The persistence of SeAO is commonly attributed to protection against severe malaria, consistent with balanced polymorphism analogous to sickle cell disease and thalassaemia.[4] Although long regarded as clinically benign beyond infancy, accumulating evidence indicates that SeAO is associated with a broader spectrum of haematological and systemic manifestations. Clinical studies have linked SeAO to haemolysis, neonatal jaundice, anaemia, and distal renal tubular acidosis (dRTA), reflecting the dual erythrocyte and renal expression of Band 3.[5] Additionally, SeAO-associated SLC4A1 variants have been shown to influence HbA1c levels independently of glycaemia, with important implications for diabetes diagnosis in affected populations.[6]
Despite these emerging clinical insights and substantial population burden, SeAO research has not been systematically examined. This study aims to map global research trends, citation impact, collaborative networks, and thematic evolution in SeAO research to identify knowledge gaps and inform future haematological and translational research priorities.
Material and Methods
Data Source and Search Strategy. A bibliometric analysis was conducted using the Scopus database. A structured concept-block search strategy was applied to the TITLE-ABS-KEY fields using the following queries:Primary search: (“southeast asian ovalocytosis” OR “south-east asian ovalocytosis” OR “hereditary ovalocytosis”) AND (SLC4A1 OR “band 3” OR AE1)
Supplementary search: ovalocytosis AND (SLC4A1 OR “band 3” OR AE1)
Eligible publications comprised peer-reviewed original research articles and reviews addressing human SeAO or SLC4A1-related ovalocytosis, including clinical, haematological, pathophysiological, and genetic aspects. Grey literature, including conference abstracts, preprints, and non-peer-reviewed reports, was excluded. The search was restricted to English-language human studies with no limits on publication year or geographical region. The final search was conducted on 18 January 2026.
Study Screening and Bibliometric Analysis. Records from both searches were merged and deduplicated using EndNote. Screening was conducted manually at the title and abstract level. Bibliographic records, including authors, institutional affiliations, article titles, abstracts, keywords, source titles, cited references, publication year, and citation counts, were exported to VOSviewer (version 1.6.20).[7] Citation counts included self-citations as reported in the Scopus database. Harmonisation of author and index keywords was performed using a controlled thesaurus to standardise spelling variants, abbreviations, and synonymous terms (e.g., “SAO” vs “Southeast Asian ovalocytosis”; “Band 3,” “AE1,” and “SLC4A1”). The harmonised dataset was analysed for keyword co-occurrence, co-authorship and country collaboration networks, co-citation analysis, and overlay visualisation.
Results
Study Selection and Descriptive Characteristics. The main and supplementary searches identified 700 and 730 records, respectively. After merging, deduplication, and screening, 228 publications were included for analysis. These studies were published between 1990 and 2024, comprising predominantly original research articles (n=212, 92.6%) and review articles (n=16, 7.4%). Collectively, the publications accumulated 9,158 citations. Study selection is illustrated in the modified PRISMA flow diagram (Figure 1).Scientific Production and Journal Dispersion. SeAO research output was consistently low and intermittent, averaging 2-15 publications annually. After emerging in the early 1990s, publications increased modestly from the late 1990s to the mid-2000s, then went into sporadic output. Despite their small number, early foundational studies[8,9] achieved the highest citation impact. Citation distribution was highly skewed toward a small group of journals. Blood, Journal of Biological Chemistry, and Biochemical Journal accounted for over 80% of total citations (3,166 citations from 43 documents). In contrast, clinically oriented journals such as the British Journal of Haematology (168 citations; 7 documents) and Journal of Human Genetics (109 citations; 6 documents) generated substantially fewer citations.
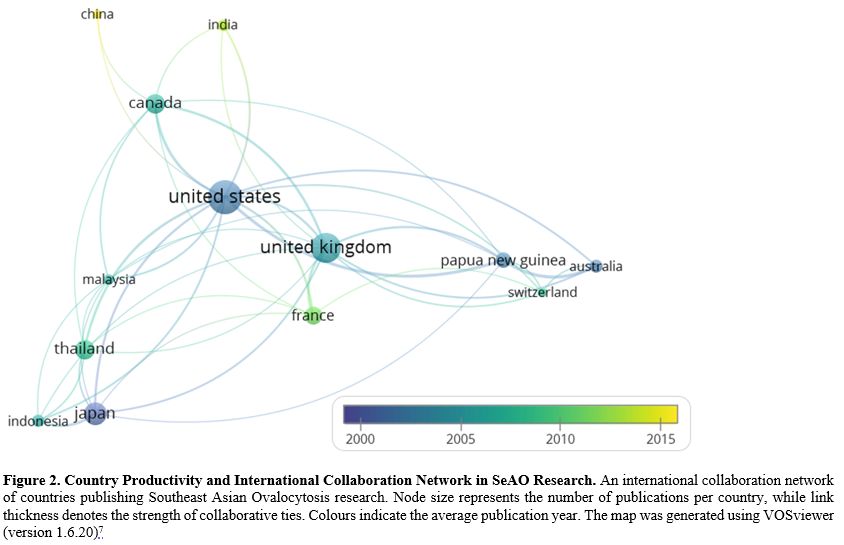
Author Productivity and Geographical Distribution. An analysis of author productivity showed a highly centralised collaboration network dominated by a few prolific investigators. Michael J. A. Tanner was the most influential author (25 publications; 1,926 citations; total link strength = 31). Early contributors such as Petr Jarolím, Jiri Palek, and David J. Anstee achieved high citation impact despite fewer publications, reflecting their foundational roles in erythrocyte membrane biology. At the country level, the United States (72 publications; 3,607 citations) and the United Kingdom (56 publications; 3,168 citations) led both output and international collaboration. Authors from endemic countries, including Pa-Thai Yenchitsomanus, Somkiat Vasuvattakul, and Hélène Guizouarn, formed smaller, less connected clusters with more recent publication activity. Figure 2 presents the country-level productivity and collaboration network in SeAO research.
Thematic Mapping and Evolution. The most prominent keywords identified through co-occurrence analysis were ovalocytosis (occurrences=97; total link strength=554), membrane (80; 441), AE1/band 3 (68; 417), and SLC4A1 (51; 313), underscoring the central role of erythrocyte membrane structure and anion exchange biology. Clinical-genetic links were represented by distal renal tubular acidosis (54; 378) and gene (59; 365). In contrast, population- and region-related keywords such as Southeast Asia (14: 87), Papua New Guinea (18; 112), and malaria (24; 148) formed smaller, less dominant clusters. Overlay analysis revealed a clear temporal shift in research focus. Early studies (average publication years 1996-2001) centred on structural and functional terms (e.g., cytoplasmic domain, ankyrin), whereas later publications increasingly emphasised clinical-genetic themes, including distal renal tubular acidosis, SLC4A1, and haemolytic anaemia. The network visualisation of keyword co-occurrence is illustrated in Figure 3.
 |
|
Discussion
This bibliometric analysis demonstrates a phased thematic evolution of SeAO research over more than three decades. Early foundational work (1995–2005) centred on SLC4A1 (Band 3) molecular biology, with paradigm-defining genetic and structural studies establishing the field’s enduring mechanistic framework.[8,9] From the early 2000s to 2010, research expanded into clinical haematology, focusing on phenotypic expression and differentiation from other red cell membrane disorders.[10] Between 2010 and 2018, attention shifted toward evolutionary and biomechanical themes, particularly malaria resistance and parasite invasion dynamics.[11] Since 2019, emerging studies have extended beyond erythrocytes to extra-haematological manifestations, notably distal renal tubular acidosis and renal-metabolic associations.[12] Despite this thematic broadening, the field remains structurally anchored to Band 3–centred mechanistic paradigms, with clinically oriented and translational research streams remaining comparatively limited and weakly integrated, often limited to case series or phenotype reports.[5]Author and country collaboration networks reveal a pronounced asymmetry between research leadership and disease distribution. Investigators and institutions in high-income countries, particularly the United States and the United Kingdom, occupy central positions within global collaboration networks, despite SeAO being endemic to Southeast Asia and the Pacific. Although researchers from Thailand and Papua New Guinea contribute substantially, their work is frequently embedded within externally led collaborations. Output from other endemic countries, including Malaysia and Indonesia, remains comparatively limited, highlighting the underrepresentation of locally driven genomic and clinical research in regions bearing the highest disease burden. It should be noted that this analysis is restricted to the Scopus database and does not capture publications indexed exclusively in other databases such as PubMed, Web of Science, or regional repositories. While Scopus provides comprehensive multidisciplinary coverage, this restriction may underrepresent contributions from journals with limited international indexing, particularly those published in endemic regions.
This imbalance has significant structural and policy implications. Limited region-specific epidemiological data hinders context-appropriate screening, delays recognition of SeAO complications, particularly undiagnosed dRTA, and misinterpretation of HbA1c. Local clinical guidelines routinely adapt international frameworks that may not fully account for region-specific genetic disorders. Consequently, diabetes algorithms in Southeast Asia do not routinely recognise SeAO as a potential HbA1c confounder,[13] and paediatric and urology protocols lack explicit recommendations for SeAO-directed screening despite its local prevalence.[14]
In endemic Southeast Asian and Pacific populations, limited clinical integration of SeAO research may contribute to underdiagnosis and delayed recognition of its complications. Subclinical distal renal tubular acidosis (dRTA) may remain undetected without targeted renal evaluation in individuals presented with growth failure or nephrolithiasis, while altered erythrocyte turnover can lead to HbA1c underestimation and potential misclassification of diabetes. In malaria-endemic settings, unrecognised SeAO may confound the interpretation of haemolysis severity during acute infection.
Incorporation of SeAO into routine diagnostic considerations is essential to prevent avoidable morbidity and ensure accurate clinical assessment in affected populations. For instance, peripheral blood film examination should be considered in patients presenting with unexplained neonatal jaundice, growth failure, nephrolithiasis, or metabolic bone disease, while HbA1c measurements should be supplemented with fasting plasma glucose or oral glucose tolerance testing in individuals with SeAO. In the management of malaria-infected patients, haemolysis that appears disproportionate to the level of parasitaemia should prompt evaluation for an underlying red cell membrane disorder.
Taken together, these findings highlight the urgent need for a coordinated regional response to address the translational gap in Southeast Asian Ovalocytosis (SeAO). A concrete, immediately actionable step would be the formal inclusion of SeAO as a recognised HbA1c confounder and renal risk factor in national diabetes and paediatric clinical practice guidelines in endemic Southeast Asian countries. Such integration should mandate supplementary glucose-based testing in suspected or confirmed cases and recommend targeted renal evaluation in patients presenting with growth failure, nephrolithiasis, or unexplained metabolic acidosis. Embedding SeAO into routine diagnostic algorithms would represent a low-cost, high-impact policy intervention capable of preventing avoidable misdiagnosis, delayed recognition of complications, and inequitable standards of care in high-prevalence populations.
Acknowledgements
The authors thank the Director of Sultan Ahmad Shah Medical Centre (SASMEC) and the Department of Education and Research, SASMEC, for providing research grant support. We also acknowledge Wan Nurul Najwa and Azrini Asri for their assistance in this study.References
- Nixon
CP, Satyagraha AW, Baird GL, Harahap A, Panggalo LV, Ekawati LL,
Sutanto I, Syafruddin D, Baird J. Accurate light microscopic diagnosis
of South-East Asian ovalocytosis. Int J Lab Hematol. 2018;40:655-662. https://doi.org/10.1111/ijlh.12900
- Flatt
JF, Bruce LJ. The molecular basis for altered cation permeability in
hereditary stomatocytic human red blood cells. Front Physiol.
2018;9:367. https://doi.org/10.3389/fphys.2018.00367
- Lavinya
AA, Razali RA, Razak MA, Mohamed R, Moses EJ, Soundararajan M, Bruce
LJ, Eswaran J, Yusoff NM. Homozygous Southeast Asian ovalocytosis in
five live-born neonates. Haematologica. 2021;106:1758-1761. https://doi.org/10.3324/haematol.2020.268581
- de
Meulenaere K, Prajapati SK, Villasis E, Cuypers B, Kattenberg JH,
Kasian B, Laman M, Robinson LJ, Gamboa Vilela D, Laukens K,
Rosanas-Urgell A. Band 3–mediated Plasmodium vivax invasion is
associated with transcriptional variation in PvTRAg genes. Front Cell
Infect Microbiol. 2022;12:1011692. https://doi.org/10.3389/fcimb.2022.1011692
- Gunaratne
W, Dissanayake D, Jayaratne K, Premawardhana NP, Siribaddana S. A case
series of distal renal tubular acidosis, Southeast Asian ovalocytosis
and metabolic bone disease. BMC Nephrol. 2020;21:327. https://doi.org/10.1186/s12882-020-01959-7
- Chai
JF, Kao SL, Wang C, Jun-Yu-Lim V, Khor IW, Dou J, Podgornaia AI,
Chothani S, Cheng CY, Sabanayagam C, Wong TY, van Dam RM, Liu J, Reilly
DF, Paterson AD, Sim X. Genome-wide association for HbA1c in Malay
identified deletion on SLC4A1 that influences HbA1c independent of
glycemia. J Clin Endocrinol Metab. 2020;105:3854-3864. https://doi.org/10.1210/clinem/dgaa591
- Van
Eck NJ, Waltman L. Software survey: VOSviewer, a computer program for
bibliometric mapping. Scientometrics. 2010;84:523-538. https://doi.org/10.1007/s11192-010-0251-7
- Jarolím
P, Rubin HL, Zhai S, Sahr KE, Liu SC, Mueller TJ, Palek J. Band 3
Memphis: A widespread polymorphism with abnormal electrophoretic
mobility of erythrocyte band 3 protein caused by substitution AAG → GAG
(Lys → Glu) in codon 56. Blood. 1992;80:1592-1598. https://doi.org/10.1182/blood.V80.6.1592.1592
- Tanner MJA. The structure and function of band 3 (AE1): Recent developments. Mol Membr Biol. 1997;14:155-165. https://doi.org/10.3109/09687689709048178
- Fucharoen
G, Fucharoen S, Singsanan S, Sanchaisuriya K. Coexistence of Southeast
Asian ovalocytosis and β-thalassemia: A molecular and hematological
analysis. Am J Hematol. 2007;82:381-385. https://doi.org/10.1002/ajh.20818
- Goheen
MM, Campino S, Cerami C. The role of the red blood cell in host defence
against falciparum malaria: An expanding repertoire of evolutionary
alterations. Br J Haematol. 2017;179:543-556. https://doi.org/10.1111/bjh.14635
- Guo W, Ji P, Xie Y. Genetic diagnosis and treatment of inherited renal tubular acidosis. Kidney Dis (Basel). 2023;9:371-383. https://doi.org/10.1159/000531556
- Mirasol
R, Thai AC, Salahuddin AA, Tan K, Deerochanawong C, Mohamed M,
Saraswati MR, Sethi BK, Shah S, Soetedjo NN. A consensus of key opinion
leaders on the management of pre-diabetes in the Asia-Pacific region. J
ASEAN Fed Endocr Soc. 2017;32:6-12. https://doi.org/10.15605/jafes.032.01.02
- Skolarikos
A, Geraghty R, Somani B, Tailly T, Jung H, Neisius A, Petřík A,
Kamphuis GM, Davis N, Bezuidenhout C, Lardas M, Gambaro G, Sayer JA,
Lombardo R, Tzelves L. European Association of Urology guidelines on
the diagnosis and treatment of urolithiasis. Eur Urol. 2025;88:64-75. https://doi.org/10.1016/j.eururo.2025.03.011