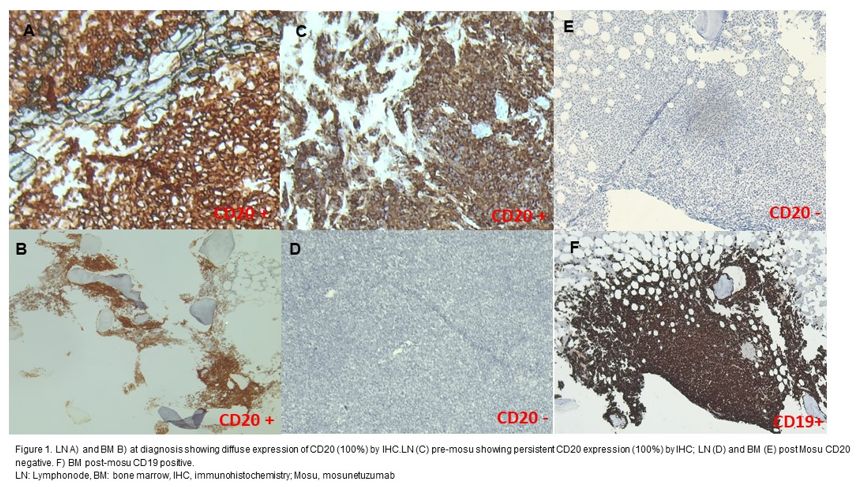
A 49-year-old man was diagnosed in 2019 with FL involving the mediastinum, abdomen, and bone marrow (Figure 1A-1B). First-line R-CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) induced a Lugano-defined CR. He relapsed in 2021; biopsy confirmed FL, and treatment with obinutuzumab–bendamustine followed by autologous stem cell transplantation again achieved CR. His post-transplant course was complicated by SARS-CoV-2 pneumonia, invasive aspergillosis, and cytomegalovirus (CMV) reactivation—an infectious trifecta that would make even the hardiest immune system consider a career change.
In January 2023, PET-CT revealed mediastinal progression, managed with 40 Gy radiotherapy. In March 2024, new abdominal disease appeared (SUV 12.5). Biopsy showed relapse of FL (Figure 1C) At this time the patient initiated mosu (Nov 2024–Apr 2025). A CT scan in May 2025 -after 6 cycles- showed progression, and lymph node biopsy demonstrated FL with complete CD20 loss (Figure 1D, 1E). The patient was evaluated for CAR-T, received bridging R-GemOx (gemcitabine 1000 mg/m² and oxaliplatin 100 mg/m²), and underwent axicabtagene ciloleucel infusion in July 2025. Grade 2 cytokine release syndrome (CRS) resolved with tocilizumab and dexamethasone, while hemophagocytic lymphohistiocytosis (HLH) syndrome on day +23 from CAR-T infusion improved with anakinra. He currently remains clinically well, with a 3-month post-infusion PET-CT showing the reduction of the previous lymphoma mass.
Recent investigations have shed light on the biology of CD20 loss in the context of CD20-directed bispecific antibodies. Schuster et al.[3] reported that although CD20 loss is not common, it correlates with disease progression in a subset of GO29781 participants, with approximately one-third of evaluable biopsies demonstrating marked downregulation — particularly among patients with low baseline antigen density. However, most patients progressing on mosu retain CD20, suggesting additional resistance pathways, including MS4A1 transcriptional suppression, CD20 structural alterations, and microenvironment-driven immune dysfunction. Olszewski et al.[4] have proposed three temporal patterns of failure under bispecific therapy: (1) early progression, shaped by an immunosuppressive tumor microenvironment and insufficient T-cell infiltration; (2) intermediate progression, driven by MS4A1 mutations or silencing leading to reduced CD20 expression; and (3) late progression, associated with T-cell exhaustion. Our clinical case clearly fits the intermediate pattern, in which antigen loss results in complete abrogation of the therapeutic target. In our patient, CD20 expression — consistently assessed using the same immunohistochemical protocol and antibody clone — was 100% at diagnosis, while a complete loss of CD20 expression was documented during treatment. This finding plausibly places our case within the second hypothesis proposed by Olszewski et al.[4] This case underscores the need for systematic reassessment of antigen expression at each relapse, especially when progression occurs on CD20-directed therapies. In patients with diminished or absent CD20 expression, alternative therapeutic routes — such as CD19-directed bispecific antibodies, CAR-T cell products,[5] or investigational strategies aimed at restoring antigen expression or augmenting T-cell function — may offer greater benefit. For this reason, assessing CD19 expression was decisive in determining eligibility for CAR-T therapy.[6-8] In conclusion, while mosu has meaningfully expanded the treatment landscape for R/R FL, emerging resistance mechanisms — particularly CD20 loss — highlight the importance of dynamic antigen reassessment. Incorporating routine evaluation at diagnosis and at each relapse may facilitate more refined therapeutic sequencing and decision-making to improve patient outcomes.
Author contributions
MC conceived the manuscript and drafted the text. SF contributed to the histopathological section and provided the corresponding images. ADR assisted with the literature review and bibliography. EA critically reviewed the clinical case and provided final revisions.References
- Bartlett NL, Assouline S, Giri P, Schuster SJ,
Cheah CY, Matasar M, Gregory GP, Yoon DH, Shadman M, Fay K, Yoon SS,
Panizo C, Flinn I, Johnston A, Bosch F, Sehn LH, Wei MC, Yin S, To I,
Li CC, Huang H, Kwan A, Penuel E, Budde LE. Mosunetuzumab monotherapy
is active and tolerable in patients with relapsed/refractory diffuse
large B-cell lymphoma. Blood Adv. 2023;7(17):4926–4935. https://doi.org/10.1182/bloodadvances.2022009260
- Budde
LE, Assouline S, Sehn LH, Schuster SJ, Yoon SS, Yoon DH, Matasar MJ,
Bosch F, Kim WS, Nastoupil LJ, Flinn IW, Shadman M, Diefenbach C,
O'Hear C, Huang H, Kwan A, Li CC, Piccione EC, Wei MC, Yin S, Bartlett
NL. Single-agent mosunetuzumab shows durable complete responses in
patients with relapsed or refractory B-cell lymphomas: Phase I
dose-escalation study. J Clin Oncol. 2022;40(5):481–491 https://doi.org/10.1200/JCO.21.00931
- Schuster
SJ, Huw LY, Bolen CR, et al. Loss of CD20 expression as a mechanism of
resistance to mosunetuzumab in relapsed/refractory B-cell lymphomas.
Blood. 2024;143(9):822-832. https://doi.org/10.1182/blood.2023022348 PMid:38048694 PMCid:PMC10934296
- Olszewski
AJ. When B cells rebuff bispecifics. Blood. 2024 Feb 29;143(9):744-746.
https://doi.org/10.1182/blood.2023023312 PMID: 38421818.
- Testa
U., D'Alò F., Pelosi E., Castelli G., Leone G. CAR-T cell therapy for
follicular lymphomas. Mediterr J Hematol Infect Dis 2024, 16(1):
e2024012 https://doi.org/10.4084/MJHID.2024.012 PMid:38223488 PMCid:PMC10786124
- Plaks V, Rossi JM, Chou J, et al. CD19 target evasion after axi-cel. Blood. 2021;138(12):1081-1085. https://doi.org/10.1182/blood.2021010930 PMid:34041526 PMCid:PMC8462361
- Sotillo
E, Barrett DM, Black KL, et al. CD19 mutations and alternative splicing
enable CAR-T resistance. Cancer Discov. 2015;5(12):1282- 1295. https://doi.org/10.1158/2159-8290.CD-15-1020 PMid:26516065 PMCid:PMC4670800
- Hernani
R, Ventura L, Heras B, et Al. Clinical Impact of CD19 Expression
Assessed by Quantitative PCR in Lymphoma Patients Undergoing CAR-T
Therapy. E J Haem. 2025 ;6(2):e270015. https://doi.org/10.1002/jha2.70015 PMid:40110072 PMCid:PMC11920814