria were as follows: previously diagnosed structural or ischemic heart disease; acute coronary syndrome not ruled out or confirmed on admission; atrial fibrillation; failure to undergo blood count or hs-cTnT testing within 0–6 hours after t0, or absence of interpretable echocardiography within 24–72 hours; death within 24 hours of admission with unattainable outcome assessment; pregnancy or lactation; non-sepsis-related hospitalization; significant and uncorrectable volume overload; severe arrhythmia or pacemaker dependency; and significant immunosuppression. The study obtained ethical approval and used de-identified data.Outcome assessment of Sepsis-3 and SICM. According to the Third International Consensus on Sepsis and Septic Shock,[20] Sepsis-3 is defined as organ dysfunction occurring in the context of, or with strong suspicion of, infection, with an operational criterion of a ≥2-point increase in the SOFA score from baseline. The time point at which this criterion was first met was defined as t0, which determined the time windows for laboratory sampling and ultrasound assessment in this study. The outcome was SICM, independently assessed by two qualified sonographers under blinded conditions. Discrepancies were resolved by a third expert, and cases with poor image quality were considered uninterpretable. SICM was diagnosed when any one of the following criteria was met: ① left ventricular ejection fraction (LVEF) <50% or a decrease of ≥10% from baseline; ② absolute global longitudinal strain (GLS) <16%; or ③ right ventricular dysfunction (TAPSE <17 mm or tricuspid annular S′ <10 cm/s).[21-23] Cases meeting these criteria were classified as SICM+, and the remainder as non-SICM. Echocardiographic assessments used for classification were limited to examinations performed within 24–72 hours after t0; when multiple examinations were available, the first interpretable result was used.
Clinical Data Collection and Variables. The first available value within the 0–24-hour window after t0 was extracted using a standardized template. Collected data included demographics and comorbidities (e.g., age, sex, and chronic disease history), vital signs and therapeutic support (e.g., blood pressure, heart rate, mechanical ventilation, and hemodynamic/device support), and organ function scores (e.g., SOFA). All information was obtained from routine hospital records, and ultrasound interpreters remained blinded to the clinical and laboratory data.
Laboratory and Parameter Calculation. Blood was collected within 0–6 hours after t0 according to hospital standard operating procedures, using either peripheral venipuncture or an indwelling venous catheter. Two milliliters of EDTA-K₂ anticoagulated whole blood were collected for complete blood count analysis on a Beckman Coulter hematology analyzer. In addition, 3–5 mL of blood was collected into a serum separator tube for immunological and biochemical assays (hs-cTnT, CRP, PCT, and IL-6). After complete coagulation, serum samples were centrifuged at 1,500 g for 10 minutes at room temperature, and testing and quality control were performed on the Beckman Coulter immunoassay platform according to the manufacturer's instructions. PBIIs were calculated from the concurrent complete blood count as follows: NLR = Neu/Lym, PLR = Plt/Lym, SII = (Plt×Neu)/Lym, SIRI = (Neu×Mon)/Lym, and PIV = (Neu×Plt×Mon)/Lym.
Statistical analysis. Statistical analyses were performed in R 4.4.3, using the main packages glmnet, pROC, rms, and rmda. Univariate logistic regression was first conducted for each candidate variable, and the associations with the outcome were presented as odds ratios (ORs) with 95% confidence intervals (CIs). Candidate variables were then entered into multivariable logistic regression. The penalty parameter for LASSO regression (α=1) was selected by 10-fold cross-validation, with preference given to λ.1se. Stability was further assessed using bootstrap resampling (B=500). The final model was established on the basis of the screening results, and individual predicted probabilities were calculated.
Model discrimination was evaluated using ROC curves and the AUC with DeLong 95% CIs. Thresholds were determined using Youden's index, and the corresponding sensitivity, specificity, false-positive rate, false-negative rate, Youden's index, threshold value, and P value were reported. Calibration was assessed by comparing predicted probabilities with observed event rates across deciles; an apparent LOESS calibration curve (span=0.75) was plotted, bootstrap bias correction was performed with 500 resamples, and a Hosmer–Lemeshow test (g=10) was conducted. Decision curve analysis (DCA) was used to calculate net benefit across threshold probabilities from 0.05 to 0.95. All statistical tests were two-sided, and P<0.05 was considered statistically significant.
Results
Baseline characteristics. This study enrolled 319 hospitalized patients with sepsis who met the inclusion criteria, including 115 patients (36.1%) in the SICM group and 204 in the non-SICM group (Figure 1). No statistically significant differences were observed between the two groups in age, sex, or BMI (all P>0.05). Regarding vital signs, the SICM group had lower systolic blood pressure (112±13 vs 115±13 mmHg, P=0.049) and diastolic blood pressure (62±8 vs 66±8 mmHg, P<0.001), whereas heart rate was similar between groups (93.8±11.1 vs 93.0±11.0 beats/min, P=0.502). With respect to hematologic and inflammatory markers, the SICM group had higher white blood cell and neutrophil counts, together with lower lymphocyte and monocyte counts (all P<0.001). C-reactive protein, procalcitonin, and IL-6 levels were also significantly higher (all P<0.001), whereas platelet counts did not differ significantly (P=0.749). Regarding therapeutic support, the SICM group more frequently received mechanical ventilation (41.7% vs 24.5%, P=0.002) and hemodynamic/mechanical circulatory support (20.0% vs 4.4%, P<0.001), with a trend toward a difference in renal replacement therapy (16.5% vs 8.8%, P=0.060). Among comorbidities, hypertension was more common in the SICM group (40.0% vs 27.5%, P=0.029), whereas chronic kidney disease and coronary heart disease did not differ significantly. The SICM group also had higher SOFA scores (9.34±1.18 vs 8.89±1.19, P=0.001) (Table 1).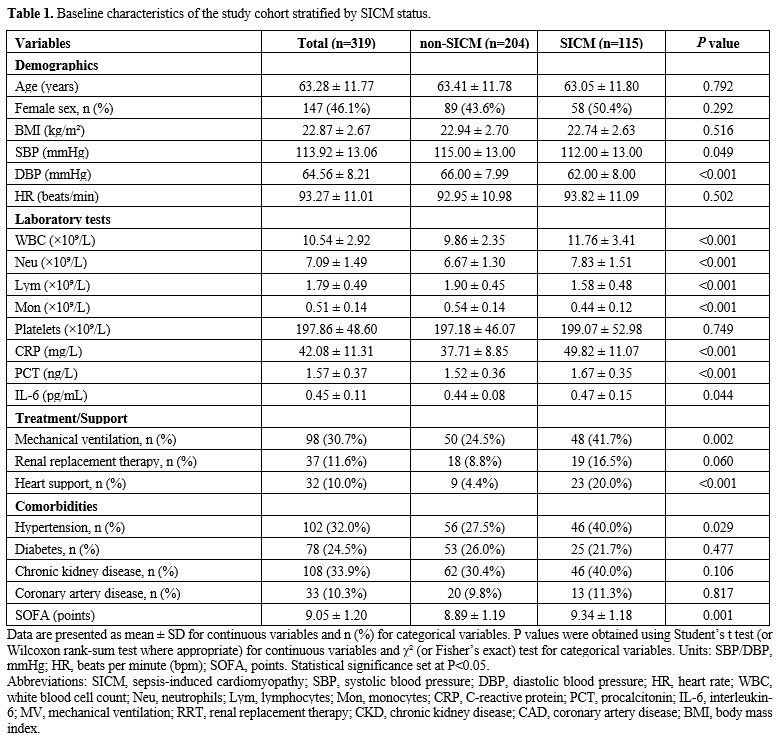 |
Table 1. Baseline characteristics of the study cohort stratified by SICM status. |
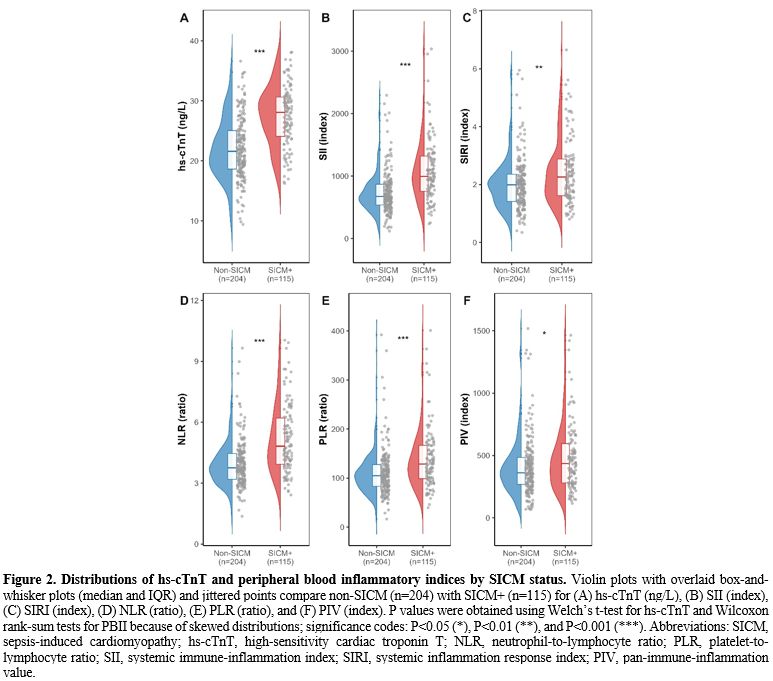
Distribution of hs-cTnT and peripheral inflammatory immune markers stratified by SICM. Among the 319 patients with sepsis, those with SICM had significantly higher hs-cTnT levels than those without SICM (27.44 ± 4.99 vs 22.05 ± 5.10 ng/L, P<0.001), indicating more severe myocardial injury. Peripheral inflammatory immune indices were also generally higher in the SICM group: NLR was 4.82 [3.94, 6.21] vs 3.75 [3.19, 4.46] (P<0.001), SII was 996.50 [756.16, 1318.82] vs 674.63 [539.62, 870.17] (P<0.001), PLR was 128.65 [98.73, 166.59] vs 104.78 [83.29, 127.59] (P<0.001), SIRI was 2.26 [1.62, 2.88] vs 1.99 [1.42, 2.35] (P=0.001), and PIV was 435.94 [280.18, 595.86] vs 361.61 [267.88, 485.28] (P=0.016) (Figure 2). Among these indices, the between-group differences were most pronounced for SII and NLR, whereas the differences for SIRI and PIV were smaller but still statistically significant. Overall, these findings suggest that patients with SICM exhibit more marked abnormalities in both myocardial injury markers and inflammation-related indices.
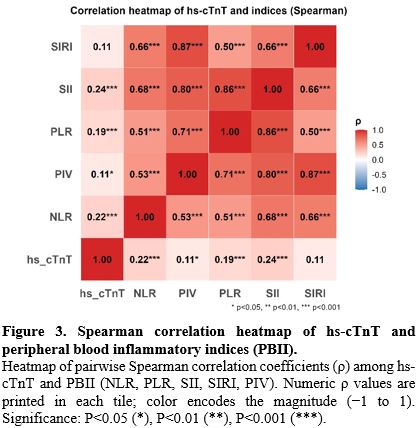
Correlation between hs-cTnT and PBII. Spearman correlation analysis showed that hs-cTnT was weakly correlated with the peripheral inflammatory indices (ρ=0.11–0.24). Specifically, the correlation coefficient was 0.22 with NLR (P<0.001), 0.19 with PLR (P<0.001), 0.24 with SII (P<0.001), and 0.11 with PIV (P<0.05), whereas the correlation with SIRI was not statistically significant (ρ=0.11, P≥0.05). In contrast, significant and strong positive correlations were observed among the inflammatory indices themselves. For example, the correlation coefficient was 0.87 between PIV and SIRI (P<0.001), 0.86 between PLR and SII (P<0.001), 0.80 between SII and PIV (P<0.001), 0.68 between NLR and SII (P<0.001), and 0.66 between NLR and SIRI (P<0.001). These results indicate substantial collinearity among the inflammatory indices, whereas hs-cTnT shows only weak positive correlations with PBII (Figure 3).
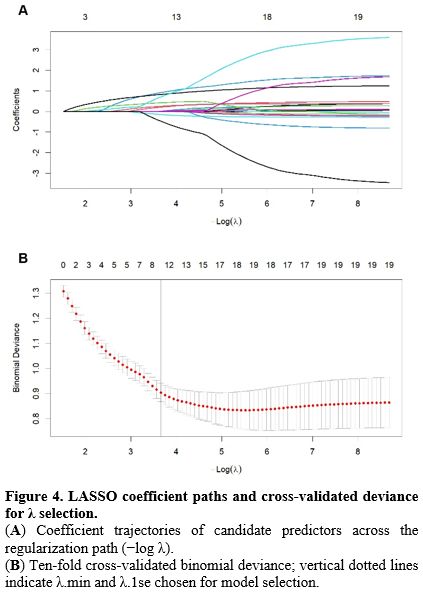
LASSO-Based Variable Selection and Stability Assessment. For variable selection, hs-cTnT, the various PBIIs, and clinical covariates were entered into LASSO logistic regression. The penalty parameter was selected using 10-fold cross-validation, and stability was further assessed with 500 bootstrap resamples (Figure 4). Variables with a stable selection frequency of at least 70% included hs-cTnT, SII, NLR, PIV, SOFA, systolic blood pressure, hemodynamic/mechanical circulatory support, and mechanical ventilation. Among these, hs-cTnT had a selection frequency of 100%, SII 99.4%, NLR 98.4%, PIV 96.2%, SOFA 97.6%, systolic blood pressure 91.2%, cardiac support 99.4%, and mechanical ventilation 76.6%. These findings suggest robust predictive contributions from myocardial injury markers, inflammatory indices, disease severity scores, and support-related variables for SICM prediction.
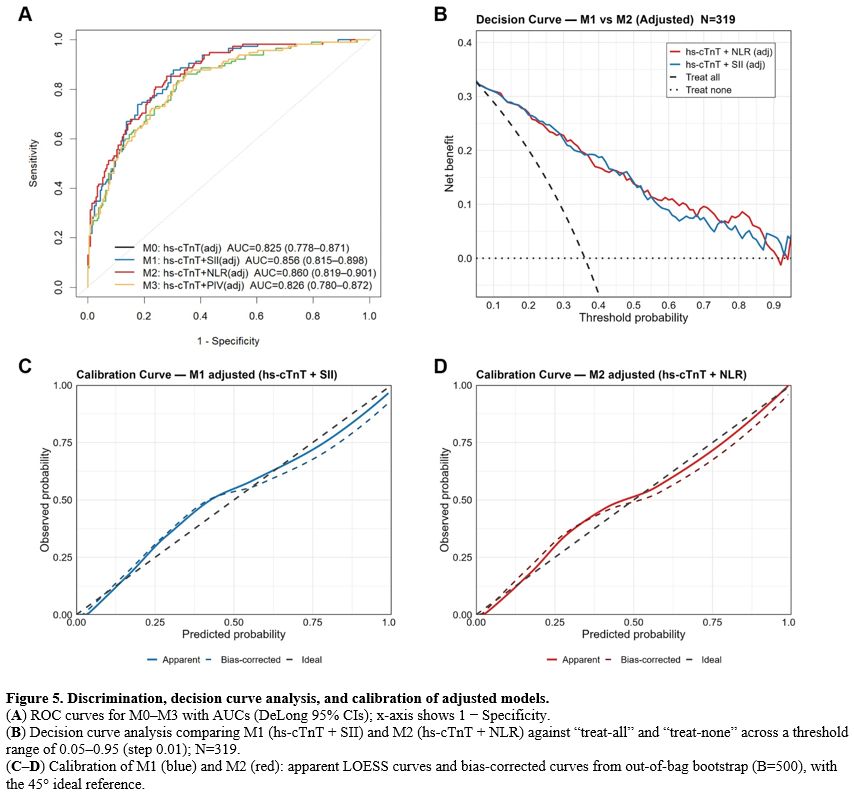
hs-cTnT Combined with elevated PBII for SICM discrimination and calibration. In multivariable logistic regression analysis, hs-cTnT remained an independent predictor, and after the addition of PBII, both SII and NLR also remained independently associated with SICM, whereas PIV did not (Table 2). In terms of discriminative performance, the AUC of the baseline model M0 (hs-cTnT, adjusted for Heart support, SOFA, MV, and SBP) was 0.825 (95% CI 0.778–0.871). After incorporating PBII, the AUC increased to 0.856 for M1 (hs-cTnT + SII; 95% CI 0.815–0.898, DeLong P=0.009) and to 0.860 for M2 (hs-cTnT + NLR; 95% CI 0.819–0.901, P=0.008), both significantly higher than M0. By contrast, M3 (hs-cTnT + PIV) had an AUC of 0.826 (95% CI 0.780–0.872), which was similar to M0 (P=0.694) (Figure 5A, Table 3). At the optimal Youden threshold, M1 and M2 showed higher Youden indices than M0 (approximately 0.57 vs 0.52), with improved specificity while maintaining high sensitivity. M1 showed a greater gain in sensitivity, whereas M2 showed a greater gain in specificity. Decision curve analysis showed that, across threshold probabilities of 0.05–0.95, M1 and M2 generally provided higher net benefit than the treat-all and treat-none strategies, particularly within the clinically relevant range of 0.10–0.60 (Figure 5B). Calibration plots showed that the apparent and bias-corrected curves of M1 and M2 were generally close to the ideal 45° line. Agreement between predicted and observed probabilities was good in the low- to medium-risk range. Although slight deviation was observed at the high-risk end, this was attenuated after bias correction, with no clear evidence of systematic over- or under-prediction (Figure 5C–D). Overall, adding SII or NLR to hs-cTnT improved model discrimination while preserving acceptable calibration, whereas PIV provided limited incremental value.
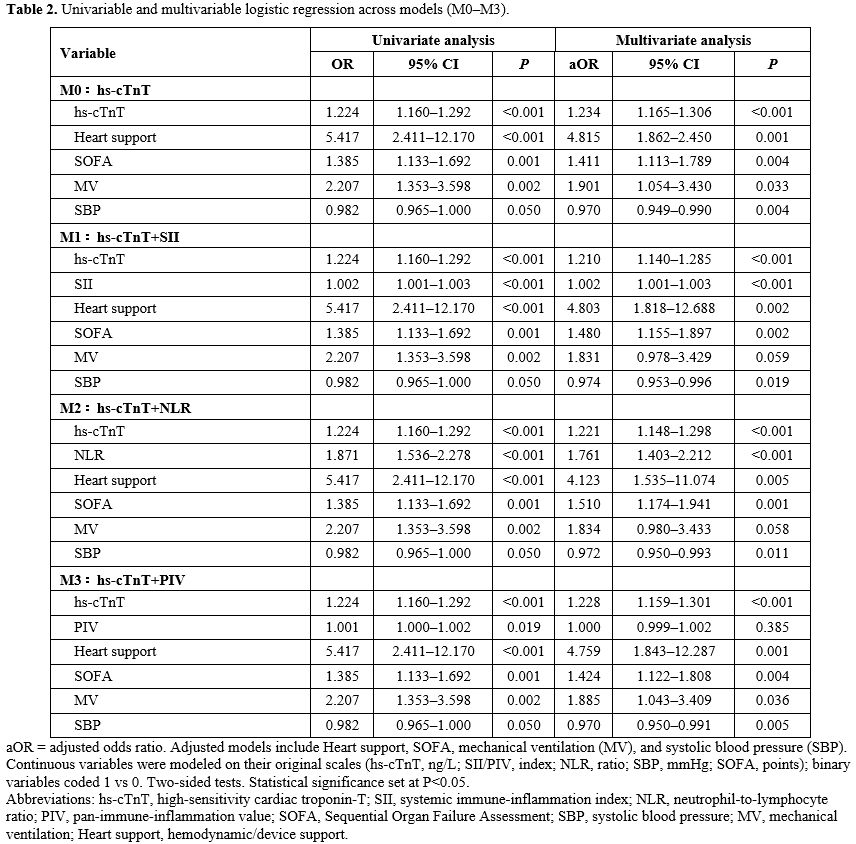 |
Table 2. Univariable and multivariable logistic regression across models (M0–M3). |
Discussion
Previous studies have consistently shown that SICM is a common complication of sepsis and is closely associated with adverse outcomes. Reported incidence rates range from approximately 20% to 43%. The incidence observed in the present study was 36.1%, which is broadly consistent with previous reports.[24-27] The development of SICM not only aggravates circulatory dysfunction but may also contribute to the progression of multiple organ failure. Accordingly, early prediction remains an important challenge in critical care medicine.Traditional studies have largely focused on single biomarkers, including myocardial injury markers such as troponin and inflammatory markers such as CRP, PCT, and IL-6. Although these indicators can partly reflect myocardial injury or systemic inflammation, their predictive value is still limited when used alone. For example, one study reported that elevated cardiac troponin I (cTnI) was an independent predictor of short-term mortality in patients with septic shock, but its standalone accuracy was insufficient.[28] Another study found that combining blood glucose with hs-cTnT/I improved early risk identification in acute myocardial infarction, but the incremental diagnostic gain over hs-cTn alone was limited.[29] In patients with sepsis, admission hs-cTnT levels were significantly associated with 30-day and 1-year mortality, and among acute-phase survivors, hs-cTnT also remained predictive of mortality from 30 to 365 days.[30] In addition, some studies have suggested that although cTnI, CRP, and NLR are independent risk factors, their individual predictive performance is modest, supporting the rationale for combined assessment.[31]
In recent years, PBIIs have attracted increasing attention. These indices are derived from routine blood count parameters and have the advantages of simplicity, low cost, and high accessibility, while also reflecting inflammatory burden and immune status. Existing studies suggest that NLR, SII, PLR, SIRI, and PIV all have clinical value across multiple diseases.[17-20,32,33] Among them, NLR and SII are thought to better capture inflammatory burden and immune dysregulation. Multiple studies have confirmed their association with infection-related adverse outcomes, providing theoretical support for their use in sepsis-associated myocardial injury.[34,35]
NLR has accumulated substantial supporting evidence in sepsis populations. Ni et al. reported in an emergency department cohort that admission NLR was significantly associated with in-hospital mortality.[36] Li et al. analyzed the MIMIC-IV database and found that elevated NLR levels were independently associated with 28-day all-cause mortality in sepsis patients with concomitant coronary artery disease.[37] Zhang et al. further showed in a large database study that the time-weighted average NLR was nonlinearly associated with 90-day in-hospital mortality, suggesting potential value for dynamic monitoring.[38] In studies of septic cardiomyopathy, Lan et al. identified NLR as an independent risk factor, and its diagnostic performance improved when combined with CRP and PLR.[31]
Research on SII is also expanding. Ou et al. found in patients with bloodstream infections that both SII and NLR were significantly associated with mortality risk, with nonlinear effects.[34] A review by Islam et al. emphasized the stability and reproducibility of SII and NLR under infectious stress conditions.[35] A meta-analysis of nine cohorts showed that high admission SII was associated with increased short-term mortality in sepsis.[40] Mangalesh et al. further suggested that combining SII with the SOFA score may improve mortality risk prediction in sepsis.[41] Chen et al. reported a U-shaped association between SII and hypertension risk in the NHANES cohort,[39] which, although outside the sepsis setting, supports the broader biological relevance of SII.
Overall, both NLR and SII can reflect inflammatory burden and immune dysregulation to some extent, and both show potential value for risk assessment in sepsis and its complications. On this basis, the present study further combined NLR and SII with hs-cTnT to evaluate their potential for early identification of SICM. The results showed that hs-cTnT levels were markedly elevated in patients with SICM, indicating more severe myocardial injury. Multiple PBIIs were also elevated, supporting the involvement of inflammatory burden and immune dysregulation in this condition. Among these indices, SII and NLR showed the most pronounced between-group differences. When combined with hs-cTnT in multivariable models, both markers improved discrimination, maintained acceptable calibration, and yielded more favorable net benefit on decision curve analysis. These findings support the complementary roles of inflammatory and myocardial injury markers in early SICM risk assessment. Similarly, Lan et al. reported that the combination of NLR, CRP, and PLR achieved good predictive accuracy for SICM, further supporting the value of multi-marker integration.[31]
It should also be noted that previous studies of NLR and SII have mainly focused on mortality or longer-term prognosis in sepsis,[34-40] and this body of evidence provides an important background for their application in critically ill populations. However, compared with mortality, SICM is a more proximal and potentially reversible complication. Earlier recognition of patients at higher risk of SICM may facilitate closer monitoring and timely supportive management. From this perspective, evaluating these inflammatory markers during the early phase of SICM may be more clinically informative than using them only for mortality stratification. In this context, the present findings suggest that combining hs-cTnT with either NLR or SII may provide a practical approach for early SICM risk assessment. Future studies should validate these findings in larger multicenter cohorts and determine whether the combined biomarker strategy remains informative in other inflammation-related cardiovascular settings. Exploratory questions may include conditions such as restenosis, post-cardiac inflammatory syndromes, including Dressler’s syndrome, or treatment contexts involving biologic drugs. These extensions remain speculative and require dedicated prospective evaluation.
Conclusions
This study shows that combining hs-cTnT with PBIIs, particularly SII and NLR, may improve early risk assessment for SICM in patients with sepsis. Compared with single markers, these combined models showed better discrimination together with acceptable calibration and decision-curve performance. Because these markers are derived from routine laboratory tests, they are easy to obtain and may have practical value. Further validation in larger, multicenter cohorts is still needed. Integration with cardiac imaging and other multimodal approaches may provide a more robust basis for the early identification of sepsis-associated cardiomyopathy.Funding
2025 Jiangsu Provincial Research Hospital Association Special Research Fund Project (SYHKJ-XF-2025-05). Nantong University Clinical Medicine Research Fund (2024LQ013) Nantong Municipal Health Commission Fund (MS2025025 QNZ2025018).Data available
Data is available from the corresponding author on request.Ethics statement
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by Nantong First People’s Hospital.Author’s Contribution
Conceptualization, ChunYan Zong and Xin Chu, methodology, BaoFeng Zhu and FeiFeng Wu, formal analysis, FeiFeng Wu and FenFen Chen; investigation, JiaoJiao Yang, data curation, Hong Zhou, writing original draft preparation, ChunYan Zong. Writing review and editing, BaoFeng Zhu. All author shave read and agreed to the published version of the manuscript.References
- Borges A, Bento L. Organ crosstalk and dysfunction in sepsis. Ann Intensive Care. 2024;14(1):147. https://doi.org/10.1186/s13613-024-01377-0 PMid:39298039 PMCid:PMC11413314
- Gauer R, Forbes D, Boyer N. Sepsis: Diagnosis and Management. Am Fam Physician. 2020;101(7):409-18.
- Aissaoui N, Boissier F, Chew M, Singer M, Vignon P. Sepsis-induced cardiomyopathy. Eur Heart J. 2025;46(34):3339-53. https://doi.org/10.1093/eurheartj/ehaf340 PMid:40439150
- Singer
M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al.
The Third International Consensus Definitions for Sepsis and Septic
Shock (Sepsis-3). Jama. 2016;315(8):801-10. https://doi.org/10.1001/jama.2016.0287 PMid:26903338 PMCid:PMC4968574
- Hollenberg
SM. Sepsis-Associated Cardiomyopathy: Long-Term Prognosis, Management,
and Guideline-Directed Medical Therapy. Curr Cardiol Rep. 2025;27(1):5.
https://doi.org/10.1007/s11886-024-02175-7 PMid:39776326
- Hollenberg SM, Singer M. Pathophysiology of sepsis-induced cardiomyopathy. Nat Rev Cardiol. 2021;18(6):424-34. https://doi.org/10.1038/s41569-020-00492-2 PMid:33473203
- Lin
H, Wang W, Lee M, Meng Q, Ren H. Current Status of Septic
Cardiomyopathy: Basic Science and Clinical Progress. Front Pharmacol.
2020;11:210. https://doi.org/10.3389/fphar.2020.00210 PMid:32194424 PMCid:PMC7062914
- Kuroshima
T, Kawaguchi S, Okada M. Current Perspectives of Mitochondria in
Sepsis-Induced Cardiomyopathy. Int J Mol Sci. 2024;25(9). https://doi.org/10.3390/ijms25094710 PMid:38731929 PMCid:PMC11083471
- Song
J, Fang X, Zhou K, Bao H, Li L. Sepsis‑induced cardiac dysfunction and
pathogenetic mechanisms (Review). Mol Med Rep. 2023;28(6). https://doi.org/10.3892/mmr.2023.13114 PMid:37859613 PMCid:PMC10619129
- Fan
D, Wu R. Mechanisms of the septic heart: From inflammatory response to
myocardial edema. J Mol Cell Cardiol. 2024;195:73-82. https://doi.org/10.1016/j.yjmcc.2024.08.003 PMid:39142438
- Lörstad
S, Wang Y, Tehrani S, Shekarestan S, Åstrand P, Gille-Johnson P, et al.
Development of an Extended Cardiovascular SOFA Score Component
Reflecting Cardiac Dysfunction with Improved Survival Prediction in
Sepsis: An Exploratory Analysis in the Sepsis and Elevated Troponin
(SET) Study. J Intensive Care Med. 2025;40(3):320-30. https://doi.org/10.1177/08850666241282294 PMid:39350606 PMCid:PMC11915778
- Røsjø
H, Varpula M, Hagve TA, Karlsson S, Ruokonen E, Pettilä V, et al.
Circulating high sensitivity troponin T in severe sepsis and septic
shock: distribution, associated factors, and relation to outcome.
Intensive Care Med. 2011;37(1):77-85. https://doi.org/10.1007/s00134-010-2051-x PMid:20938765 PMCid:PMC3020309
- Violi
F, Calvieri C, Cangemi R. Effect of corticosteroids on myocardial
injury among patients hospitalized for community-acquired pneumonia:
rationale and study design. The colosseum trial. Intern Emerg Med.
2020;15(1):79-86. https://doi.org/10.1007/s11739-019-02117-0 PMid:31152308 PMCid:PMC7223724
- deFilippi
C, McCallum S, Zanni MV, Fitch KV, Diggs MR, Bloomfield GS, et al.
Association of Cardiac Troponin T With Coronary Atherosclerosis in
Asymptomatic Primary Prevention People With HIV. JACC Adv.
2024;3(9):101206. https://doi.org/10.1016/j.jacadv.2024.101206 PMid:39253712 PMCid:PMC11381782
- Hu
W, Huang Y, Zhou Q, Huang X. Relation between peripheral blood
inflammatory indices and severity of central retinal artery occlusion.
PeerJ. 2024;12:e18129. https://doi.org/10.7717/peerj.18129 PMid:39364366 PMCid:PMC11448653
- Shen
X, Xiang M, Tang J, Xiong G, Zhang K, Xia T, et al. Evaluation of
peripheral blood inflammation indexes as prognostic markers for
colorectal cancer metastasis. Sci Rep. 2024;14(1):20489. https://doi.org/10.1038/s41598-024-68150-y PMid:39227608 PMCid:PMC11372090
- Rezazadeh
M, Kamyabi A, Pisheh RG, Noroozie S, Amiri BS, Negahi A, et al.
Diagnostic value of peripheral blood inflammatory indices for
clinicopathological profile of colorectal cancer: a retrospective
observational study. BMC Gastroenterol. 2025;25(1):127. https://doi.org/10.1186/s12876-025-03681-w PMid:40033197 PMCid:PMC11874807
- Li
D, Liu L, Lv J, Xiong X. The Clinical significance of Peripheral
Blood-related Inflammatory Markers in patients with AECOPD.
Immunobiology. 2025;230(3):152903. https://doi.org/10.1016/j.imbio.2025.152903 PMid:40286421
- Hu
H, Chen WJ, Sun C, Xie JP. D-D and PLR, NLR combined forecasting
first-line treatment: The short-term curative effect and prognosis of
patients with SCLC. Medicine (Baltimore). 2023;102(45):e34877. https://doi.org/10.1097/MD.0000000000034877 PMid:37960828 PMCid:PMC10637545
- Ramasamy
J, Murugiah V, Dhanapalan A, Balasubramaniam G. Diagnostic Utility of
Pan-Immune-Inflammation Value (PIV) in Predicting Insulin Resistance:
Results from the National Health and Nutrition Examination Survey
(NHANES) 2017-2020. Ejifcc. 2024;35(2):100-10. https://doi.org/10.21203/rs.3.rs-3945289/v1
- L'Heureux
M, Sternberg M, Brath L, Turlington J, Kashiouris MG. Sepsis-Induced
Cardiomyopathy: a Comprehensive Review. Curr Cardiol Rep.
2020;22(5):35. https://doi.org/10.1007/s11886-020-01277-2 PMid:32377972 PMCid:PMC7222131
- Nyberg
J, Jakobsen EO, Østvik A, Holte E, Stølen S, Lovstakken L, et al.
Echocardiographic Reference Ranges of Global Longitudinal Strain for
All Cardiac Chambers Using Guideline-Directed Dedicated Views. JACC
Cardiovasc Imaging. 2023;16(12):1516-31. https://doi.org/10.1016/j.jcmg.2023.08.011 PMid:37921718
- Hameed
A, Condliffe R, Swift AJ, Alabed S, Kiely DG, Charalampopoulos A.
Assessment of Right Ventricular Function-a State of the Art. Curr Heart
Fail Rep. 2023;20(3):194-207. https://doi.org/10.1007/s11897-023-00600-6 PMid:37271771 PMCid:PMC10256637
- Hasegawa
D, Ishisaka Y, Maeda T, Prasitlumkum N, Nishida K, Dugar S, et al.
Prevalence and Prognosis of Sepsis-Induced Cardiomyopathy: A Systematic
Review and Meta-Analysis. J Intensive Care Med. 2023;38(9):797-808. https://doi.org/10.1177/08850666231180526 PMid:37272081
- Liang
YW, Zhu YF, Zhang R, Zhang M, Ye XL, Wei JR. Incidence, prognosis, and
risk factors of sepsis-induced cardiomyopathy. World J Clin Cases.
2021;9(31):9452-68. https://doi.org/10.12998/wjcc.v9.i31.9452 PMid:34877280 PMCid:PMC8610866
- Sha
T, Jiang H, Bai L. An interpretable machine learning model for
predicting sepsis-induced cardiomyopathy in ICU patients: development
and validation using the MIMIC-IV database. Health Inf Sci Syst.
2025;13(1):49. https://doi.org/10.1007/s13755-025-00367-1 PMid:40814334 PMCid:PMC12343452
- Lu
NF, Jiang L, Zhu B, Yang DG, Zheng RQ, Shao J, et al. Elevated Plasma
Histone H4 Levels Are an Important Risk Factor in the Development of
Septic Cardiomyopathy. Balkan Med J. 2020;37(2):72-8. https://doi.org/10.4274/balkanmedj.galenos.2019.2019.8.40
- Yang
Z, Qdaisat A, Hu Z, Wagar EA, Reyes-Gibby C, Meng QH, et al. Cardiac
Troponin Is a Predictor of Septic Shock Mortality in Cancer Patients in
an Emergency Department: A Retrospective Cohort Study. PLoS One.
2016;11(4):e0153492. https://doi.org/10.1371/journal.pone.0153492 PMid:27077648 PMCid:PMC4831781
- Yufera-Sanchez
A, Lopez-Ayala P, Nestelberger T, Wildi K, Boeddinghaus J, Koechlin L,
et al. Combining glucose and high-sensitivity cardiac troponin in the
early diagnosis of acute myocardial infarction. Sci Rep.
2023;13(1):14598. https://doi.org/10.1038/s41598-023-37093-1 PMid:37670005 PMCid:PMC10480296
- Lörstad
S, Shekarestan S, Jernberg T, Tehrani S, Åstrand P, Gille-Johnson P, et
al. First Sampled High-Sensitive Cardiac Troponin T is Associated With
One-Year Mortality in Sepsis Patients and 30- to 365-Day Mortality in
Sepsis Survivors. Am J Med. 2023;136(8):814-23.e8. https://doi.org/10.1016/j.amjmed.2023.04.029 PMid:37156347
- Lan
X, Mai H. Predictive value of combined inflammatory markers for septic
cardiomyopathy: a retrospective study. BMC Infect Dis. 2025;25(1):1033.
https://doi.org/10.1186/s12879-025-11447-8 PMid:40826335 PMCid:PMC12363019
- Wen
X, Wang F, Tang T, Xu B, Yuan M, Li Y, et al. Sex-specific association
of peripheral blood cell indices and inflammatory markers with
depressive symptoms in early adolescence. J Affect Disord.
2024;362:134-44. https://doi.org/10.1016/j.jad.2024.06.098 PMid:38960333
- Yang
W., Dong W.Heparin-binding protein as a diagnostic and prognostic
marker of infections: A systematic review and meta-analysis. Mediterr J
Hematol Infect Dis 2025, 17(1): e2025029, https://doi.org/10.4084/MJHID.2025.029 PMid:40375914 PMCid:PMC12081054
- Ou
S, Lu H, Qu R, Cui X, Xiong Z, Fan F, et al. The Clinical Value of
Systemic Immune Inflammatory Index in Predicting the Prognosis of
Patients with Bloodstream Infection. J Inflamm Res. 2025;18:10181-92. https://doi.org/10.2147/JIR.S531272 PMid:40761379 PMCid:PMC12318860
- Islam
MM, Satici MO, Eroglu SE. Unraveling the clinical significance and
prognostic value of the neutrophil-to-lymphocyte ratio,
platelet-to-lymphocyte ratio, systemic immune-inflammation index,
systemic inflammation response index, and delta neutrophil index: An
extensive literature review. Turk J Emerg Med. 2024;24(1):8-19. https://doi.org/10.4103/tjem.tjem_198_23 PMid:38343523 PMCid:PMC10852137
- Ni
J, Wang H, Li Y, Shu Y, Liu Y. Neutrophil to lymphocyte ratio (NLR) as
a prognostic marker for in-hospital mortality of patients with sepsis:
A secondary analysis based on a single-center, retrospective, cohort
study. Medicine (Baltimore). 2019;98(46):e18029. https://doi.org/10.1097/MD.0000000000018029 PMid:31725679 PMCid:PMC6867781
- Li
X, Chen Y, Yuan Q, Zhou H, Lu L, Guo R. Neutrophil-to-lymphocyte ratio,
monocyte-to-lymphocyte ratio, platelet-to-lymphocyte ratio associated
with 28-day all-cause mortality in septic patients with coronary artery
disease: a retrospective analysis of MIMIC-IV database. BMC Infect Dis.
2024;24(1):749. https://doi.org/10.1186/s12879-024-09516-5 PMid:39075364 PMCid:PMC11288105
- Zhang
G, Wang T, An L, Hang C, Wang X, Shao F, et al. The
neutrophil-to-lymphocyte ratio levels over time correlate to all-cause
hospital mortality in sepsis. Heliyon. 2024;10(16):e36195. https://doi.org/10.1016/j.heliyon.2024.e36195 PMid:39253154 PMCid:PMC11381600
- Chen
Y, Li Y, Liu M, Xu W, Tong S, Liu K. Association between systemic
immunity-inflammation index and hypertension in US adults from NHANES
1999-2018. Sci Rep. 2024;14(1):5677. https://doi.org/10.1038/s41598-024-56387-6 PMid:38454104 PMCid:PMC10920861
- Liang
L, Su Q. Systemic immune-inflammation index and the short-term
mortality of patients with sepsis: A meta-analysis. Biomol Biomed.
2025;25(4):798-809. https://doi.org/10.17305/bb.2024.11494 PMid:39739368 PMCid:PMC11959392
- Mangalesh
S, Dudani S, Malik A. The systemic immune-inflammation index in
predicting sepsis mortality. Postgrad Med. 2023;135(4):345-51. https://doi.org/10.1080/00325481.2022.2140535 PMid:36287784