Case Presentation
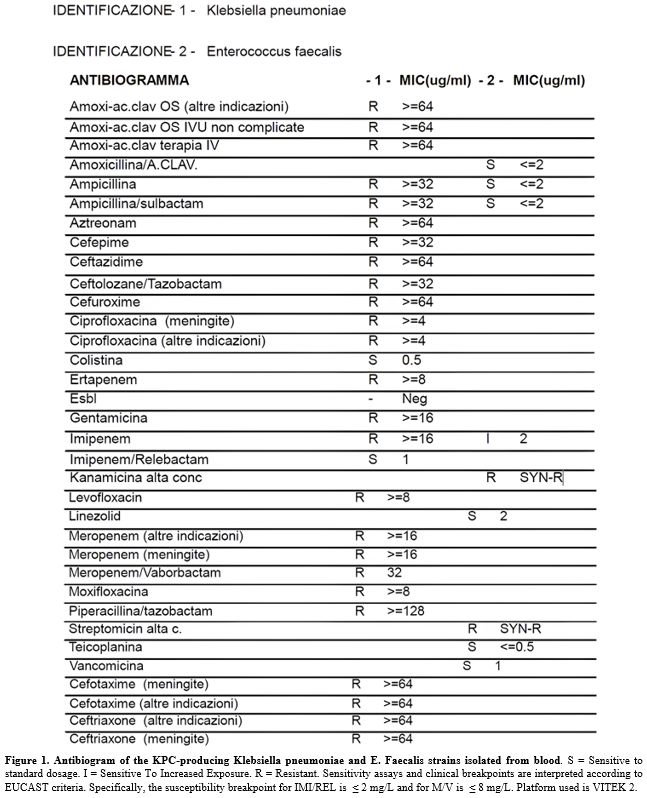
An 80-year-old male, residing in a long-term care facility with advanced Parkinson’s disease and complete loss of autonomy, presented to the Emergency Department with fever and tachycardia. His medical history was significant for chronic bed-bound status, hypertensive heart disease, multifactorial anemia, and previous episodes of sepsis. Upon admission, laboratory tests revealed marked leukocytosis (WBC 45,510/µL), elevated CRP (10.07 mg/dL), and a critical procalcitonin level (88.24 ng/mL). Chest X-ray showed an inflammatory consolidation in the left lower lobe. The patient was admitted to the Internal Medicine department and, based on preliminary blood cultures identifying Klebsiella pneumoniae, treatment with meropenem/vaborbactam and vancomycin was started.Final blood cultures results identified a polymicrobial bacteremia involving Enterococcus faecalis (susceptible to ampicillin and vancomycin) and KPC-Kp. Notably, the KPC-Kp isolate was resistant to meropenem/vaborbactam (MIC>8μg/mL) but susceptible to IMI/REL (MIC≤2μg/mL) (Figure 1).
Following the antibiogram, the regimen was switched to IMI/REL (500/500/250 mg every 6 hours). The patient experienced rapid clinical and laboratory improvement without adverse effects. Blood cultures repeated at 72 hours showed no growth. After 7 days of treatment, white blood cell count normalised (6,320/µL), and procalcitonin increased to 1.56 ng/mL. Echocardiography ruled out endocarditis, and the patient was discharged in improved clinical condition.
Discussion
To our knowledge, this is the first clinical report of KPC-producing K. pneumoniae bacteremia resistant to meropenem/vaborbactam and concurrently susceptible to imipenem/cilastatin/relebactam, with documented microbiological clearance and favorable outcome. Evidence of this divergent resistance pattern has so far been largely limited to in vitro studies and pharmacodynamic models, without in vivo confirmation.[2,4,7-9]In vitro and genomic data indicate that meropenem/vaborbactam resistance is often linked to alterations in the OmpK35 and OmpK36 porins (including the GD134-135 insertion), which restrict carbapenem uptake.[2,5-7] Relebactam appears to be less affected by these porin changes than vaborbactam, which may contribute to the divergent susceptibilities observed in some KPC-producing K. pneumoniae isolates.[5,6,9] In our case, these mechanisms were not confirmed at the molecular level and remain a hypothesis based on the literature.
Recent Italian and US surveys confirm emerging resistance to novel BL/BLI, while showing that susceptibility is not always cross-reactive.[2,6,9] Our case illustrates the successful use of IMI/REL as rescue therapy in an elderly, high-risk patient — a group often underrepresented in randomized trials.[3-4,12-13] Consistent with our findings, real-world data, including an Italian series by Gaibani et al. and larger US cohorts, report clinical success rates around 70% with IMI/REL, supporting its role even in severe bacteremia and polymicrobial infections.[10,13-15]
Conclusions
This case highlights the central role of antimicrobial stewardship in managing infections caused by multidrug-resistant organisms. Microbiology-driven susceptibility testing of novel β-lactam/β-lactamase inhibitor combinations is essential, as resistance to one agent does not imply cross-resistance. Targeted use of imipenem/cilastatin/relebactam led to clinical and microbiological cure while avoiding unnecessary therapeutic escalation, reinforcing stewardship as a key strategy to preserve last-line antibiotics and optimize patient outcomes.Ethical approval
Written informed consent for publication was obtained from the patient’s legally authorized representative in accordance with institutional policy.References
- Palomba E, Comelli A, Saluzzo F, Di Marco et al.
Activity of imipenem/relebactam against KPC-producing Klebsiella
pneumoniae and the possible role of Ompk36 mutation in determining
resistance: an Italian retrospective analysis. Ann Clin Microbiol
Antimicrob. 2025, 24(1):23. https://doi.org/10.1186/s12941-025-00792-w
- Bianco
G, Boattini M, Lupo L, Ambretti S et al. In vitro activity and genomic
characterization of KPC-producing Klebsiella pneumoniae clinical blood
culture isolates resistant to ceftazidime/avibactam,
meropenem/vaborbactam, imipenem/relebactam: an Italian nationwide
multicentre observational study (2022-23). J Antimicrob Chemother. 2025
Feb 3;80(2):583-592. https://doi.org/10.1093/jac/dkae450
- Lei
W, Duan Y, Xin M, Tian M, Xu J. Efficacy and safety of
imipenem/cilastatin/relebactam (IMI/CS/REL): a meta-analysis of
randomized controlled clinical trials. BMC Infect Dis. 2025 Sep
26;25(1):1149. https://doi.org/10.1186/s12879-025-11499-w
- Caniff
KE, Rebold N, Xhemali X, Tran N et al. Real-World Applications of
Imipenem-Cilastatin-Relebactam: Insights From a Multicenter
Observational Cohort Study. Open Forum Infect Dis. 2025 Feb
26;12(4):ofaf112. https://doi.org/10.1093/ofid/ofaf112
- Leanza
C, Mascellino MT, Volpicelli L, Covino S et al. Real-world use of
imipenem/cilastatin/relebactam for the treatment of KPC-producing
Klebsiella pneumoniae complex and difficult-to-treat resistance (DTR)
Pseudomonas aeruginosa infections: a single-center preliminary
experience. Front Microbiol. 2024 Jul 16;15:1432296. https://doi.org/10.3389/fmicb.2024.1432296
- Rogers
TM, Kline EG, Griffith MP, Jones CE, et al. Impact of ompk36 genotype
and KPC subtype on the in vitro activity of ceftazidime/avibactam,
imipenem/relebactam and meropenem/vaborbactam against KPC-producing K.
pneumoniae clinical isolates. JAC Antimicrob Resist. 2023 Mar
23;5(2):dlad022. https://doi.org/10.1093/jacamr/dlad022
- Gaibani
P, Lombardo D, Bussini L, Bovo F, et al. Epidemiology of
Meropenem/Vaborbactam Resistance in KPC-Producing Klebsiella pneumoniae
Causing Bloodstream Infections in Northern Italy, 2018. Antibiotics
(Basel). 2021 May 6;10(5):536. https://doi.org/10.3390/antibiotics10050536
- Lombardo
D, Ambretti S, Lazzarotto T, Gaibani P. In vitro activity of
imipenem-relebactam against KPC-producing Klebsiella pneumoniae
resistant to ceftazidime-avibactam and/or meropenem-vaborbactam. Clin
Microbiol Infect. 2022 May;28(5):749-751. https://doi.org/10.1016/j.cmi.2022.01.025
- Karaiskos
I, Galani I, Daikos GL, Giamarellou H. Breaking Through Resistance: A
Comparative Review of New Beta-Lactamase Inhibitors (Avibactam,
Vaborbactam, Relebactam) Against Multidrug-Resistant Superbugs.
Antibiotics (Basel). 2025 May 21;14(5):528. https://doi.org/10.3390/antibiotics14050528
- Heo YA. Imipenem/Cilastatin/Relebactam: A Review in Gram-Negative Bacterial Infections. Drugs. 2021 Feb;81(3):377-388. https://doi.org/10.1007/s40265-021-01471-8
- Motsch
J, Murta de Oliveira C, Stus V, Köksal I, et al. RESTORE-IMI 1: A
Multicenter, Randomized, Double-blind Trial Comparing Efficacy and
Safety of Imipenem/Relebactam vs Colistin Plus Imipenem in Patients
With Imipenem-nonsusceptible Bacterial Infections. Clin Infect Dis.
2020 Apr 15;70(9):1799-1808. https://doi.org/10.1093/cid/ciz530
- Motsch
J, Murta de Oliveira C, Stus V, Köksal I, et al A. RESTORE-IMI 1: A
Multicenter, Randomized, Double-blind Trial Comparing Efficacy and
Safety of Imipenem/Relebactam vs Colistin Plus Imipenem in Patients
With Imipenem-nonsusceptible Bacterial Infections. Clin Infect Dis.
2020 Apr 15;70(9):1799-1808. https://doi.org/10.1093/cid/ciz530
- Shields
RK, Yücel E, Turzhitsky V, Merchant S, Min JS, Watanabe AH. Real-world
evaluation of imipenem/cilastatin/relebactam across US medical centres.
J Glob Antimicrob Resist. 2024 Jun;37:190-194. https://doi.org/10.1016/j.jgar.2024.03.002
- Gaibani
P, Bussini L, Amadesi S, Bartoletti M, et al. Successful Treatment of
Bloodstream Infection due to a KPC-Producing Klebsiella Pneumoniae
Resistant to Imipenem/Relebactam in a Hematological Patient.
Microorganisms. 2022 Apr 5;10(4):778. https://doi.org/10.3390/microorganisms10040778
- Istituto Superiore di Sanità(ISS). Antibiotico-resistenza per Klebsiella pneumoniae, Italia 2024. AR-ISS Report. 2024. https://www.epicentro.iss.it/antibiotico-resistenza/ar-iss-rapporto-klebsiella-pneumoniae