Among individuals with hematologic malignancies, reported colonization rates range from 3.8% to 21%,[6,7] while in hematopoietic stem cell transplantation (HSCT) recipients, colonization prevalence may reach 26–36%.[8] HSCT recipients are uniquely predisposed to invasive infections due to prolonged neutropenia, mucosal barrier injury, exposure to broad-spectrum antibiotics, graft-versus-host disease (GVHD), and frequent healthcare contact. Despite this high-risk profile, factors associated with progression from CRE colonization to bacteremia in HSCT recipients remain incompletely characterized, and most available data derive from mixed oncologic populations.
Materials and Methods
We therefore conducted a retrospective cohort study to identify clinical factors associated with progression from CRE colonization to bacteremia among HSCT recipients at a tertiary referral center in Bogotá, Colombia, between January 2017 and June 2024.All adult patients (≥18 years) with hematologic malignancies undergoing HSCT and documented CRE colonization were eligible. At our institution, CRE colonization screening is performed using rectal swabs at admission and weekly thereafter. Samples are processed with the HB&L® system (Alifax, Italy), an automated laser nephelometry–based analyzer that detects bacterial growth in liquid culture within a few hours, with reported sensitivities of approximately 85% and specificities of 100%.[9]
To capture known carriers while maintaining clinical relevance, we included patients colonized from 365 days prior to HSCT to 30 days after HSCT. For analytical purposes, the most recent colonization episode prior to transplantation (or prior to bacteremia in affected patients) was considered. The primary outcome was the cumulative incidence of CRE bacteremia during hospitalization and up to 90 days post-transplant. Platelet engraftment was defined as the first of three consecutive days with platelet counts ≥20,000/µL without transfusion; neutrophil engraftment was defined as three consecutive days with neutrophils ≥500/µL. Time-to-event analyses were conducted using the Nelson–Aalen estimator and compared using log-rank testing.
Results
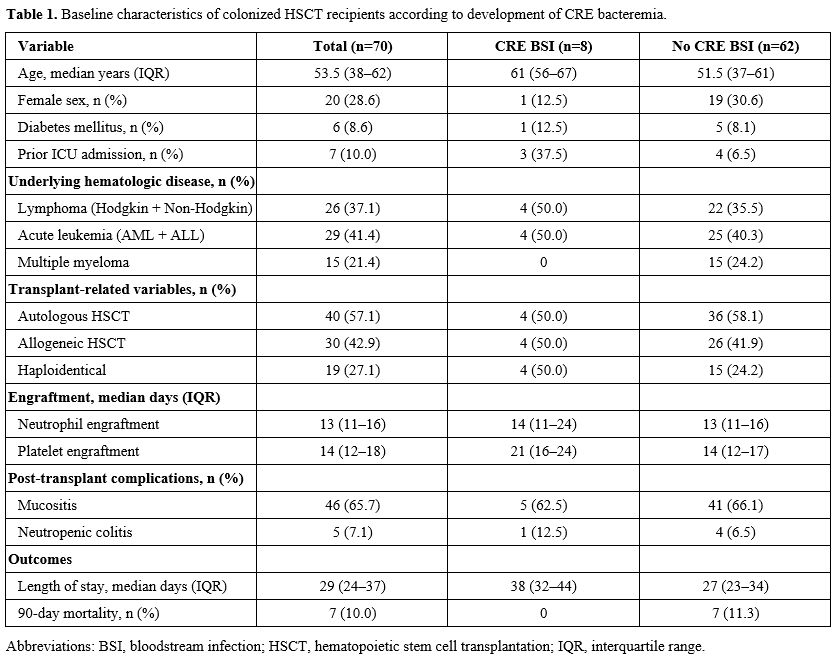
During the study period, 790 HSCT procedures were performed (593 autologous and 197 allogeneic). Ninety-seven patients were identified as colonized with CRE. After exclusions based on predefined criteria, 70 colonized patients were included in the final analysis. Eight patients (11%) developed CRE bacteremia.The median age of the cohort was 53.5 years (IQR 38–62). Patients who developed CRE bacteremia were significantly older than those who did not (median 61 vs. 51.5 years, p=0.03). Baseline characteristics, including sex, hematologic diagnosis, transplant type (autologous vs. allogeneic), conditioning regimen, mucositis, neutropenic colitis, and neutrophil engraftment time, were comparable between groups (Table 1).
 |
|
Prior intensive care unit (ICU) admission was more frequent among patients who developed CRE bacteremia (37.5% vs. 6.5%). Moreover, platelet engraftment occurred later in the bacteremia group (median 21 vs. 14 days), while neutrophil engraftment time was comparable between groups.
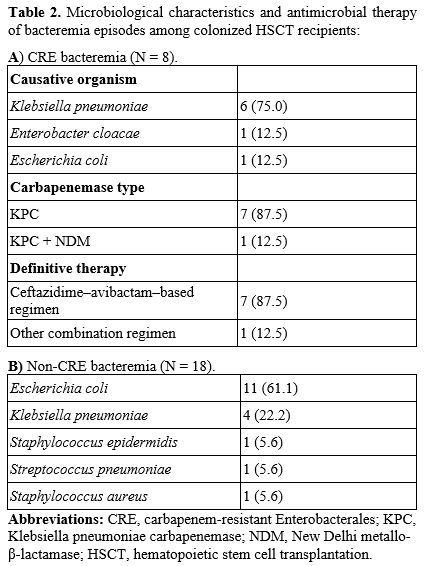
Within 90 days post-transplantation, 26 patients (37.1%) developed bacteremia from any cause, of which 8 episodes (11.4% of the cohort) were attributable to CRE. The median interval from documented colonization to CRE bacteremia was 13 days (IQR 9–15.5). All CRE bacteremia cases occurred in patients colonized prior to HSCT, and 87.5% developed within the first 15 days following transplantation (Table 2). No episodes occurred beyond 54 days after colonization.
 |
|
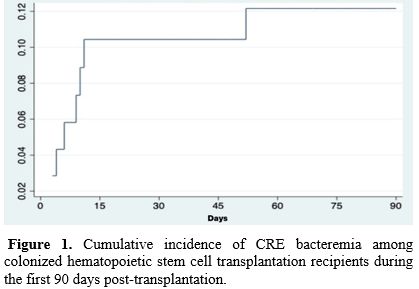
Cumulative incidence curves revealed a higher probability of CRE bacteremia among patients with platelet engraftment >20 days (log-rank p=0.006) and among those with prior ICU admission (log-rank p=0.0073) (Figure 1). A comparable pattern was observed in patients aged ≥60 years, though it was not statistically significant (log-rank p=0.11).
 |
|
Klebsiella pneumoniae was the predominant causative organism (75%), largely KPC-producing strains. Definitive therapy most commonly included ceftazidime–avibactam, administered either as monotherapy or in combination. Patients who developed CRE bacteremia experienced prolonged hospitalization (median 38 vs. 27 days). Ninety-day mortality was similar between groups.
Discussion
The 11% progression rate observed in our cohort is lower than the 15–20% rates previously reported among colonized HSCT recipients and patients with hematologic malignancies.[12–15] This difference may be partly explained by the high proportion of autologous transplants in our cohort, which are generally associated with shorter neutropenia durations and less profound immunosuppression than allogeneic transplants.Age was associated with bacteremia in the univariate analysis; however, cumulative incidence stratification did not reach statistical significance. Prior ICU admission emerged as one of the strongest associations, consistent with previous reports demonstrating increased infection risk among critically ill CRE carriers.[15–17] ICU admission likely reflects a composite marker of disease severity, exposure to invasive devices, broad-spectrum antibiotic pressure, and mucosal barrier disruption, all of which may facilitate bacterial translocation and hematogenous dissemination. These associations should be interpreted with caution and considered exploratory and hypothesis-generating rather than definitive predictors.
Delayed platelet engraftment was also associated with CRE bacteremia. Platelet recovery may represent a surrogate marker of bone marrow resilience and immune reconstitution.[18] Conversely, prolonged cytopenia may indicate sustained systemic inflammation or subclinical infection. Given the retrospective design, causality cannot be established, and reverse causation remains a plausible explanation. These findings should be interpreted with caution and considered exploratory.
The temporal clustering of CRE bacteremia within the first two weeks following transplantation underscores the clinical relevance of recent colonization status. Prior studies have similarly demonstrated that infection risk is highest shortly after colonization acquisition.[5,19] These observations support the importance of close clinical surveillance during the early post-transplant period and may help identify patients who could benefit from a more individualized approach to empirical antimicrobial therapy.
This study has several limitations. The small number of CRE bacteremia events (n=8) limits statistical power and the precision of the estimates. Therefore, multivariable analysis was not performed due to the limited number of outcome events and the risk of model overfitting. The retrospective single-center design may restrict generalizability. Furthermore, phenotypic screening methods based on laser nephelometry may be less sensitive than molecular assays, potentially leading to underestimation of colonization despite their high specificity. Additionally, the predominance of autologous HSCT recipients in our cohort may limit the extrapolation of these findings to allogeneic transplant populations. Nevertheless, our study specifically focuses on colonized HSCT recipients, an underrepresented population in prior analyses, and provides clinically meaningful insights into early risk stratification.
Conclusions
In conclusion, among HSCT recipients colonized with CRE, prior ICU admission and delayed platelet engraftment were associated with an increased probability of subsequent CRE bacteremia. Most infections occurred within the first two weeks following transplantation. These findings underscore the importance of close clinical monitoring and may inform empirical antimicrobial strategies in high-risk colonized patients. However, these findings should be interpreted cautiously as hypothesis-generating, and prospective multicenter studies are needed to validate these associations and develop predictive models for targeted intervention.Ethics Approval
Approved by the Institutional Ethics Committee of Hospital Universitario San Ignacio (FM-CIE-1269-24). Informed consent was waived due to retrospective design.Authors’ contributions
DRF contributed to study design, data collection, statistical analysis, and manuscript drafting. CDB contributed to study conception, methodological supervision, data interpretation, and critical revision of the manuscript. JG participated in data collection and clinical interpretation. OM contributed to the study design and critical revision of the manuscript. All authors read and approved the final manuscript.Availability of data and materials
The datasets generated and/or analyzed during the current study are not publicly available due to institutional data protection policies, but are available from the corresponding author on reasonable request.References
- Budia-Silva
M, Kostyanev T, Ayala-Montaño S, Bravo-Ferrer Acosta J, Garcia-Castillo
M, Cantón R, et al. International and regional spread of
carbapenem-resistant Klebsiella pneumoniae in Europe. Nat Commun.
2024;15(1):5092. https://doi.org/10.1038/s41467-024-49349-z
- Cassini
A, Högberg LD, Plachouras D, Quattrocchi A, Hoxha A, Simonsen GS, et
al. Attributable deaths and disability-adjusted life-years caused by
infections with antibiotic-resistant bacteria in the EU and the
European Economic Area in 2015: a population-level modelling analysis.
Lancet Infect Dis. 2019;19(1):56–66. https://doi.org/10.1016/S1473-3099(18)30605-4
- Trecarichi
EM, Pagano L, Martino B, Candoni A, Di Blasi R, Nadali G, et al.
Bloodstream infections caused by Klebsiella pneumoniae in
onco-hematological patients: clinical impact of carbapenem resistance
in a multicentre prospective survey. Am J Hematol. 2016;91(11):1076–81.
https://doi.org/10.1002/ajh.24489
- Gutiérrez-Gutiérrez
B, Salamanca E, de Cueto M, Hsueh PR, Viale P, Paño-Pardo JR, et al.
Effect of appropriate combination therapy on mortality of patients with
bloodstream infections due to carbapenemase-producing
Enterobacteriaceae (INCREMENT): a retrospective cohort study. Lancet
Infect Dis. 2017;17(7):726–34. https://doi.org/10.1016/S1473-3099(17)30228-1
- Tischendorf
J, de Avila RA, Safdar N. Risk of infection following colonization with
carbapenem-resistant Enterobacteriaceae: a systematic review. Am J
Infect Control. 2016;44(5):539–43. https://doi.org/10.1016/j.ajic.2015.12.005
- Andria
N, Henig O, Kotler O, Domchenko A, Oren I, Zuckerman T, et al.
Mortality burden related to infection with carbapenem-resistant
Gram-negative bacteria among haematological cancer patients: a
retrospective cohort study. J Antimicrob Chemother.
2015;70(11):3146–53. https://doi.org/10.1093/jac/dkv218
- Jaiswal
SR, Gupta S, Kumar RS, Sherawat A, Rajoreya A, Dash SK, et al. Gut
colonization with carbapenem-resistant Enterobacteriaceae adversely
impacts the outcome in patients with hematological malignancies:
results of a prospective surveillance study. Mediterr J Hematol Infect
Dis. 2018;10(1):e2018025. https://doi.org/10.4084/MJHID.2018.025
- Girmenia C, Rossolini GM, Piciocchi A, Bertaina A, Pisapia G, Pastore D, et al. Infections by carbapenem-resistant Klebsiella pneumoniae in SCT recipients: a nationwide retrospective survey from Italy. Bone Marrow Transplant. 2015;50(2):282–8. https://doi.org/10.1038/bmt.2014.231
- Josa
DF, Bustos G, Torres IC, Esparza G. Evaluación de tres métodos de
tamizaje para detección de Enterobacteriaceae productoras de
carbapenemasas en hisopados rectales. Rev Chil Infectol.
2018;35(3):253–61. https://doi.org/10.4067/s0716-10182018000300253
- Lalaoui
R, Javelle E, Bakour S, Ubeda C, Rolain JM. Infections due to
carbapenem-resistant bacteria in patients with hematologic
malignancies. Front Microbiol. 2020;11:1422. https://doi.org/10.3389/fmicb.2020.01422
- Averbuch
D, Tridello G, Hoek J, Mikulska M, Akan H, Yanez San Segundo L, et al.
Antimicrobial resistance in gram-negative rods causing bacteremia in
hematopoietic stem cell transplant recipients: intercontinental
prospective study. Clin Infect Dis. 2017;65(11):1819–28. https://doi.org/10.1093/cid/cix646
- Cao
W, Zhang J, Bian Z, Li L, Zhang S, Qin Y, et al. Active screening of
intestinal colonization of carbapenem-resistant Enterobacteriaceae for
subsequent bloodstream infection in allogeneic hematopoietic stem cell
transplantation. Infect Drug Resist. 2022;15:5993–6006. https://doi.org/10.2147/IDR.S387615
- Kömürcü
B, Tükenmez Tigen E, Toptaş T, Fıratlı Tuğlular T, Korten V. Rectal
colonization with multidrug-resistant gram-negative bacteria in
patients with hematological malignancies: a prospective study. Expert
Rev Hematol. 2020;13(8):923–7. https://doi.org/10.1080/17474086.2020.1787145
- Chen
X, Wen X, Jiang Z, Yan Q. Prevalence and factors associated with
carbapenem-resistant Enterobacterales infection among hematological
malignancies patients with CRE intestinal colonization. Ann Clin
Microbiol Antimicrob. 2023;22(1):3. https://doi.org/10.1186/s12941-023-00554-6
- Giannella
M, Trecarichi EM, De Rosa FG, Del Bono V, Bassetti M, Lewis RE, et al.
Risk factors for carbapenem-resistant Klebsiella pneumoniae bloodstream
infection among rectal carriers: a prospective observational
multicentre study. Clin Microbiol Infect. 2014;20(12):1357–62. https://doi.org/10.1111/1469-0691.12747
- Wang
X, Liu J, Li A. Incidence and risk factors for subsequent infections
among rectal carriers with carbapenem-resistant Klebsiella pneumoniae:
a systematic review and meta-analysis. J Hosp Infect. 2024;145:11–21. https://doi.org/10.1016/j.jhin.2023.12.002
- Paramythiotou
E, Routsi C. Association between infections caused by
multidrug-resistant gram-negative bacteria and mortality in critically
ill patients. World J Crit Care Med. 2016;5(2):111–20. https://doi.org/10.5492/wjccm.v5.i2.111
- Kimura
F, Kobayashi S, Ohashi K, Taniguchi S, Mori T, Inoue M, et al. Platelet
engraftment failure leads to poor overall survival even after
neutrophil engraftment without relapse. Biol Blood Marrow Transplant.
2013;19(2 Suppl):S120–1. https://doi.org/10.1016/j.bbmt.2012.11.048
- Cano
Á, Gutiérrez-Gutiérrez B, Machuca I, Torre-Giménez J, Gracia-Ahufinger
I, Natera AM, et al. Association between timing of colonization and
risk of developing carbapenemase-producing Klebsiella pneumoniae
infection in hospitalized patients. Microbiol Spectr.
2022;10(2):e01970-21. https://doi.org/10.1128/spectrum.01970-21