Case Report
Case 1. A 63-year-old male presented to a local hospital with a three-month history of bone pain. CT imaging revealed multiple fractures in bilateral ribs and the 8th, 10th, 11th, and 12th thoracic vertebrae. Peripheral blood counts showed a white blood cell count of 20.4x10⁹/L, hemoglobin of 58 g/L, and platelet count of 259x10⁹/L. Serum biochemistry indicated total protein 48.1 g/L, albumin 29 g/L, urea 49.78 mmol/L, creatinine 626 µmol/L, and uric acid 785 µmol/L. Serum immunofixation electrophoresis demonstrated a lambda light chain type. A diagnosis of multiple myeloma was suspected at the local hospital, and the patient received supportive care, including denosumab for bone protection.The patient was subsequently transferred to our institution for further management. Urinary immunoglobulin light chain analysis showed an elevated kappa light chain level of 8.38 mg/L and a markedly elevated lambda light chain level >240.00 mg/L. Serum protein electrophoresis revealed an M-protein of 4.30 g/L of the lambda light chain type. The β2-microglobulin level was significantly elevated (>10,000.00 µg/L). Bone marrow aspiration cytology demonstrated 10.5% plasmablasts and 19.5% mature plasma cells. Flow cytometric analysis of the bone marrow identified a population of 25% abnormal plasma cells positive for CD38++, CD138++, and cytoplasmic lambda light chain restriction. Bone marrow biopsy showed approximately 50% plasma cell infiltration. Immunohistochemistry results were positive for CD38 (partial), CD138 (partial), Lambda, and MUM-1 (partial), while negative for Kappa, CD3, CD20, and CD56. A diagnosis of multiple myeloma was confirmed.
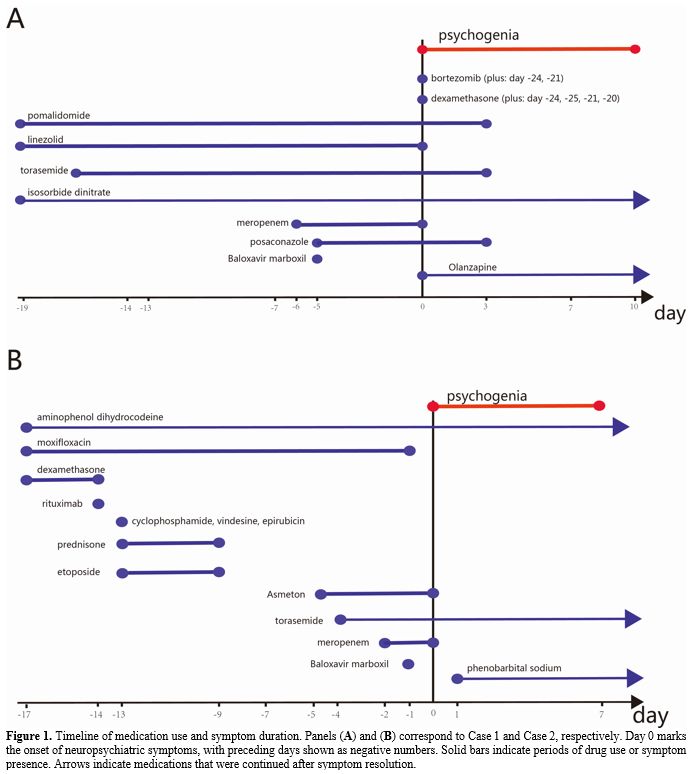
The patient started on the VPD induction regimen (bortezomib 1mg on days 1 and 2, and 2.0mg on day 4; dexamethasone 20.25mg on days 1-2 and 4-5; pomalidomide 4mg on days 1-21). During treatment, he developed acute heart failure and tested positive for influenza antigen A. Chest CT indicated pulmonary infection, suggesting a mixed etiology. Management included a single oral dose of Baloxavir marboxil (80mg), along with meropenem, linezolid, and posaconazole for infection control. Furosemide and isosorbide dinitrate were administered for heart failure. Antineoplastic therapy was temporarily withheld. While the infectious symptoms gradually improved, the patient developed delirium on the fifth day after Baloxavir marboxil administration. Clinical manifestations included generalized tremors, agitation, logorrhea, incoherent speech, disorientation, and fragmented recollection of past events. Psychiatric assessment revealed an altered level of consciousness with poor communication; he was largely unresponsive to questions, preventing formal evaluation of perception and thought content. A head CT scan showed no abnormalities. Delirium severity was assessed using the Delirium Rating Scale-Revised-98 (DRS-R-98), yielding a score of 23.[1] Olanzapine (2.5-5mg/day) was initiated, and meropenem was discontinued. The psychiatric symptoms resolved after ten days (Figure 1A).
Case 2. A 40-year-old female was admitted to the hospital with a 17-day history of cough and fever, and a 10-day history of a discovered mediastinal mass. PET-CT revealed a hypermetabolic mass in the anterior and middle mediastinum, with swelling in bilateral neck regions, chest walls, and breasts, alongside inflammatory reactive hyperplasia in bilateral axillary lymph nodes. A patchy shadow with increased FDG uptake was noted in the subpleural area of the right lung. Pathological examination of a biopsy from the anterior mediastinal mass showed numerous severely crushed small round cells with hyperchromatic, atypical nuclei within proliferative fibrous tissue, accompanied by extensive necrosis. Immunohistochemistry results were as follows: CKpan (-), CD3 (-), CD20 (+), Pax-5 (+), Bcl-2 (+), Bcl-6 (+), CD10 (-), MUM1 (+), c-Myc (+, 5%), CD5 (-), CD21 (-), CD23 (+), CyclinD1 (-), Ki-67 (+, 80%), CD30 (-). A diagnosis of primary mediastinal diffuse large B-cell lymphoma was established. Upper-extremity vascular ultrasound showed thrombosis in the bilateral subclavian and jugular veins.
The patient was initiated on the R-CHOPE chemotherapy regimen (rituximab 0.6g on day 0; cyclophosphamide 1.2g, vindesine 4mg, and epirubicin 100mg on day 1; prednisone 100mg and etoposide 100mg on days 1-5). Concurrent medications included furosemide for diuresis and rivaroxaban for anticoagulation. Despite antimicrobial therapy with moxifloxacin and meropenem, intermittent fever persisted. On day 13 post-chemotherapy, testing returned a weakly positive result for influenza A virus IgM antibody. The patient subsequently received a single 80mg oral dose of Baloxavir marboxil. On the following night, she abruptly developed psychiatric abnormalities, manifesting as tightly closed eyes, refusal to communicate, and occasional agitation. Non-contrast CT of the brain showed no significant abnormalities. Her clinical presentation was scored using the Delirium Detection Scale (DDS), yielding a score of 16.[2] A single dose of phenobarbital sodium was administered. Meropenem was discontinued and piperacillin-tazobactam was substituted. The patient's psychiatric symptoms resolved one week later (Figure 1B).
Discussion
Influenza infection can induce acute neuropsychiatric symptoms, primarily manifesting as delirium and abnormal behavior. Delirium is more commonly observed in children and adolescent patients, characterized by confusion, disorientation, anxiety, or hallucinations. Several Japanese studies describe influenza-associated delirium as a transient, reversible cognitive dysfunction that often accompanies fever and lasts for minutes to hours.[3] Population-based studies from Western cohorts have quantified the risk of serious influenza-associated neuropsychiatric events requiring hospitalization, confirming their occurrence beyond the well-described Japanese context.[4]Early clinical trials of Baloxavir marboxil did not specifically mention psychiatric symptoms. In the phase III trial conducted by Hayden et al., the incidence of adverse events was similar between the Baloxavir marboxil and placebo groups (20.7% vs. 24.6%), with no reports of serious neuropsychiatric events.[5]
However, post-marketing safety surveillance data have revealed new signals. For instance, Zhou et al., in their analysis of FAERS data from 2018 to 2023, identified a significant safety signal linking Baloxavir marboxil to psychiatric symptoms. Among 1700 relevant reports, 49 Preferred Terms (PTs) were flagged as significant signals. This included 89 reports categorized under psychiatric disorders, encompassing hallucinations, abnormal behavior, and delirium.[6]
The study by Zhou et al. indicated that adverse events (AEs) of psychiatric disorders associated with baloxavir marboxil include abnormal behavior, hallucinations, and confusion, while the incidence of psychiatric system abnormalities was significantly lower than that associated with oseltamivir.[7]
Furthermore, when comparing the safety profiles of Baloxavir marboxil and oseltamivir, Li et al. observed that Baloxavir marboxil did not exhibit significant neurological safety signals in patients younger than 18 years.[8] In adult patients, however, the reporting odds ratios (ROR) for depressed level of consciousness and encephalopathy were 4.52 and 6.74, respectively,[8] suggesting that age may be an important factor influencing the occurrence of these adverse events.
Reports on Baloxavir marboxil's neuropsychiatric adverse events have also focused on concomitant medications. Zhou et al. found that the signal strength for abnormal behavior increased significantly when Baloxavir marboxil was co-administered with Calonal (an acetaminophen preparation). Conversely, concomitant use with acetaminophen was associated with a notably prominent signal for febrile delirium.[6]
We present two patients with hematologic malignancies who developed psychiatric symptoms following influenza A infection. Both patients received a single dose of baloxavir shortly after influenza diagnosis and subsequently developed neuropsychiatric symptoms within days. Applying the Naranjo Adverse Drug Reaction Probability Scale yielded a score of 2 (“possible”) for both, indicating an uncertain causal association with Baloxavir marboxil.[9] This reflects the significant challenge of attribution in such complex clinical scenarios. This "possible" classification and the inherent uncertainty are consistent with the nature of signals identified in pharmacovigilance studies, which indicate a potential association requiring further scrutiny rather than confirming causation. The observed delirium in these patients likely resulted from a confluence of factors. First, active influenza infection is a known precipitant of acute encephalopathy.[3] Second, both patients were receiving neurotoxic chemotherapy: Case 1 was on bortezomib and high-dose dexamethasone; Case 2 received high-dose corticosteroids as part of R-CHOPE.[10] Third, Case 1 had significant renal failure (creatinine 626 µmol/L), which can cause uremic encephalopathy.[11] Fourth, and notably, both patients were on meropenem — a carbapenem antibiotic with well-documented neurotoxic potential, including delirium, especially in the setting of renal impairment.[12] Furthermore, both patients were newly diagnosed with high tumor burden, poor overall performance status, and multiple comorbidities.
Conclusions
The etiology of influenza A-associated psychiatric symptoms is likely multifactorial. Further investigation is required to determine whether a causal relationship exists between Baloxavir marboxil and the reported neuropsychiatric adverse events.Author contributions
Zhan Su and Yao Li designed the study and drafted the manuscript. Xiaojia Bu collected clinical information. All authors read and approved the final manuscript.Ethics approval
The study protocol was approved by the Medical Ethics Committee of the affiliated hospital of Qingdao University (No.: QYFY WZLL 28785).References
- Trzepacz, P. T.; Mittal, D.; Torres, R.; Kanary,
K.; Norton, J.; Jimerson, N. Validation of the Delirium Rating
Scale-Revised-98: Comparison with the Delirium Rating Scale and the
Cognitive Test for Delirium. J Neuropsychiatry Clin Neurosci, 2001; 13
(2): 229-242. https://doi.org/10.1176/jnp.13.2.229 PMid:11449030 PMCid:PMC12398295
- Grover, S. Assessment Scales for Delirium: A Review. World J Psychiatr, 2012; 2 (4): 58. https://doi.org/10.5498/wjp.v2.i4.58 PMid:24175169 PMCid:PMC3782167
- Mizuguchi, M. Influenza Encephalopathy and Related Neuropsychiatric Syndromes. Influenza Resp Viruses, 2013; 7 (s3): 67-71. https://doi.org/10.1111/irv.12177 PMid:24215384 PMCid:PMC6492655
- Antoon,
J. W.; Williams, D. J.; Bruce, J.; Sekmen, M.; Zhu, Y.; Edwards, K. M.;
Grijalva, C. G. Population-Based Incidence of Influenza-Associated
Serious Neuropsychiatric Events in Children and Adolescents. Jama
Pediatr, 2023; 177 (9): 967. https://doi.org/10.1001/jamapediatrics.2023.2304 PMid:37486679 PMCid:PMC10366945
- Hayden,
F. G.; Sugaya, N.; Hirotsu, N.; Lee, N.; De Jong, M. D.; Hurt, A. C.;
Ishida, T.; Sekino, H.; Yamada, K.; Portsmouth, S.; et al. Baloxavir
Marboxil for Uncomplicated Influenza in Adults and Adolescents. N Engl
J Med, 2018; 379 (10): 913-923. https://doi.org/10.1056/NEJMoa1716197 PMid:30184455
- Zhou,
J.; Ye, J.; Chen, M.; Zheng, X. A Real-World Disproportionality
Analysis of Baloxavir Marboxil: Post-Marketing Pharmacovigilance Data.
Expert Opinion on Drug Safety, 2025;24(10):1231-1239. https://doi.org/10.1080/14740338.2024.2393269 PMid:39234783
- Zhou,
Y.; Jin, L.; Lai, X.; Li, Y.; Sheng, L.; Xie, G.; Fang, J. Adverse
Events Associated with Oseltamivir and Baloxavir Marboxil in against
Influenza Virus Therapy: A Pharmacovigilance Study Using the FAERS
Database. PLoS ONE, 2024; 19 (11): e0308998. https://doi.org/10.1371/journal.pone.0308998 PMid:39536015 PMCid:PMC11560017
- Li,
Y.; Wang, X.; Liao, Y.; Zeng, Y.; Lin, W.; Zhuang, W. Safety Analysis
of Oseltamivir and Baloxavir Marboxil after Market Approval: A
Pharmacovigilance Study Based on the FDA Adverse Event Reporting
System. Bmc Infect Dis, 2024; 24 (1): 446. https://doi.org/10.1186/s12879-024-09339-4 PMid:38724914 PMCid:PMC11080077
- Naranjo,
C. A.; Busto, U.; Sellers, E. M.; Sandor, P.; Ruiz, I.; Roberts, E. A.;
Janecek, E.; Domecq, C.; Greenblatt, D. J. A Method for Estimating the
Probability of Adverse Drug Reactions. Clin Pharmacol Ther, 1981; 30
(2): 239-245. https://doi.org/10.1038/clpt.1981.154 PMid:7249508
- Pasyk,
S.; Maharaj, S.; Bourgeois, J. A. Psychotoxicity of Immunomodulators:
Corticosteroids, Mycophenolate, Tacrolimus, Cyclophosphamide, and
Hydroxychloroquine. In Psychonephrology; Hategan, A., Bourgeois, J. A.,
Gangji, A. S., Woo, T. K. W., Eds.; Springer International Publishing:
Cham, 2022; pp 241-252. https://doi.org/10.1007/978-3-030-84740-1_14
- Brouns, R.; De Deyn, P. P. Neurological Complications in Renal Failure: A Review. Clin Neurol Neurosur, 2004;107 (1): 1-16. https://doi.org/10.1016/j.clineuro.2004.07.012 PMid:15567546
- Grill,
M. F.; Maganti, R. K. Neurotoxic Effects Associated with Antibiotic
Use: Management Considerations. Brit J Clin Pharmaco, 2011; 72 (3):
381-393. https://doi.org/10.1111/j.1365-2125.2011.03991.x PMid:21501212 PMCid:PMC3175508