Dynamic Hb monitoring is increasingly recognized as a crucial factor in capturing disease activity, treatment response, and prognostic risk. This approach is supported by evidence from various fields where latent class trajectory modeling has been applied to identify distinct patterns of disease progression and associated risks. For instance, in type 2 diabetes, different HbA1c trajectories have been linked to varying risks of complications [6]. Despite its potential, this modeling approach is underutilized in the management of CKD anemia. Frailty, a parallel and understudied outcome in CKD, shares biological pathways with anemia and is associated with adverse outcomes like falls and hospitalizations.[7,8] The integration of dynamic assessments of anemia and frailty is crucial, as both contribute to cardiovascular risk, with anemia-related hemodynamic stress and frailty acting as independent cardiovascular risk enhancers.[9] The need for an integrated assessment of anemia dynamics, frailty changes, and cardiovascular events is underscored by the potential to improve prognostic accuracy and patient stratification, as demonstrated in other diseases through dynamic modeling.[10,11] These approaches, such as dynamic phenotype modeling and trajectory alignment, have shown promise for enhancing understanding of disease progression and improving clinical outcomes by capturing the temporal dynamics of disease states.[12] Therefore, adopting similar methodologies in CKD could provide significant clinical benefits, offering a more comprehensive and personalized approach to patient care.
The existing literature highlights a significant knowledge gap regarding the simultaneous examination of longitudinal Hb trajectories, frailty progression, and incident cardiovascular events in patients with stage 3–5 non-dialysis-dependent CKD (ND-CKD). Most studies have primarily focused on single baseline Hb measurements, neglecting the dynamic nature of Hb levels over time and the impact of frailty on clinical outcomes.[13-15] For instance, the CKD-REIN cohort study identified five distinct Hb trajectory profiles, revealing that while two-thirds of patients maintained stable Hb levels, the remaining third exhibited declining trajectories associated with increased risks of major adverse cardiovascular events.[15] Furthermore, frailty, prevalent in advanced CKD, has been linked to adverse outcomes, yet longitudinal studies exploring its progression in this population remain scarce.[16] This underscores the need for comprehensive research that integrates these factors to better understand their interrelationships and implications for patient management in CKD.[17]
The present study sought to delineate 12 month Hb trajectories in adults with stage 3-5 ND-CKD and to evaluate whether these longitudinal patterns are associated with two clinically salient outcomes: frailty worsening and a composite of major cardiovascular events. We hypothesized that a persistently declining Hb pattern, relative to a stable pattern, would be associated with greater frailty worsening and higher risk of cardiovascular events.
Methods
Study design and setting. We conducted a prospective, single center cohort study at Affiliated Hospital of Hebei University from June 2023 to December 2023. Each participant was followed for 12 months or until death, kidney replacement therapy initiation, or withdrawal of consent, whichever occurred first. The local Institutional Review Board approved the study, and all participants gave written informed consent.Participants. Adults ≥18 years were eligible if they met all of the following at screening: (1) estimated glomerular filtration rate (eGFR, CKD EPI 2021)<60 mL/min/1.73 m², classifying as CKD stage3a–5 and not receiving dialysis or having a functioning kidney transplant; (2) renal anemia defined by Hb<13 g/dL for men or <12 g/dL for women on two occasions ≥7 days apart; and (3) ability to attend monthly study visits. Exclusion criteria were: active malignancy, pregnancy or lactation, acute bleeding episode in the preceding 3 months, planned dialysis or transplantation within 3 months, acute kidney injury, or participation in another interventional trial likely to affect Hb concentration.
Data collection and variable definitions. At baseline, we recorded demographics, CKD stage, comorbidities (Charlson index), medication use (including erythropoiesis stimulating agents [ESAs], hypoxia inducible factor prolyl hydroxylase inhibitors [HIF PHIs], ACE inhibitors/ARBs, and SGLT2 inhibitors), blood pressure, body mass index (BMI), and laboratory tests (Hb, ferritin, transferrin saturation [TSAT], C reactive protein [CRP], serum albumin, serum creatinine). Hb was measured monthly using a Sysmex XN 1000 hematology analyzer. All other blood tests were repeated every 3 months or when clinically indicated. Medication exposure variables used in adjusted models reflected baseline ESA/HIF-PHI use. Protocolized time-updated dose-escalation data were unavailable, so whether treatment intensification preceded or followed the Hb decline could not be reliably determined.
Frailty was assessed at baseline, 6 months and 12 months using: (i) the 5 item FRAIL scale (Fatigue, Resistance, Ambulation, Illnesses, Loss of weight; score 0–5) and (ii) objective performance measures: hand grip strength (Jamar dynamometer, average of three tests; <26 kg men or <18 kg women considered weak) and 4 metre gait speed (slow <0.8 m/s). Frailty worsening was predefined as a ≥1 point increase in the FRAIL score or new onset of either weak grip or slow gait. Because frailty was reassessed only at these prespecified visits, short-term fluctuations between visits were not captured.
Cardiovascular events comprised a prespecified composite of (a) non fatal myocardial infarction, (b) non fatal stroke or transient ischemic attack, (c) hospitalization for new onset or acutely decompensated heart failure, (d) sustained ventricular arrhythmia or atrial fibrillation/flutter requiring hospital care, and (e) cardiovascular death. Two blinded cardiologists adjudicated all suspected events, with disagreements resolved by a third reviewer.
Sample size justification. Using Schoenfeld’s formula for time to event analyses, we assumed three trajectory classes with 40% of patients in the “Declining” class, a 12 month composite CV event rate of 25%, and a clinically meaningful hazard ratio (HR) of 2.5 versus the “Stable” class. With α = 0.05 (two sided) and 80% power, 39 events were required, translating to 156 patients; inflating by 15% for attrition yielded a target enrolment of 190. This sample provided>80% power to detect a 30 percentage point difference in frailty worsening risk (30% vs 60%).
Statistical analysis. Continuous variables are reported as mean ± standard deviation (SD) or median (interquartile range, IQR), and categorical variables are reported as counts (%). Baseline differences across trajectory classes were evaluated with ANOVA or Kruskal–Wallis tests (continuous) and χ2 tests (categorical).
Hemoglobin trajectories were derived using latent-class mixed models based on all available monthly Hb measurements during follow-up. Competing 2- to 5-class models were compared using BIC, AIC, entropy, mean posterior class-membership probabilities, minimum class size, and clinical interpretability. Because class membership is estimated from repeated Hb measurements collected during follow-up, the trajectory classes are interpreted as summaries of longitudinal Hb patterns observed during the study period.
Frailty worsening was evaluated at 6 months and 12 months relative to baseline, yielding up to two repeated binary outcomes per participant. Mixed-effects logistic regression was used with a random intercept for participant and fixed effects for trajectory class and prespecified covariates: age, sex, baseline eGFR, diabetes status, CRP, ferritin, TSAT, baseline ESA/HIF-PHI exposure, and ACE inhibitor/ARB use.
The time to first composite CV event was examined using cause-specific Cox proportional-hazards models. Non-CV death and kidney replacement therapy (KRT) initiation were treated as censoring events in cause-specific analyses. Proportional-hazards assumptions were checked with Schoenfeld residuals.
Sensitivity analyses included: (1) a joint longitudinal–time-to-event model linking longitudinal Hb and time to CV event; (2) Fine–Gray subdistribution hazards treating non-CV death or KRT initiation as competing events; (3) multiple imputation by chained equations (20 datasets) for missing covariates; (4) exclusion of participants with baseline ferritin >800 ng/mL; and (5) stratified analyses by CKD stage (3 vs 4–5).
Exploratory analyses examined whether frailty worsening might lie on the pathway between Hb trajectory and CV events; given the discrete timing of frailty assessments and the possibility that CV events could occur before frailty reassessment, these analyses were considered hypothesis-generating and are presented in the Supplement. All tests were two sided with significance set at p < 0.05. Effect estimates are reported as odds ratios (OR) or hazard ratios (HR) with 95% confidence intervals (CI). Analyses were performed in Rv4.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
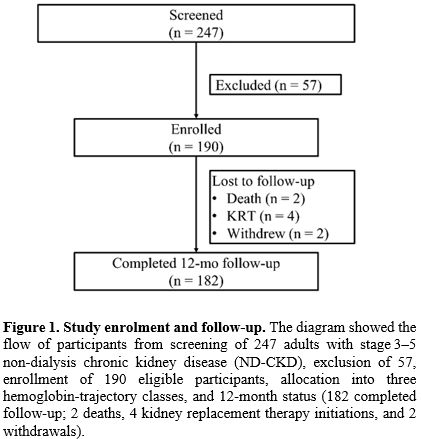
Study population. We screened 247 adults with stage 3–5 ND-CKD, enrolled 190 who met eligibility criteria, and had 12-month follow-up assessments available for 182 (95.8%) (Figure 1). Two participants died, four initiated kidney replacement therapy, and two withdrew consent before the 12-month visit. The cohort’s mean ± SD age was 63 ± 12 years, 44% were women, and 58% had CKD stage 4–5. Baseline characteristics were broadly similar across hemoglobin trajectory classes, although baseline use of erythropoiesis stimulating agents or HIF prolyl hydroxylase inhibitors was higher in the Declining group (59%) than in the Stable (43%) or Fluctuating (53%) groups (Table 1).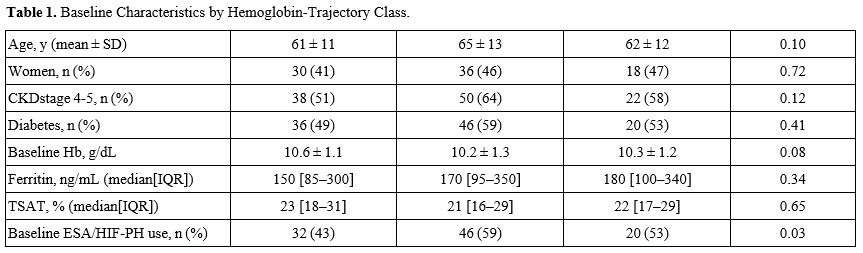 |
Table 1. Baseline Characteristics by Hemoglobin Trajectory Class.Suggeste |
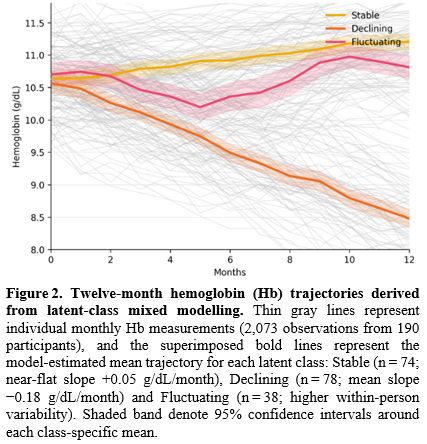
A three class latent class linear mixed effects model best described the 2,073 hemoglobin observations (mean 10.9 Hb measurements per participant), yielding a Stable class of 74 participants (39%) with an almost flat monthly slope (+0.05 g/dL), a Declining class of 78 participants (41%) exhibiting a mean decrease of −0.18 g/dL per month, and a Fluctuating class of 38 participants (20%) whose linear course had a within person coefficient of variation exceeding 12%. Model fit statistics were optimal at a Bayesian Information Criterion of 4,015 and entropy of 0.83, and 86% of individuals had posterior class membership probabilities ≥0.90 (Table 2 and Figure 2). The four-class model had a higher BIC (4,028), lower entropy (0.79), and less clinically interpretable class sizes, supporting retention of the three-class solution.
During follow up, frailty worsened in 25% of Stable, 57% of Declining, and 45% of Fluctuating participants (p < 0.001). In a mixed effects logistic regression adjusted for demographic, renal, inflammatory and treatment covariates, the Declining trajectory was associated with nearly threefold higher odds of frailty deterioration compared with the Stable trajectory (adjusted odds ratio [aOR] 2.8, 95% CI 1.6–5.0), whereas the Fluctuating trajectory showed an elevated but non significant odds ratio of 1.9 (95% CI 0.9–4.0) (Table 3).
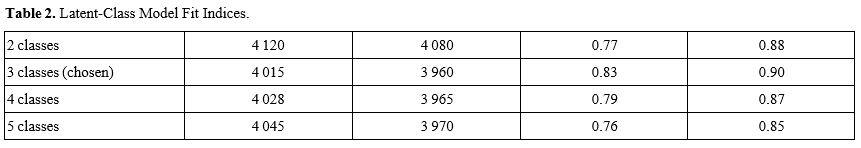 |
Table 2. Latent Class Model Fit Indices. |
 |
Table 3. Association Between Hb Trajectory and Frailty Worsening. |
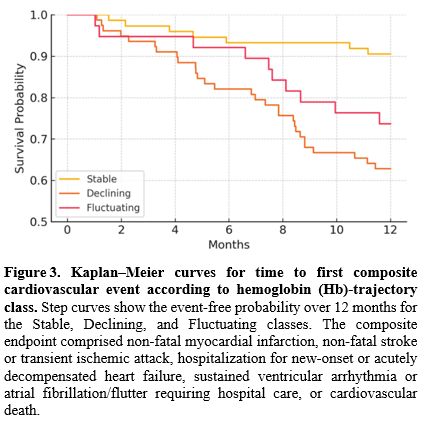
Over 185 person years (median follow up 364 days), 46 composite cardiovascular events occurred — 29 in the Declining, 10 in the Fluctuating, and 7 in the Stable class — yielding an incidence of 24.9 per 100 person years and clearly separated Kaplan–Meier estimates of time-to-first composite CV events (Figure 3). After multivariable adjustment, the Declining trajectory conferred a 2.6 fold higher hazard of cardiovascular events relative to the Stable trajectory (adjusted hazard ratio [aHR] 2.6, 95% CI 1.4–4.9; p = 0.003), whereas the Fluctuating trajectory carried a non significant 70 % increase (aHR 1.7, 95% CI 0.8–3.6; p = 0.17) (Table 4); proportional hazards assumptions were satisfied.
 |
Table 4. Association Between Hb Trajectory and Composite Cardiovascular Events. |
Discussion
This prospective cohort study examined whether distinct 12-month Hb trajectory patterns are associated with frailty worsening and cardiovascular outcomes in adults with stage 3–5 ND-CKD and renal anemia. Using latent-class mixed modeling, we identified three patterns — Stable, Declining, and Fluctuating. The Declining group was independently associated with substantially higher odds of frailty worsening and more than doubled hazard of composite CV events compared with the Stable class, whereas the Fluctuating class showed intermediate, non-significant associations. These longitudinal Hb trajectories should be interpreted as prognostic pattern markers rather than proven mediators of frailty progression or cardiovascular injury.Recent work in oncology and cardiology has already illustrated that the direction and velocity of hemoglobin change can matter more than its absolute value at a single visit: in non small cell lung cancer and colorectal cancer a post diagnosis shift of |∆Hb| > 2.6 g/dL heralds poorer survival,[18] while in heart failure with reduced ejection fraction declining Hb portends higher rehospitalization and death.[19] In nephrology, however, most observational or interventional studies, including the landmark CREATE, TREAT, and PIVOTAL trials, categorized anemia exposure by a baseline or target Hb threshold and therefore could not capture within patient trends.[20-22] Our identification of three 12 month Hb trajectories (Stable, Declining, Fluctuating) suggests that a persistently declining profile was associated with a substantially higher probability of frailty worsening and an increased risk of cardiovascular events compared with a stable trajectory, consistent with recent CKD trajectory analyses.[15] Notably, the 57% frailty worsening rate observed in the Declining group suggests that progressive anemia may identify a particularly vulnerable subgroup within ND-CKD.
The association between a declining Hb pattern and CV events is likely multifactorial. The observed pattern may reflect a combination of worsening underlying disease, inflammation, iron-restricted erythropoiesis, and treatment hyporesponsiveness rather than a direct causal effect of Hb decline itself. Anemia amplifies myocardial workload, while activation of hypoxia inducible factors drives both renal fibrosis and cardiomyocyte hypertrophy.[23-25] Concurrent inflammation and iron dysregulation accelerate muscle catabolism and sarcopenia, which may help explain why declining Hb co-occurred with frailty worsening.[24,26] Because baseline ESA/HIF-PHI use was more frequent in the Declining group and time-updated dose-escalation data were not modeled, confounding by indication remains possible. The continued Hb fall despite greater treatment exposure is also compatible with ESA/HIF-PHI hyporesponsiveness, suboptimal titration, or treatment resistance.[27] By contrast, the Fluctuating group showed no significant excess CV risk, a pattern that may reflect reversible Hb dips during intercurrent illness or preserved physiological reserve.[28] This heterogeneity highlights the need to interpret episodic anemia in its clinical context.
Besides hemodynamic overload, oxidative stress, and endothelial dysfunction, frailty may also contribute to the association between a declining Hb trajectory and CV events.[29,30] Clinically, these data support that trajectory based risk stratification may complement conventional anemia assessment.[31,32,33] Hb trajectories should be viewed as risk markers that may identify patients who merit closer evaluation of reversible causes of anemia, frailty, and cardiovascular vulnerability within existing guideline-based care.[31,32,33] This approach is best considered an adjunct to, not a replacement for, conventional anemia assessment and individualized clinical judgment.[31,32] More frequent hemoglobin monitoring could help detect an emerging decline in routine care, but whether intervening guided by the trajectory pattern improves outcomes remains unproven.[31,33] Any treatment modification should continue to follow established anemia guidelines and the broader clinical context rather than the trajectory pattern alone.[31,32,33]
Several limitations should be noted for this study. The single-center setting may limit generalizability. The modest sample and event counts limit statistical power for subgroup or interaction testing. Residual confounding is possible because we lacked repeated high-sensitivity inflammatory markers, detailed dietary data, objective measures of physical activity, and protocolized time-updated anemia-treatment dosing. Trajectory assignment, although supported by high entropy values, remains vulnerable to misclassification, and frailty was assessed only at baseline, 6, and 12 months, potentially missing short-term fluctuations. In addition, some CV events may have preceded the next frailty reassessment, limiting temporal sequencing. Because trajectory assignment used Hb information accrued during follow-up, the CV analyses should be interpreted as associations with the longitudinal Hb pattern rather than as time-zero causal estimates. Future research should therefore focus on multicenter validation in larger, ethnically diverse cohorts with extended follow-up to capture kidney-replacement outcomes, on pragmatic randomized trials that test whether trajectory-guided anemia management or frailty-targeted interventions improve hard clinical endpoints, and on mechanistic studies that unravel the roles of iron metabolism, erythropoietin resistance, and skeletal-muscle bioenergetics in progressive anemia.
In summary, with this observational cohort, the shape of a patient’s hemoglobin curve may provide additional prognostic information beyond a single reading: a persistently declining trajectory was associated with higher observed rates of frailty worsening and cardiovascular events in adults with stage 3-5 ND-CKD. Dynamic trajectory-based assessment may help risk recognition, but interventional studies are needed before trajectory patterns are used to guide treatment decisions.
Founding
Hebei Province Medical Applicable Technology Tracking Project (Grant No. GZ2024096); Baoding Science and Technology Program Project (Grant No. 2041ZF160).Ethics approval and consent to participate
The protocol conformed to the Declaration of Helsinki. The Institutional Review Board of Affiliated Hospital of Hebei University approved the study, and all participants gave written informed consent.Data availability statement
Data sets generated during the current study are available from the corresponding author on reasonable request.Author Contribution Statement
The authors confirm contribution to the paper as follows: study conception and design: L G; data collection: L-R, Z-Z H, Y-P Z, J-D L, S-S G; analysis and interpretation of results: L-R, Z-Z H, Y-P Z, J-D L, S-S G; draft manuscript preparation: L-R, Z-Z H, Y-P Z, J-D L, S-S G, L G. All authors reviewed the results and approved the final version of the manuscript.References
- Kovesdy, C. P. (2022). Epidemiology of chronic kidney disease: an update 2022. Kidney International Supplements, 12 1(1), 7–11. https://doi.org/10.1016/j.kisu.2021.11.003
- Bello,
A. K., Okpechi, I. G., Levin, A., & Johnson, D. W. (2024).
Variations in kidney care management and access: regional assessments
of the 2023 International Society of Nephrology Global Kidney Health
Atlas (ISN-GKHA). Kidney International Supplements, 13(1), 1–5. https://doi.org/10.1016/j.kisu.2023.12.001
- Sarnak,
M. J., Levey, A. S., Schoolwerth, A. C., Coresh, J., Culleton, B. F.,
Hamm, L. L., McCullough, P. A., Kasiske, B. L., Kelepouris, E., Klag,
M. J., Parfrey, P. S., Pfeffer, M. A., Raij, L., Spinosa, D. J., &
Wilson, P. W. F. (2003). Kidney Disease as a Risk Factor for
Development of Cardiovascular Disease: A Statement From the American
Heart Association Councils on Kidney in Cardiovascular Disease, High
Blood Pressure Research, Clinical Cardiology, and Epidemiology and
Prevention. Hypertension, 42(5), 1050–1065. https://doi.org/10.1161/01.HYP.0000102971.85504.7C
- Raza,
I. I., Younus, S., Azhar, H., Fatima, H., Anwar, Z., Farah, A. A.,
& Rangwala, H. S. (2024). Transforming the management of chronic
kidney disease-associated anemia using daprodustat. Annals of medicine
and surgery (2012), 86(7), 3824–3826. https://doi.org/10.1097/MS9.0000000000002207
- Mills,
K. T., Xu, Y., Zhang, W., Bundy, J. D., Chen, C. S., Kelly, T. N.,
Chen, J., & He, J. (2015). A systematic analysis of worldwide
population-based data on the global burden of chronic kidney disease in
2010. Kidney International, 88(5), 950–957. https://doi.org/10.1038/ki.2015.230
- Handley
D, Gillett AC, Bala R, Tyrrell J, Lewis CM. Latent class growth mixture
modeling of HbA1C trajectories identifies individuals at high risk of
developing complications of type 2 diabetes mellitus in the UK Biobank.
BMJ Open Diabetes Research & Care. 2025;13(5):e004826. https://doi.org/10.1136/bmjdrc-2024-004826
- Chowdhury,
R., Peel, N. M., Krosch, M., & Hubbard, R. E. (2017). Frailty and
chronic kidney disease: A systematic review. Archives of gerontology
and geriatrics, 68, 135–142. https://doi.org/10.1016/j.archger.2016.10.007
- Afilalo,
J., Karunananthan, S., Eisenberg, M. J., Alexander, K. P., &
Bergman, H. (2009). Role of frailty in patients with cardiovascular
disease. The American journal of cardiology, 103(11), 1616–1621. https://doi.org/10.1016/j.amjcard.2009.01.375
- James,
K., Jamil, Y., Kumar, M., Kwak, M. J., Nanna, M. G., Qazi, S., Troy, A.
L., Butt, J. H., Damluji, A. A., Forman, D. E., & Orkaby, A. R.
(2024). Frailty and Cardiovascular Health. Journal of the American
Heart Association, 13(15), e031736. https://doi.org/10.1161/JAHA.123.031736
- Proust-Lima,
C., & Taylor, J. M. (2009). Development and validation of a dynamic
prognostic tool for prostate cancer recurrence using repeated measures
of posttreatment PSA: a joint modeling approach.Biostatistics (Oxford,
England),10(3), 535–549. https://doi.org/10.1093/biostatistics/kxp009
- Spohr
P, Froehlich RC, Scharf S, Rommerskirchen A, Bobak J, Schweier S,
Jaeger P, Kobbe G, Dietrich S, Dilthey AT, Henrich B, Pfeffer KT, Haas
R, Klau GW. Dynamic Prediction of Mortality Risk Following Allogeneic
Hematopoietic Stem Cell Transplantation. Machine Learning: Health.
2025;1(1). https://doi.org/10.1088/3049-477X/adf74e
- Paik,
H., & Kim, J. (2021). Condensed trajectory of the temporal
correlation of diseases and mortality extracted from over 300,000
patients in hospitals. PLOS ONE, 16(10). https://doi.org/10.1371/JOURNAL.PONE.0257894
- Prezelin-Reydit,
M., Combe, C., Massy, Z. A., Lange, C., Stengel, B., Alencar de Pinho,
N., Harambat, J., & Leffondré, K. (2023). #3381 profiles of
hemoglobin trajectory in ckd patients and associated risks of major
adverse cardiovascular events: the ckd-rein cohort study. Nephrology
Dialysis Transplantation, 38(Supplement_1). https://doi.org/10.1093/ndt/gfad063a_3381
- Kuragano,
T., Okami, S., Tanaka, S., Uenaka, H., Kimura, T., Ishida, Y., James,
G., & Hayasaki, T. (2023). #2560 hemoglobin variability and adverse
clinical events in patients with non-dialysis-dependent chronic kidney
disease and anemia in continuous care. Nephrology Dialysis
Transplantation, 38(Supplement_1). https://doi.org/10.1093/ndt/gfad063a_2560
- Le
Gall, L., Harambat, J., Combe, C., Philipps, V., Proust-Lima, C.,
Dussartre, M., Drüeke, T., Choukroun, G., Fouque, D., Frimat, L.,
Jacquelinet, C., Laville, M., Liabeuf, S., Pecoits-Filho, R., Massy, Z.
A., Stengel, B., Alencar de Pinho, N., Leffondré, K., Prezelin-Reydit,
M., & CKD-REIN study group (2024). Haemoglobin trajectories in
chronic kidney disease and risk of major adverse cardiovascular events.
Nephrology, dialysis, transplantation : official publication of the
European Dialysis and Transplant Association - European Renal
Association, 39(4), 669–682. https://doi.org/10.1093/ndt/gfad235
- Walker,
S. R., Brar, R., Eng, F., Komenda, P., Rigatto, C., Prasad, B., Bohm,
C., Storsley, L., & Tangri, N. (2015). Frailty and physical
function in chronic kidney disease: the CanFIT study. Canadian Journal
of Kidney Health and Disease, 2(1), 32. https://doi.org/10.1186/S40697-015-0067-4
- Le
Gall, L., Harambat, J., Combe, C., Alencar de Pinho, N., Stengel, B.,
Lange, C., Leffondré, K., & Prezelin-Reydit, M. (2024). Anaemia in
CKD and cardiovascular risk; all cut from the same cloth? Nephrology
Dialysis Transplantation, 39(Suppl. 1), gfae069–0670–1122. https://doi.org/10.1093/ndt/gfae069.670
- Wan,
S., Lai, Y., Myers, R. E., Li, B., Palazzo, J. P., Burkart, A. L.,
Chen, G., Xing, J., & Yang, H. (2013). Post-diagnosis hemoglobin
change associates with overall survival of multiple malignancies –
results from a 14-year hospital-based cohort of lung, breast,
colorectal, and liver cancers. BMC Cancer, 13(1), 340. https://doi.org/10.1186/1471-2407-13-340
- Suenaga,
T., Fujino, T., Hashimoto, T., Ishikawa, Y., Shinohara, K., Matsushima,
S., Komman, H., Toyosawa, M., Ide, T., Tsutsui, H., Shiose, A., &
Kinugawa, S. (2024). Hemoglobin Level Can Predict Heart Failure
Hospitalization in Patients with Advanced Heart Failure Awaiting Heart
Transplantation without Inotropes or Mechanical Circulatory Support.
International Heart Journal, 65(4), 667–675. https://doi.org/10.1536/ihj.24-067
- Drüeke,
T. B., Locatelli, F., Clyne, N., Eckardt, K. U., Macdougall, I. C.,
Tsakiris, D., Burger, H. U., Scherhag, A., & CREATE Investigators
(2006). Normalization of hemoglobin level in patients with chronic
kidney disease and anemia. The New England journal of medicine,
355(20), 2071–2084. https://doi.org/10.1056/NEJMoa062276
- Pfeffer,
M. A., Burdmann, E. A., Chen, C. Y., Cooper, M. E., de Zeeuw, D.,
Eckardt, K. U., Feyzi, J. M., Ivanovich, P., Kewalramani, R., Levey, A.
S., Lewis, E. F., McGill, J. B., McMurray, J. J., Parfrey, P., Parving,
H. H., Remuzzi, G., Singh, A. K., Solomon, S. D., Toto, R., & TREAT
Investigators (2009). A trial of darbepoetin alfa in type 2 diabetes
and chronic kidney disease. The New England journal of medicine,
361(21), 2019–2032. https://doi.org/10.1056/NEJMoa0907845
- Macdougall,
I. C., White, C., Anker, S. D., Bhandari, S., Farrington, K., Kalra, P.
A., McMurray, J. J. V., Murray, H., Tomson, C. R. V., Wheeler, D. C.,
Winearls, C. G., Ford, I., & PIVOTAL Investigators and Committees
(2019). Intravenous Iron in Patients Undergoing Maintenance
Hemodialysis. The New England journal of medicine, 380(5), 447–458. https://doi.org/10.1056/NEJMoa1810742
- Taddei,
S., Nami, R., Bruno, R. M., Quatrini, I., & Nuti, R. (2011).
Hypertension, left ventricular hypertrophy and chronic kidney disease.
Heart Failure Reviews, 16(6), 615–620.
https://doi.org/10.1007/S10741-010-9197-Z
- Todorova, G. V.,
Akisheva, A., & Stoimenova, M.-Y. (2022). Anemia and Left
Ventricular Hypertrophy in Chronic Renal Failure. Journal of Biomedical
and Clinical Research, 15(2), 151–157. https://doi.org/10.2478/jbcr-2022-0021
- Liu,
J., Wei, Q., Guo, C., Dong, G., Liu, Y., Tang, C., Dong, Z., &
Dong, Z. (2017). Hypoxia, HIF, and Associated Signaling Networks in
Chronic Kidney Disease. International Journal of Molecular Sciences,
18(5), 950. https://doi.org/10.3390/IJMS18050950
- Butler,
K. G. (2002). Hemoglobin Levels, Cardiovascular Disease, and Left
Ventricular Hypertrophy in Patients with Chronic Kidney Disease.
Nephrology Nursing Journal, 29(2), 189–192.
https://dialnet.unirioja.es/servlet/articulo?codigo=1330742
- Mase,
K., Yamagata, K., Yamamoto, H., Tsuruya, K., Hase, H., Nishi, S.,
Nangaku, M., Wada, T., Hayashi, T., Uemura, Y., & Hirakata, H.
(2023). Predictors of hyporesponsiveness to erythropoiesis-stimulating
agents in patients with non-dialysis-dependent chronic kidney disease
(RADIANCE-CKD study). American Journal of Nephrology, 54(11-12),
471-478. https://doi.org/10.1159/000534438
- Habas, E., Sr, Al
Adab, A., Arryes, M., Alfitori, G., Farfar, K., Habas, A. M., Akbar, R.
A., Rayani, A., Habas, E., & Elzouki, A. (2023). Anemia and Hypoxia
Impact on Chronic Kidney Disease Onset and Progression: Review and
Updates. Cureus, 15(10), e46737. https://doi.org/10.7759/cureus.46737
- Volis,
I., & Zafrir, B. (2024). Frailty and Cardiovascular Disease: A
Bidirectional Association. Cardiology, 149(3), 205–207. https://doi.org/10.1159/000535494
- Govindarajulu,
U. S. & Qadri, M. (2019). Survival and Mediation Analysis with
Correlated Frailty. Current Research in Biostatistics, 9(1), 21-30. https://doi.org/10.3844/amjbsp.2019.21.30
- Locatelli,
F., Bárány, P., Covic, A., de Francisco, A. L. M., Del Vecchio, L.,
Goldsmith, D., Hörl, W. H., London, G. M., Vanholder, R., & Van
Biesen, W. (2013). Kidney Disease: Improving Global Outcomes guidelines
on anaemia management in chronic kidney disease: a European Renal Best
Practice position statement. Nephrology Dialysis Transplantation,
28(6), 1346–1359. https://doi.org/10.1093/NDT/GFT033
- Locatelli,
F., Aljama, P., Canaud, B., Covic, A., de Francisco, A. L. M.,
Macdougall, I. C., Wiecek, A., & Vanholder, R. (2010). Target
haemoglobin to aim for with erythropoiesis-stimulating agents: a
position statement by ERBP following publication of the Trial to Reduce
Cardiovascular Events with Aranesp® Therapy (TREAT) Study. Nephrology
Dialysis Transplantation, 25(9), 2846–2850. https://doi.org/10.1093/NDT/GFQ336
- Kidney
Disease: Improving Global Outcomes (KDIGO) Anemia Work Group. (2026).
KDIGO 2026 clinical practice guideline for the management of anemia in
chronic kidney disease (CKD). Kidney International, 109(1 Suppl),
S1-S99. https://doi.org/10.1016/j.kint.2025.06.006