According to the European LeukemiaNet (ELN) guidelines, a change in TKI therapy should be considered for patients who fail to achieve specific response milestones.[3]
Therefore, accurately predicting the likelihood of treatment failure in individuals with chronic phase CML is crucial to achieve precise response targets when selecting the initial TK.
The risk stratification of CML patients was based on scores predicting overall survival since the pre-imatinib era. The Sokal risk score was published in 1984, while the EURO score was established in 1998 using data from CML patients receiving interferon-α therapy.[4-5] After the introduction of imatinib, the EUTOS risk score was established in 2011 to predict the likelihood of achieving complete cytogenetic response (CCyR) at 18 months, serving as a proxy for survival.[6]
In 2022, Zhang et al.[7] proposed an imatinib therapy failure score (IMTF) for patients affected by chronic phase CML. The score was applied to 1364 patients to predict progression-free survival and overall survival (all p-values < 0.001). These data were also confirmed in a real-world experience.[8]
In 2024, Zhang et al. applied a new scoring system to predict the risk of first-line TKI[9] therapy in patients with CML treated in the chronic phase, as first-line therapy with imatinib or 2-generation TKI. The score is based on six prognostic factors: gender, age, hemoglobin level, blast percentage, spleen size, and the presence of additional cytogenetic abnormalities (ACAs), and was developed by analyzing data from 3,454 patients across 76 centers.
In our monocentric cohort, we retrospectively evaluated 135 patients to determine whether they experienced treatment failure during first-line therapy from 2000 to 2025, with a median follow-up of 8 years. Patients who switched TKI for toxicity reasons were excluded from the analysis. Chronic phase was defined according to World Health Organization (WHO) criteria.[10] Conventional cytogenetic analysis was performed on bone marrow samples using R-banding techniques, with at least 20 metaphases analyzed from both direct and short-term (24-hour) cultures.
Quantitative real-time polymerase chain reaction monitoring was performed at diagnosis and every 3 months thereafter until a major molecular response (MMR) was achieved, and every 3 to 6 months thereafter.[11] TKI-therapy failure was defined as meeting “failure” milestones in the 2020 ELN recommendations: loss of response, including CHR, CCyR, or MMR, or transformation to an advanced phase as defined by the ELN recommendations.[12]
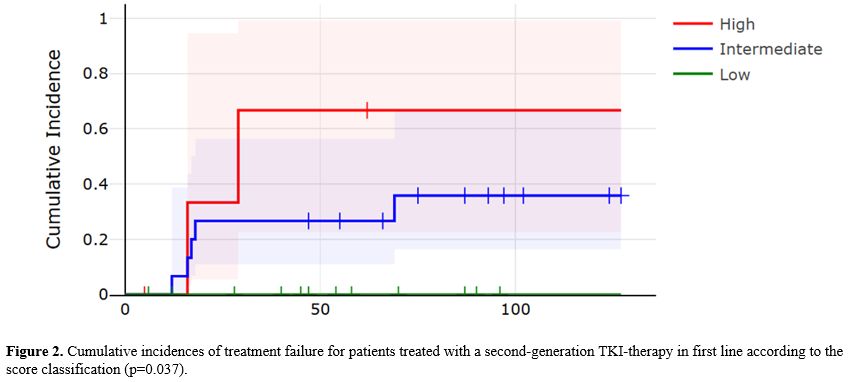
The patients in our cohort were treated with imatinib (75%), dasatinib (12%), or nilotinib (13%). Regarding the Zhang score, the 135 patients were divided into three risk categories: low-risk (48 patients, 36%), intermediate-risk (76 patients, 56%), and high-risk (11 patients, 8%) (Table 1).
In the original validation cohort, 8-year cumulative incidences of treatment failure were 10%, 34%, and 69% for the low-, intermediate-, and high-risk groups, respectively (p < .001).
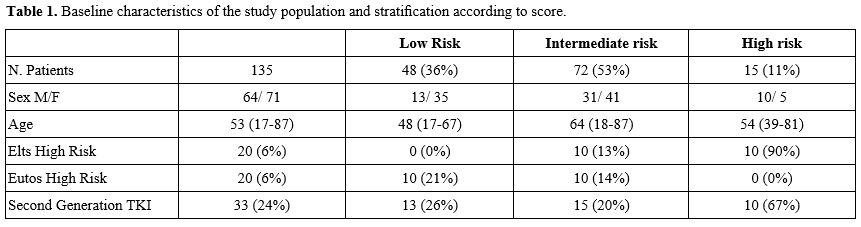
In our cohort, the 8-year cumulative incidence of treatment failure was 18% in the low-risk group, 27% in the intermediate-risk group, and 68% in the high-risk group (p. 0.0002; Figure 1).
Using the low-risk group as the reference, in univariate analysis, the hazard ratios (HRs) for treatment failure were 1.79 (95% CI, 0.72–4.40; p = .21) for the intermediate-risk group and 7.50 (95% CI, 1.8–17.5; p = .002) for the high-risk group. These findings were compared to the original cohort, which reported HRs of 3.8 (95% CI, 2.9–5.0; p < .001) and 10.4 (95% CI, 7.7–14.0; p < .001) for the intermediate- and high-risk groups, respectively.
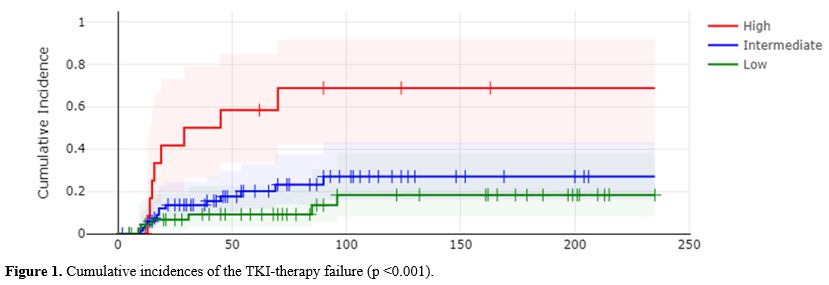
In the cohort of patients treated with second-generation TKIs, the model demonstrated good capacity to stratify risk of treatment failure (p = 0.037; Figure 2). At 8 years, the cumulative incidence of failure was 66% in the high-risk group and 35% in the intermediate-risk group, while no treatment failure occurred among low-risk patients.
 |
|
Despite the limited size and single-center nature of our cohort, our results support the predictive validity of this novel scoring system in a demographically distinct population. Overall, the Zhang score demonstrated good discriminative and predictive accuracy, which could help physicians optimize the selection of initial TKI therapy in clinical practice.
References
- Hehlmann R, Lauseker M,
Saußele S, et al. Assessment of imatinib as first-line treatment of
chronic myeloid leukemia: 10-year survival results of the randomized
CML study IV and impact of non-CML determinants. Leukemia.
2017;31(11):2398-2406. https://doi.org/10.1038/leu.2017.253
PMid:28804124 PMCid:PMC5668495
- Lau
A, Seiter K. Second-line therapy for patients with chronic myeloid
leukemia resistant to first-line imatinib. Clin Lymphoma Myeloma Leuk.
2014 Jun;14(3):186-96. doi: 10.1016/j.clml.2013.11.002. Epub 2013 Nov
14. https://doi.org/10.1016/j.clml.2013.11.002
PMid:24456839
- Hochhaus
A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020
recommendations for treating chronic myeloid leukemia. Leukemia.
2020;34(4):966-984. https://doi.org/10.1038/s41375-020-0776-2
PMid:32127639 PMCid:PMC7214240
- Sokal
JE, Cox EB, Baccarani M, Tura S, Gomez GA, Robertson JE, et al.
Prognostic discrimination in "good-risk" chronic granulocytic leukemia.
Blood. 1984;63:789-99. https://doi.org/10.1182/blood.V63.4.789.789
PMid:6584184
- Hasford
J, Pfirrmann M, Hehlmann R, Allan NC, Baccarani M, Kluin-Nelemans JC,
et al. A new prognostic score for survival of patients with chronic
myeloid leukemia treated with interferon alfa. Writing Committee for
the Collaborative CML Prognostic Factors Project Group. J Natl Cancer
Inst. 1998;90:850-8. https://doi.org/10.1093/jnci/90.11.850
PMid:9625174
- Hasford
J, Baccarani M, Hoffmann V, Guilhot J, Saussele S, Rosti G, et al.
Predicting complete cytogenetic response and subsequent
progression-free survival in 2060 patients with CML on imatinib
treatment: the EUTOS score. Blood. 2011;118:686-92. https://doi.org/10.1182/blood-2010-12-319038
PMid:21536864
- Zhang,
XS., Gale, R.P., Zhang, MJ.et al.A predictive scoring system for
therapy-failure in persons with chronic myeloid leukemia receiving
initial imatinib therapy. Leukemia 36, 1336-1342 (2022). https://doi.org/10.1038/s41375-022-01527-y
PMid:35194158
- Ielo,
C., Scalzulli, E., Carmosino, I., et al. (2023). Validation of imatinib
therapy failure score (IMTF) in chronic phase chronic myeloid leukemia
in real-life practice. Leukemia & Lymphoma,64(14), 2324-2326. https://doi.org/10.1080/10428194.2023.2255804
PMid:37689986
- Zhang
X, Liu B, Huang J, et al. A predictive model for therapy failure in
patients with chronic myeloid leukemia receiving tyrosine kinase
inhibitor therapy. Blood. 2024;144(18):1951-1961. https://doi.org/10.1182/blood.2024024761
PMid:39046786 PMCid:PMC11551847
- Khoury
JD,Solary E,Abla O, et al.The 5th edition of the World Health
Organization classification of haematolymphoid tumours: myeloid and
histiocytic/dendritic neoplasms. Leukemia 2022;36(7):1703-1719. https://doi.org/10.1038/s41375-022-01613-1
PMid:35732831 PMCid:PMC9252913
- Hughes
T, Deininger M, Hochhaus A,et al Monitoring CML patients responding to
treatment with tyrosine kinase inhibitors: review and recommendations
for harmonizing current methodology for detecting BCR-ABL transcripts
and kinase domain mutations and for expressing results. Blood.
2006;108:28-37. https://doi.org/10.1182/blood-2006-01-0092
PMid:16522812 PMCid:PMC1895821
- Hochhaus
A. A further milestone towards comprehensive standardization of
quantitative RT-PCR protocols for leukemic fusion gene transcripts has
been reached. Leukemia. 2003;17:2383. https://doi.org/10.1038/sj.leu.2403137
PMid:14562123