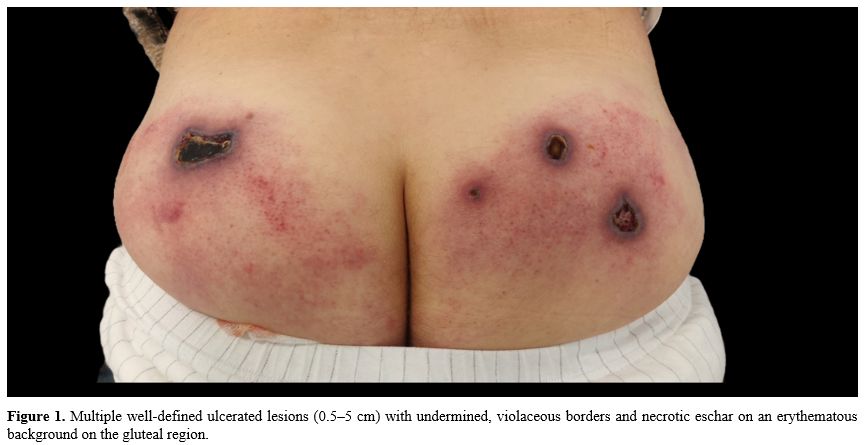
A 56-year-old man was referred for a five-week history of painful ulcerated lesions in the gluteal area, initially treated by his general physician with valaciclovir 1000 mg three times daily for seven days on suspicion of varicella zoster virus (VZV) infection, with gradual worsening. Physical examination revealed four well-defined, ulcerated lesions, ranging from 0.5 to 5 cm in diameter, with undermined, violaceous borders covered by a necrotic eschar atop a diffusely erythematous background (Figure 1). The patient had been diagnosed four years earlier with CLL with trisomy 12, hypermutated IGHV, and wild-type TP53; due to the absence of symptoms, he was not undergoing any treatment and was scheduled for regular follow-up every six months.
 |
|
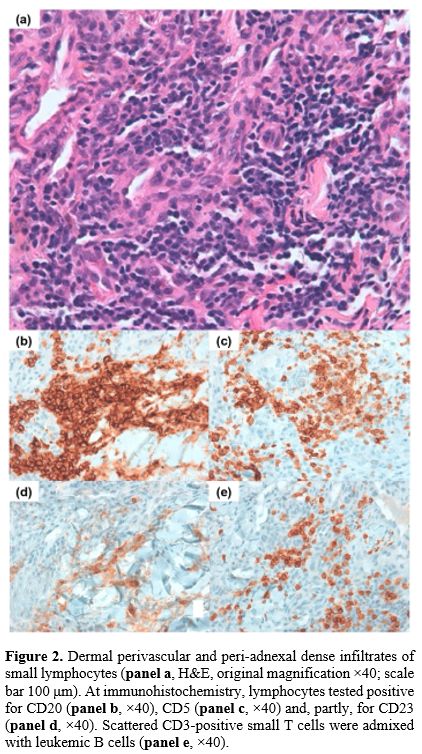
A biopsy of the edge of one lesion was performed. Histopathological examination showed superficial ulceration and extravasation of blood in the superficial dermis beneath a largely spared epidermis. In the deep dermis and subcutaneous tissue, a perivascular and peri-adnexal dense infiltrate of small lymphocytes was observed. Immunohistochemistry documented numerous small T cells (CD3+, CD5+) and clusters of B cells (CD20+, CD79a+) expressing CD5 and CD23 (Figure 2a–e). Direct immunofluorescence (DIF) revealed C1q deposition (+) around blood vessels in the papillary dermis. These findings were consistent with cutaneous infiltration of CLL. This immunophenotypic profile — CD20+ B cells coexpressing CD5 and CD23, admixed with CD3+ small T cells — is characteristic of the CLL/small lymphocytic lymphoma phenotype and supports leukemic skin infiltration by the known CLL clone rather than by another B-cell lymphoproliferative disorder.
Interestingly, the skin lesions spontaneously resolved three weeks after the biopsy. Four weeks after this remission, the patient developed systemic symptoms including fever, malaise, and asthenia, prompting urgent hematologic evaluation. Blood tests showed CLL progression with anemia requiring red blood cell transfusion, and imaging documented widespread lymphadenopathy, leading to the initiation of venetoclax and obinutuzumab. After the second infusion of obinutuzumab, the patient developed sepsis and pancytopenia, requiring broad-spectrum antibiotics and transfusion support; immunotherapy was temporarily discontinued. The patient improved clinically and was later reintroduced to targeted therapy. He completed treatment with venetoclax–obinutuzumab, achieving complete remission without recurrence of dermatological or infectious manifestations at one-year follow-up. A comprehensive summary of the clinical–hematologic timeline of events for this case is presented in Table 1.
LC is a rare and specific manifestation of systemic leukemia characterized by leukemic cell infiltration into the skin. It may occur in several hematologic malignancies, including CLL.[1] In CLL, LC is considered an uncommon manifestation, occurring in only a small proportion of patients and potentially developing months to years after the initial diagnosis, often reflecting changes in the underlying disease status.[2] LC typically presents as papules, nodules, or plaques, and only rarely as ulcers or blisters.[1-3] LC may precede, coincide with, or follow the diagnosis of leukemia, emphasizing the need to include it in the differential diagnosis of new skin lesions in patients with known hematologic disease.[3]
The AML case previously reported in this journal[4] elegantly illustrates LC as the first clue to an undiagnosed hematologic malignancy. In contrast, our patient had a well-established diagnosis of CLL and was under regular surveillance. In this context, LC did not enable a new diagnosis but instead signaled the transition from an indolent to an active disease phase. These two cases, therefore, represent complementary roles of LC: as a diagnostic gateway in de novo leukemia and as a dynamic marker of disease activity in established leukemia.
To the best of our knowledge, this is the first reported CLL case in which LC underwent spontaneous clinical remission before systemic progression. In prior reports, LC in CLL usually precedes or accompanies hematologic deterioration[5,6] and tends to resolve only after effective systemic therapy.[5,7] Furthermore, some cases of LC resolve spontaneously after biopsy without immediate progression, referred to as "aleukemic LC".[1,8,10] In our patient, LC served as a short-lived “alarm bell,” suggesting fluctuations in disease activity and reinforcing the necessity of close monitoring even in the absence of persistent skin findings.
Therapeutically, the patient achieved complete remission with venetoclax–obinutuzumab, with no further cutaneous involvement. Although LC had already regressed spontaneously before treatment initiation, durable hematologic disease control likely prevented additional episodes of skin infiltration. This supports the effectiveness of venetoclax-based regimens in stabilizing CLL in patients who present with LC.
In conclusion, this case highlights three key messages. First, in patients under surveillance for leukemia, new skin lesions warrant careful evaluation, as LC may indicate disease activation. Second, even spontaneously regressing LC should be interpreted as a transient warning sign that merits timely hematologic reassessment. Third, venetoclax-based regimens can provide effective systemic control in CLL associated with LC, underscoring the importance of close collaboration between dermatologists and hematologists.
Ethics Statement
The patient provided written informed consent for the publication of anonymized clinical data and images, in accordance with the Declaration of Helsinki and institutional policies.References
- Robak E, Braun M, Robak T. Leukemia cutis - the current view on pathogenesis, diagnosis, and treatment. Cancers. 2023;15:5393. https://doi.org/10.3390/cancers15225393 PMid:38001655 PMCid:PMC10670312
- Szukalski
Ł, Sysakiewicz-Buda M, Męcińska-Jundziłł K, Białecka A, Czyż J,
Czajkowski R. Cutaneous manifestation of chronic lymphocytic leukemia:
a case report and a literature review. Postepy Dermatol Alergol.
2019;36:778-780. https://doi.org/10.5114/ada.2019.91427 PMid:31998011 PMCid:PMC6986299
- Wagner
G, Fenchel K, Back W, Schulz A, Sachse MM. Leukemia cutis -
epidemiology, clinical presentation, and differential diagnoses. J
Dtsch Dermatol Ges. 2012;10:27-36. https://doi.org/10.1111/j.1610-0387.2011.07842.x PMid:22115500
- Buzzatti
E, Mauro C, Botti C, et al. Leukemia cutis in a patient with acute
myeloid leukemia undergoing azacitidine-venetoclax: case presentation
and review of the literature. Mediterr J Hematol Infect Dis.
2025;17:e2025013. https://doi.org/10.4084/MJHID.2025.013 PMid:40084096 PMCid:PMC11906129
- Cerroni
L, Zenahlik P, Höfler G, Kaddu S, Smolle J, Kerl H. Specific cutaneous
infiltrates of B-cell chronic lymphocytic leukemia. Am J Surg Pathol.
1996;20:1000-1010. https://doi.org/10.1097/00000478-199608000-00009 PMid:8712287
- Lazarian
G, Munger M, Quinquenel A, et al. Clinical and biological
characteristics of leukemia cutis in chronic lymphocytic leukemia: a
study of the French Innovative Leukemia Organization (FILO). Am J
Hematol. 2021;96:E353-E356. https://doi.org/10.1002/ajh.26274
- Pedreira-García
WM, Rodríguez-Lobato LG, Soto-Mera T, et al. Leukemia cutis as the
initial manifestation of chronic lymphocytic leukemia progression.
Cureus. 2022;14:e33013. https://doi.org/10.7759/cureus.33013 PMid:36712707 PMCid:PMC9879596
- Joseph S, Liu Y. Spontaneous resolution of aleukemic leukemia cutis: a case report. Ann Hematol Oncol. 2016;3:1076.
- Raufi
A, Alsharedi M, Khelfa Y, Griswold DC, Lebowicz Y. Leukemia cutis in a
patient with chronic lymphocytic leukemia presenting as bilateral
helical nodules. SAGE Open Med Case Rep. 2016;4:1-4. https://doi.org/10.1177/2050313X16683624 PMid:28228955 PMCid:PMC5308427
- Higaki-Mori H, Yamada N, Ozaki K, Nishimura MF, Yoshida Y. Spontaneous complete regression of aleukaemic cutaneous myeloid sarcoma without progression to leukaemia over a long-term follow-up period. Acta Derm Venereol. 2025;105:adv42339. https://doi.org/10.2340/actadv.v105.42339 PMid:40001342 PMCid:PMC11877853