CD26.[4-5] Recently, Huang et al. reported an increased proportion of neutrophils expressing CD38 and bright CD56 (CD56briCD38+) in bone marrow (BM) samples of CML patients. They found that the CD56briCD38+ neutrophil subset, exceeding 2.0% of total neutrophils, may serve as a highly sensitive and specific flow cytometric marker for identifying CML cases.[6] Hence, in this study, we performed a comprehensive analysis of multiparametric flow cytometry (MFC) data from suspected CML cases, focusing on the expression patterns of CD56 and CD38 on PB neutrophils. This approach aimed to validate the findings of Huang et al. and to explore whether evaluating CD56briCD38+ neutrophil subsets in PB could serve as a simple, less invasive flow cytometric signature for early CML detection.
PB cells were stained using a combination of monoclonal antibodies that included CD13/CD33/CD11b/CD15/CD10/CD16/CD56/CD38/CD34/CD26/CD45/CD117/HLA-DR. Data on standardized 12 color staining combinations were acquired on BD FACS Lyric flow cytometer (Becton Dickinson) and analyzed using the PAINT-A-GATE and FACSDIVA software (Becton Dickinson). A comprehensive and sequential MFC gating strategy was employed to ensure accurate neutrophil identification. Neutrophils were broadly identified by their characteristic CD45/SSC-A distribution, and this population was further refined by verifying the co-expression of CD33 and the presence of mature granulocytic markers, specifically CD10 and CD16.[6-7] The “neutrophil-gated” population refers to this subset of events identified as mature neutrophils based on their CD45/CD33/CD10/CD16 expression and side-scatter properties, while excluding other leukocyte subsets. The percentage of CD56briCD38+ neutrophils was calculated within the gated neutrophil population to allow comparison with previously published approaches,[6] as well as relative to total CD45+ WBCs, to assess reproducibility using an easier approach. To address potential inconsistencies and coefficient variation, a high number of events (median 50,000 CD45+ cells) was acquired for each sample. Furthermore, to mitigate inter-operator variability, MFC data were independently reviewed and validated by two expert flow cytometrists. Further flow cytometry methods are detailed in the supplementary data. Molecular detection of the BCR::ABL1 transcript was performed by PCR amplification following the primer and protocol recommendations of the BIOMED-1 Concerted Action, with the resulting amplification bands visualized by agarose gel electrophoresis.[8]
This single-center study analyzed MFC data from 41 samples. Among them, 31 were consecutively collected from patients presenting neutrophilic leukocytosis with or without circulating intermediate granulocytic precursors; 17 were later confirmed as newly diagnosed CML cases, while 14 were diagnosed with other hematological disorders (non-CML group). The non-CML cases included patients later diagnosed with polycythemia vera (n=2, 14.3%), atypical CML (MDS/MPN with neutrophilia) (n=2, 14.3%), myelofibrosis (n=1, 7.1%), acute myeloid leukemia (n=1, 7.1%), myelodysplastic syndrome (n=1, 7.1%), chronic myelomonocytic leukemia (n=1, 7.1%), and reactive leukocytosis (n=6, 42.9%). An additional 10 samples from healthy donors served as controls (HD-Co).
Patient and donor characteristics are summarized in Table 1. The median age was 62.0 years (IQR, 44.5–74.0), and 23 patients (56.1%) were male. As expected, CML patients exhibited a significantly higher median absolute WBC count at the time of sampling (p<0.001), along with higher median absolute neutrophil count (p<0.001), eosinophil count (p=0.001), and basophil count (p=0.001).
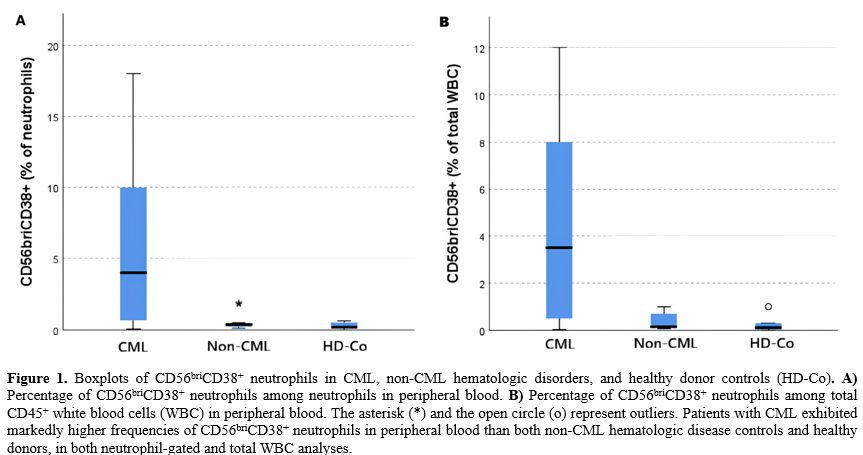
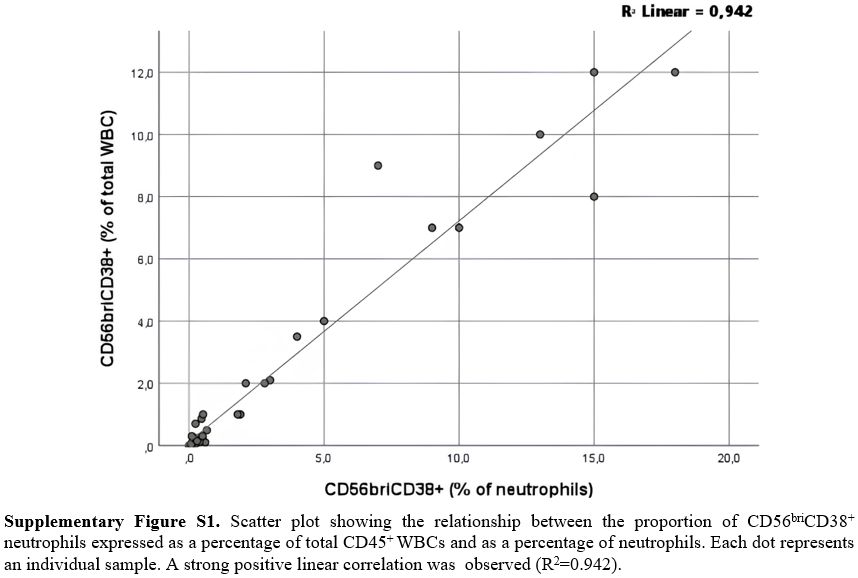
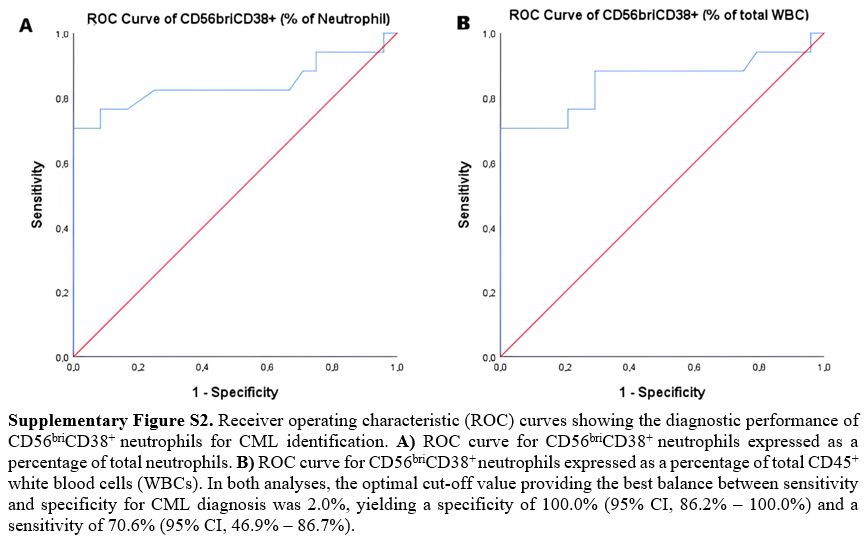
The subset of neutrophil-gated CD56briCD38+ neutrophils was markedly elevated in CML patients compared to both the non-CML group and healthy donors (HD-Co), in which it was present at minimal levels (p<0.001) (Table 1). Within the PB neutrophil compartment, the median percentage of CD56briCD38+ neutrophils was 4.0% (IQR, 0.6–11.5%) in CML cases, 0.4% (IQR, 0.2–0.5%) in the non-CML group, and 0.2% (IQR, 0.1–0.5%) in the HD-Co (p=0.001) (Figure 1A). This difference remained significant even when CD56briCD38+ neutrophils were expressed as a percentage of total WBCs (rather than within the neutrophil gate) (p<0.001) (Figure 1B) or as an absolute CD56briCD38+ neutrophil count (p<0.001) (Table 1). In fact, as expected, a strong positive linear correlation was observed between the proportion of CD56briCD38+ neutrophils calculated relative to total CD45+ positive cells and that relative to the selected neutrophil population (R²=0.942) (Supplementary Figure S1). Receiver operating characteristic (ROC) analysis was performed to evaluate the diagnostic performance of CD56briCD38+ neutrophils for CML prediction. The area under the ROC curve (AUC) for the percentage of CD56briCD38+ neutrophils within the neutrophil gate was 0.842 (95% CI, 0.693–0.991) (p<0.001) (Supplementary Figure S2A). A similar AUC value of 0.851 (95% CI, 0.712–0.992) (p<0.001) was obtained when CD56briCD38+ neutrophils were expressed as a percentage of total WBCs (Supplementary Figure S2B). The optimal cut-off value providing the best balance between sensitivity and specificity for CML diagnosis was 2.0% for both parameters. At this threshold, specificity and sensitivity were 100.0% (95% CI, 86.2% – 100.0%) and 70.6% (95% CI, 46.9% – 86.7%), respectively, in both cases. Even after adjusting for total white blood cell count, the percentage of CD56briCD38+ neutrophils remained a significant independent predictor of CML, whether expressed relative to the neutrophil population or to total WBCs. Specifically, the percentage of CD56briCD38+ neutrophil was associated with an odds ratio (OR) of 2.92 (95% CI, 1.06–8.03) (p=0.038) when expressed as a proportion of neutrophils, and a OR of 4.80 (95% CI, 1.10–20.97) (p=0.037) when expressed as a proportion of total WBCs.
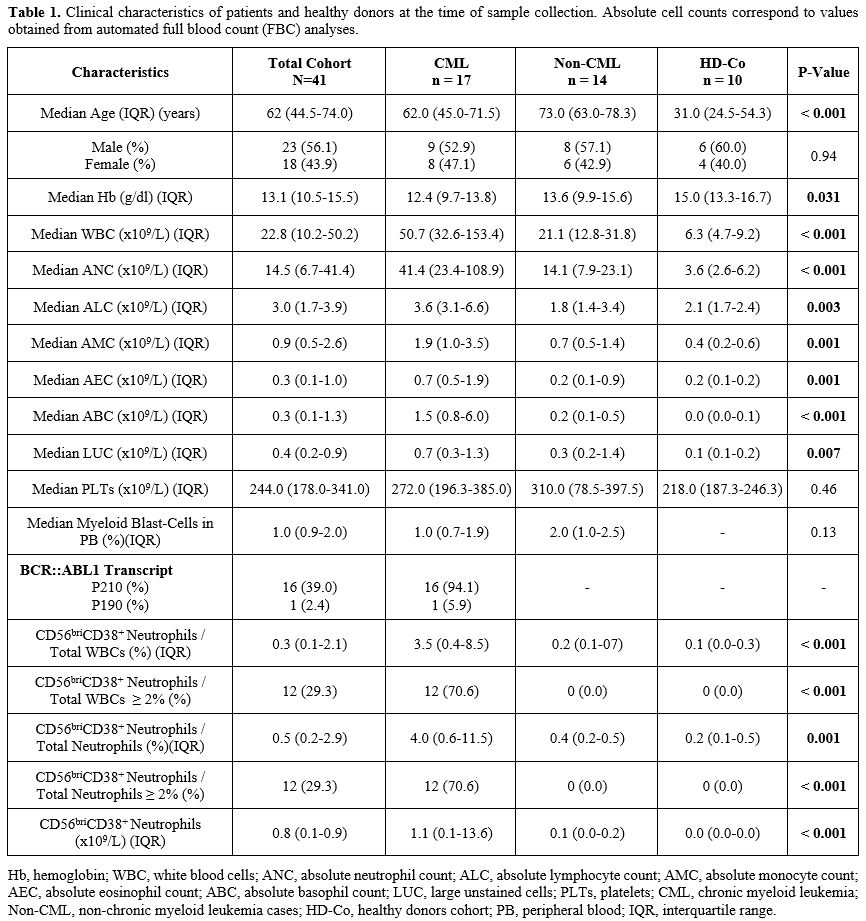 |
Table 1. Clinical characteristics of patients and healthy donors at the time of sample collection. Absolute cell counts correspond to values obtained from automated full blood count (FBC) analyses. |
Notably, in all CML cases, the CD34+/CD38− stem cells in PB, although present at low frequency [median 1.0% (IQR, 0.7-1.9)], expressed CD26, whereas stem cells from the non-CML group lacked CD26 expression.
CD56 is a membrane glycoprotein involved in homotypic adhesion, while CD38 is a cell surface glycoprotein with enzymatic activity in the synthesis and degradation of cyclic ADP-ribose. Aberrant expression of CD56 and co-expression of CD56 and CD38 have been reported in various hematological malignancies, such as abnormal plasma cells in multiple myeloma.[9] As mentioned above, Huang et al. highlighted a distinct increase in CD56briCD38+ neutrophils specifically in CML patients, a feature rarely observed in other myeloproliferative neoplasms.[6] We confirmed the findings of Huang et al. and demonstrated that this peculiar flow cytometric signature, the presence of CD56briCD38+ neutrophil subset, can also be identified in PB, enabling easier and reliable detection, with the neutrophil-gated 2.0% cutoff that appears applicable to PB specimens as well, promoting an easier approach in clinical practice. Notably, this threshold seems also to be reliably applicable as a percentage of total CD45+ WBCs, given the strong positive linear correlation between the proportion of CD56briCD38+ neutrophils calculated relative to total WBCs and that calculated relative to total neutrophils, making its usage more practical in routine analyses. Moreover, we confirmed previous reports that the CD34+/CD38−/CD26+ LSCs population in PB represents a hallmark of CML, even when present at low frequency.[4-5]
This flow cytometric approach should be considered as a supportive screening tool to prioritize gold-standard molecular or cytogenetic assays for CML diagnosis. Its clinical utility may lie in providing a rapid diagnostic suspicion, particularly when cytomorphological evaluation is inconclusive or lacks the classical features of CML, or when molecular results might be immediately unavailable or excessively delayed. In these scenarios, identifying this signature can effectively prioritize testing in patients with unexplained neutrophilic leukocytosis and ambiguous morphological profiles. This study presents several limitations, including the small size of the CML cohort and its single-center nature. Moreover, while the heterogeneity of the non-CML comparator group reflects real-world clinical practice, this cytometric approach requires further validation in larger, multicenter external datasets to fully characterize its diagnostic performance and scalability. Nevertheless, our data successfully validate the findings previously reported by Huang et al.[6] in a different cohort, confirming that the flow cytometric identification of CD56briCD38+ neutrophil subsets in the PB may represent a rapid and cost-effective screening tool to raise suspicion of CML and to prompt timely confirmatory molecular assays for BCR::ABL1 transcript detection and/or cytogenetic testing for t(9;22) identification.
Conflicts of Interest
Massimo Breccia received honoraria from Novartis, Incyte, Pfizer, BMS, AOP, Abbvie, GSK. Maurizio Martelli received honoraria from Roche, Gilead Sciences, Novartis, Abbvie, Incyte, BeiGene, Takeda, and Bristol Myers Squibb/Celgene. The other authors declare no conflict of interest.Author Contributions
Alessandro Laganà and Matteo Breccia: Writing the original draft, data collection and interpretation, and formal analysis. Loredana Elia: Molecular data collection, interpretation, and contribution to lab work. Emilia Scalzulli and Maria Laura Bisegna: Followed the patients. Claudia Ielo: Molecular data collection and contribution to lab work. Sonia Buffolino and Attilio Di Lascio: Molecular data analysis. Concetta Anna Germano, Stefania Intoppa, and Maria Laura Milani: Flow cytometry data collection, acquisition, and interpretation. Maurizio Martelli and Massimo Breccia: Supervision and manuscript editing. Maria Stefania De Propris: Data collection and interpretation, conceptualization, investigation, and writing the original draft.Data Availability Statement
The data that support the findings of this study are available in the text and from the corresponding author, Massimo Breccia, upon reasonable request.Informed Consent
Written informed consent was collected according to local practice.Acknowledgments
Concetta Anna Germano, Stefania Intoppa, Attilio Di Lascio and Maria Laura Milani were supported by ROMAIL ONLUS.Ethics Approval Statement
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. As a non-interventional study using data collected as part of routine diagnostic procedures, in accordance with local regulations, it did not require ethics committee approval.References
- Khoury JD, Solary E, Abla O, et al. The 5th edition
of the World Health Organization Classification of Haematolymphoid
Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia.
2022;36(7):1703-1719. https://doi.org/10.1038/s41375-022-01613-1
- Jabbour
E, Kantarjian H. Chronic myeloid leukemia: 2025 update on diagnosis,
therapy, and monitoring. Am J Hematol. 2024;99(11):2191-2212. https://doi.org/10.1002/ajh.27443
- Lanza
F, Bi S, Castoldi G, Goldman JM. Abnormal expression of N-CAM (CD56)
adhesion molecule on myeloid and progenitor cells from chronic myeloid
leukemia. Leukemia. 1993;7(10):1570-1575.
- Raspadori
D, Pacelli P, Sicuranza A, et al. Flow Cytometry Assessment of CD26+
Leukemic Stem Cells in Peripheral Blood: A Simple and Rapid New
Diagnostic Tool for Chronic Myeloid Leukemia. Cytometry B Clin Cytom.
2019;96(4):294-299. https://doi.org/10.1002/cyto.b.21764
- Sharma
P, Sachdeva MUS, Naseem S, et al. Identification of peripheral blood
CD26+ leukemic stem cells has a potential role in the rapid diagnosis
of chronic myeloid leukemia. Int J Lab Hematol. 2022;44(3):518-523. https://doi.org/10.1111/ijlh.13807
- Huang
P, Zhang C, Zhang A, et al. CD56briCD38+ as a novel neutrophil-specific
marker in chronic myeloid leukemia. Heliyon. 2024;10(21):e39465. https://doi.org/10.1016/j.heliyon.2024.e39465
- Wood B. Multicolor immunophenotyping: human immune system hematopoiesis. Methods Cell Biol. 2004;75:559-576. https://doi.org/10.1016/s0091-679x(04)75023-2
- Van
Dongen JJ, Macintyre EA, Gabert JA, et al. Standardized RT-PCR analysis
of fusion gene transcripts from chromosome aberrations in acute
leukemia for detection of minimal residual disease. Report of the
BIOMED-1 Concerted Action: investigation of minimal residual disease in
acute leukemia. Leukemia. 1999;13(12):1901-1928. https://doi.org/10.1038/sj.leu.2401592
- Cottini
F, Rodriguez J, Hughes T, et al. Redefining CD56 as a Biomarker and
Therapeutic Target in Multiple Myeloma. Mol Cancer Res.
2022;20(7):1083-1095. https://doi.org/10.1158/1541-7786.MCR-21-0828
Supplementary Data
Flow Cytometry Supplementary Method
Peripheral blood (PB) samples were stained within 24 hours of collection, before any treatment. Total leukocytes were incubated with an appropriate volume of monoclonal antibodies (mAbs) directed against CD13/CD33/CD11b/CD15/CD10/CD16/CD56/CD38/CD34/CD26/CD45/CD117/HLA-DR (Società Italiana Chimici, SIC, Life Sciences, Rome, Italy; Beckman Coulter, Brea, CA). Data from standardized 12-color staining combinations were acquired on FACSCanto II or BD FACS Lyric flow cytometers (Becton Dickinson) and analyzed using PAINT-A-GATE and FACSDIVA software (Becton Dickinson). Instrument performance on the BD FACS Lyric™ was monitored daily using BD™ Cytometer Setup and Tracking (CS&T) beads to maintain stable photomultiplier tube voltages and consistent fluorescence sensitivity. For each sample, at least 50.000 CD45+ events were acquired to ensure a high signal-to-noise ratio and precise quantification of rare populations. As mentioned in the manuscript, a comprehensive, sequential MFC gating strategy was employed to ensure accurate neutrophil identification. Within the neutrophil population, the subset of CD56briCD38+ neutrophils was identified by CD38 positivity and the intensity of CD56 expression. In particular, identification of CD56bri neutrophils was based on CD56 Mean Fluorescence Intensity (MFI), using internal biological references within the same sample as comparison. Mature, non-aberrant neutrophils served as the negative control, and NK cells (CD45+/SSClow/CD56+) provided the positive reference for CD56 "bright" staining intensity. Specifically, neutrophils were classified as CD56bri when their CD56 MFI was at least 1 log decade higher than that of the negative neutrophil population and similar to that of the NK-cell subset. Finally, all processed data were independently cross-validated by two expert flow cytometrists to mitigate inter-operator variability and ensure the scalability of this diagnostic tool in different clinical settings.
Supplementary Figures