Introduction
In recent years, many breakthroughs have characterized the clinical assessment of subjects with FWS; however, the final diagnosis in many cases may remain unknown. The MeMed BV assay (an automated host-immune chemiluminescence test) has been developed to differentiate bacterial and viral infections by measuring and computationally integrating 3 immune parameters in serum: C-reactive protein (CRP), tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), and interferon gamma-induced protein 10 (IP-10).[7]We conducted this pilot study using the MeMed test to provide further information on FWS diagnosis, extrapolating results for CRP, TRAIL, and IP-10 from the MeMed BV assay. The rationale for this study was to determine whether each host marker could be useful for confirming pathophysiological processes related to the final diagnosis of FWS.
The study included 22 subjects with FWS who were hospitalized at the Policlinico A. Gemelli IRCCS, Rome, Italy, during the period 2019-2023; their FWS was defined according to current guidelines or active medical literature definitions.[8] Specifically, they had a median age/IQR of 52.0/26.7 years (the M/F ratio was 17/5).
Their sera were processed through the MeMed BV on a Liaison XL platform (DiaSorin, Saluggia, Italy), as previously reported.[9] The MeMed BV cartridges are single‐use multiwell containers that work with 100 µL of subjects’ serum sample, contain all reagents with internal controls and disposables necessary to conduct the immunoassay. Upon insertion of the test cartridge into the analyzer, 3 independent assessments can be simultaneously performed to measure the cited biomarkers (CRP, TRAIL, and IP‐10). The test also provides a likelihood score (the “BV score”), ranging from 0 to 100: viral infections are associated with a MeMed BV score ≤35, while bacterial infections are associated with a MeMed BV score >65-100. We individually considered serum levels of the subanalytes CRP, TRAIL, and IP-10 rather than the BV scores. The considered cut-offs were 1,9 mg/l for CRP, 74,5 pg/ml for TRAIL, and 113,6 pg/ml for IP-10.[10]
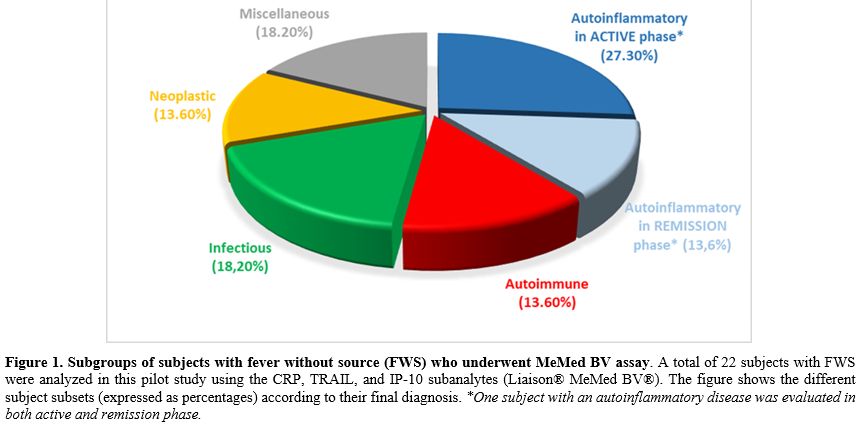
Subjects recruited in this retrospective investigation were stratified according to the final definitive diagnosis into 5 different categories: (a) autoinflammatory diseases (36.4%), (b) autoimmune diseases (13.6%), (c) infectious diseases (18.2%), (d) neoplastic diseases (13.6%), and (e) miscellaneous disorders (18.2%, see Figure 1); these diagnoses were established according to the routinely used serologic tests, autoantibody panels, histopathological results, imaging studies, and finally by the reported success of applied therapies. Frozen serum samples collected at the time of hospitalization were analyzed for CRP, TRAIL, and IP-10, and these data were compared with subjects’ diagnoses at the time of this study.
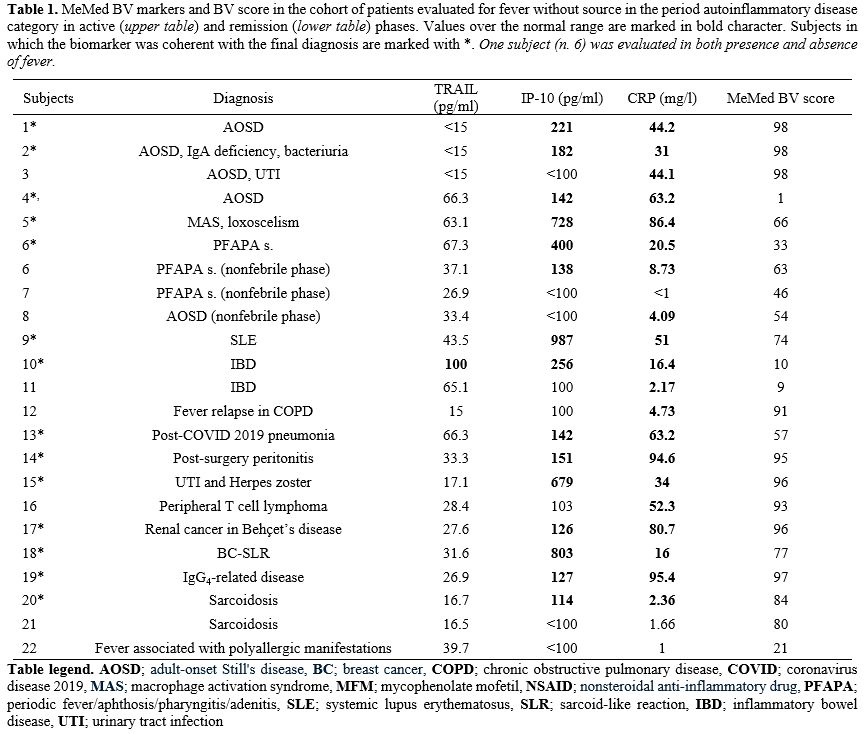
Among 8 subjects with autoinflammatory diseases (36.4% of the cohort), 5 had adult-onset Still’s disease (AOSD), 1 macrophage activation syndrome (MAS) after a spider bite of the genus Loxosceles, and 2 periodic fever/aphthosis/pharyngitis/adenitis (PFAPA) syndrome. One PFAPA patient was tested during both active and non-active disease phases. In these patients, both CRP and IP-10 were elevated during the active disease phase but low during the inactive phase; it was also possible to demonstrate that IP-10 elevation reflected interferon pathway activation in the subject with MAS. Three subjects had autoimmune disease: one with systemic lupus erythematosus (SLE) and 2 other ones with inflammatory bowel disease; these subjects showed consistently increased IP-10, variably increased CRP, and consistently low TRAIL. Infectious diseases were diagnosed in 4 subjects (18.2% of the cohort): fever relapse associated with chronic obstructive pulmonary disease (COPD), post-coronavirus disease 2019 (COVID-19) pneumonia, post-surgery peritonitis, and urinary tract infection (UTI) combined with Herpes zoster infection. In these cases, CRP values were high, TRAIL values were low or normal, and IP-10 values were almost normal or slightly higher than normal, except in the subject with UTI and Herpes zoster infection. Three/22 subjects (13.6% of the cohort) had neoplastic diseases: peripheral T cell lymphoma (a highly pyrogenic non-Hodgkin lymphoma), papillary renal cell cancer in combination with Behçet’s disease, and breast cancer (BC)-associated sarcoid-like reaction (SLR). CRP levels were high in the subject with lymphoma, revealing a chronic state of inflammation, whereas IP-10 was high in the other two. Four patients over a total of 22 were included in the miscellaneous category (18.2% of the cohort) with the following diagnoses: IgG4-related disease in 1, sarcoidosis in 2, and recurrent fevers related to severe polyallergy in the last 1; this group revealed the highest IP-10 result, which was also associated with the highest prednisone dose needed.
Discussion
The use of inflammatory markers may help in the differential diagnosis of FWS in combination with other first-level tests. In particular, CRP has been used as a simple marker of inflammation, common to both infectious and non-infectious inflammatory diseases. The biological role of CRP is linked to the host’s defense as part of the innate immune system, as it is a highly conserved plasma protein produced in response to various inflammatory triggers.[11] Indeed, high CRP levels in SLE might suggest an underlying infection,[12] but they might also disclose systemic innate immunity-mediated inflammation as in Kawasaki disease, parallel to the risk of non-responsiveness to i.v. immunoglobulin levels and risk of developing coronary artery abnormalities.[13,14] TRAIL is another marker of the TNF superfamily, capable of activating a pro-apoptotic pathway, suggesting the potential for more targeted therapies in patients with cancer.[15] Beyond oncology, TRAIL also exhibits a pro-apoptotic effect on immune cells, contributing to the regulation of many immunologic processes and anti-inflammatory activities.[16] Although TRAIL enhances neutrophil apoptosis and reduces inflammation, it can promote cell survival by inducing polarization of human macrophages toward a pro-inflammatory M1 phenotype via DR4 and DR5 death receptors.[17] A further marker acting as a ligand of chemokine (C-X-C motif) receptor 3 (CXCR3) is IP-10 (also known as CXCL10), which usually recruits immune cells to the site of inflammation.[18] Higher levels of IP-10 in the peripheral fluids can testify a T helper 1-oriented immune response, as occurring in different infections, but IP-10 and its receptor are also involved in the pathogenesis of many autoimmune diseases, either organ-specific (such as type 1 diabetes and Graves’ disease) or systemic (SLE, mixed cryoglobulinemia, Sjögren’s syndrome, and systemic sclerosis).[19,20]In this pilot study all categories of FWS of the most relevant studies have been included: autoinflammatory diseases (8 subjects, with 1 diagnosed with PFAPA syndrome considered twice as undergoing assessment during fever and not), autoimmune diseases (3 subjects), infectious diseases (4 subjects), neoplastic diseases (3 subjects), and a miscellaneous group of disorders (4 subjects). The 3 immune biomarkers in the MeMed BV test have been used to characterize the inflammatory profile of subjects with FWS and to improve the diagnostic process toward the final diagnosis that emerged during patients’ hospitalizations.
Indeed, the results of MeMed BV subanalytes in 14/22 subjects (63.6% of the whole cohort) were consistent with the final diagnosis of FWS, suggesting that routine use of this test could help address the challenging process of identifying the cause of FWS in real-life clinical practice. More specifically, in autoinflammatory diseases, CRP and IP-10 were elevated during the active phase, with IP-10 levels suggesting a marked interferon-based activation in the subject with MAS. In patients with autoimmune diseases, we observed a consistent increase in IP-10, while CRP gave variable results, and TRAIL was consistently low. The subgroup with infectious diseases had reduced TRAIL levels, despite elevated CRP and variable IP-10 levels, which were significantly increased in the case of Herpes zoster infection, as expected for a virus-induced interferon-mediated response. Two patients with a final diagnosis of malignancy had consistently elevated levels of CRP and IP-10. In the miscellaneous disease subgroup, 2/4 subjects had higher IP-10 levels, which may also correlate with the higher corticosteroid doses these patients required.
Limitations
The low number of FWS patients recruited substantially limits the significance of these preliminary results, referred to as our pilot study, reducing its statistical power, hindering generalizability, and increasing the risk of misinterpreting the true diagnostic contribution of CRP, TRAIL, and IP-10 in FWS.Conclusions
This pilot study highlights that the MeMed BV technology provides data potentially useful for describing the inflammation pattern underlying the pathology in subjects with FWS, through the individual evaluation of CRP, TRAIL, and IP-10, revealing correlations with clinical histories and mechanisms of specific diseases. These results support larger randomized studies with similar case-subgroup distributions to better understand and define the potential discriminative role of these markers, with the aim of disclosing the final diagnosis of FWS as quickly as possible.Author Contributions
All authors gave substantial contributions to the conception and design of the review, the acquisition, analysis, and interpretation of the literature, as well as to the drafting and critical revision of the manuscript. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work, ensuring its accuracy and integrity.List of Abbreviations
AOSD: Adult-onset Still's diseaseCXCL10 or IP-10: Interferon-γ-inducible protein 10
COVID: Coronavirus disease
COPD: Chronic obstructive pulmonary disease
DR: Death receptor
FWS: Fever without source
MAS: Macrophage activation syndrome
MFM: Mycophenolate mofetil
NSAID: Nonsteroidal anti-inflammatory drug
PFAPA: Periodic fever/aphthosis/pharyngitis/adenitis
SLE: Systemic lupus erythematosus
SLR: Sarcoid-like reaction
TNF: Tumor necrosis factor
TRAIL: Tumor necrosis factor-related apoptosis-inducing ligand
UTI: Urinary tract infection
References
- Antoon JW, Potisek NM, Lohr JA. Pediatric fever of unknown origin. Pediatr Rev 2015;36:380-90. https://doi.org/10.1542/pir.36.9.380 PMid:26330472
- Petersdorf RG, Beeson PB. Fever of unexplained origin: report on 100 cases. Medicine (Baltimore) 1961;40:1-30. https://doi.org/10.1097/00005792-196102000-00001 PMid:13734791
- Cunha BA, Lortholary O, Cunha CB. Fever of unknown origin: a clinical approach. Am J Med 2015;128:1138.e1-e15. https://doi.org/10.1016/j.amjmed.2015.06.001 PMid:26093175
- Jiang
L, Wu H, Zhao S, et al. Structured diagnostic scheme clinical
experience sharing: a prospective study of 320 cases of fever of
unknown origin in a tertiary hospital in North China. BMC Infect Dis
2023;23:452. https://doi.org/10.1186/s12879-023-08436-0 PMid:37420165 PMCid:PMC10327337
- Rigante
D. Autoinflammatory syndromes behind the scenes of recurrent fevers in
children. Med Sci Monit 2009;15:RA179-87. PMid: 19644432
- Cantarini
L, Iacoponi F, Lucherini OM, et al. Validation of a diagnostic score
for the diagnosis of autoinflammatory diseases in adults. Int J
Immunopathol Pharmacol 2011;24:695-702. https://doi.org/10.1177/039463201102400315 PMid: 21978701
- Papan
C, Argentiero A, Porwoll M, et al. A host signature based on TRAIL,
IP-10, and CRP for reducing antibiotic overuse in children by
differentiating bacterial from viral infections: a prospective
multicentre cohort study. Clin Microbiol Infect 2022;28:723-30. https://doi.org/10.1016/j.cmi.2021.10.019 PMid:34768022
- Durack DT, Street AC. Fever of unknown origin-reexamined and redefined. Curr Clin Top Infect Dis 1991;11:1651090.PMid: 1651090
- Hainrichson
M, Avni N, Eden E, et al. A point-of-need platform for rapid
measurement of a host-protein score that differentiates bacterial from
viral infection: analytical evaluation. Clin Biochem 2022;117:39-47. https://doi.org/10.1016/j.clinbiochem.2022.04.012 PMid:35487256
- Langedijk
AC, Rengerink KO, Harding E, et al. Natural variability of TRAIL,
IP-10, and CRP in healthy adults - The "HERACLES" study. Cytokine
2024;176:156530. https://doi.org/10.1016/j.cyto.2024.156530 PMid:38306791
- Naito
T, Torikai K, Mizooka M, et al. Relationships between causes of fever
of unknown origin and inflammatory markers: a multicenter collaborative
retrospective study. Intern Med 2015;54:1989-94. https://doi.org/10.2169/internalmedicine.54.3313 PMid:26278289
- Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front Immunol 2018;9:754. https://doi.org/10.3389/fimmu.2018.00754 PMid:29706967 PMCid:PMC5908901
- De
Rosa G, Pardeo M, Rigante D. Current recommendations for the
pharmacologic therapy in Kawasaki syndrome and management of its
cardiovascular complications. Eur Rev Med Pharmacol Sci 2007;11:301-8.
PMid: 18074939
- Rigante
D, Valentini P, Rizzo D, et al. Responsiveness to intravenous
immunoglobulins and occurrence of coronary artery abnormalities in a
single-center cohort of Italian patients with Kawasaki syndrome.
Rheumatol Int 2010;30:841-6. https://doi.org/10.1007/s00296-009-1337-1 PMid:20049445
- Lim
B, Allen JE, Prabhu VV, et al. Targeting TRAIL in the treatment of
cancer: new developments. Expert Opin Ther Targets 2015;19:1171-85. https://doi.org/10.1517/14728222.2015.1049838 PMid:26004811
- McGrath
EE, Marriott HM, Lawrie A, et al. TNF-related apoptosis-inducing ligand
(TRAIL) regulates inflammatory neutrophil apoptosis and enhances
resolution of inflammation. J Leukoc Biol 2011;90:855-65. https://doi.org/10.1189/jlb.0211062 PMid:21562052 PMCid:PMC3644175
- Gunalp
S, Helvaci DG, Oner A, et al. TRAIL promotes the polarization of human
macrophages toward a proinflammatory M1 phenotype and is associated
with increased survival in cancer patients with high tumor macrophage
content. Front Immunol 2023;14:1209249. https://doi.org/10.3389/fimmu.2023.1209249 PMid:37809073 PMCid:PMC10551148
- Lacotte S, Brun S, Muller S, et al. CXCR3, inflammation, and autoimmune diseases. Ann NY Acad Sci 2009;1173:310-7. https://doi.org/10.1111/j.1749-6632.2009.04813.x PMid:19758167
- Jain
V, Armah HB, Tongren JE, et al. Plasma IP-10, apoptotic and angiogenic
factors associated with fatal cerebral malaria in India. Malar J
2008;7:83. https://doi.org/10.1186/1475-2875-7-83 PMid:18489763 PMCid:PMC2405803
- Rubinstein A, Kudryavtsev I, Arsentieva N, et al. CXCR3-expressing T cells in infections and autoimmunity. Front Biosci (Landmark Ed) 2024;29:301. https://doi.org/10.31083/j.fbl2908301 PMid:39206903