This single-center, retrospective observational study included patients diagnosed with Philadelphia chromosome-negative MPNs treated with ruxolitinib. The variables included demographic characteristics, MPN subtype (essential thrombocythemia, polycythemia vera, primary or secondary myelofibrosis), ruxolitinib dose and duration, screening results for latent tuberculosis infection (IGRA and/or purified protein derivative [PPD] testing), isoniazid (INH) and antiviral prophylaxis status, concomitant cytoreductive or immunosuppressive therapy, and additional immunosuppressive conditions. The laboratory parameters recorded at the last visit included absolute neutrophil count (ANC), absolute lymphocyte count (ALC), and serum immunoglobulin G (IgG) levels. Neutropenia was defined as an ANC <0.5 ×10⁹/L, lymphopenia as an ALC <1.0×10⁹/L, and hypogammaglobulinemia as a serum IgG level <5 g/L. Latent tuberculosis screening and INH prophylaxis were performed according to an institutional protocol. The decision to initiate antiviral prophylaxis was made at the treating physician's discretion. The infectious outcomes included herpes zoster, recurrent oral herpes, and tuberculosis reactivation during ruxolitinib therapy. Treatment response was evaluated in patients who had completed at least six months of ruxolitinib therapy. Response was defined as achievement of target blood count parameters in patients with polycythemia vera or essential thrombocythemia, and as improvement in constitutional symptoms or at least a 50% reduction in palpable spleen size below the costal margin in patients with myelofibrosis.
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 20.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics are presented as medians with minimum–maximum values for non-normally distributed and ordinal variables. Univariate analyses were conducted using the Chi-square, Fisher’s exact, and Mann-Whitney U tests, where appropriate. Given the very small number of infectious events, statistical analyses were performed for descriptive and hypothesis-generating purposes only.
This study was conducted in accordance with the ethical standards of the World Medical Association Declaration of Helsinki. Ethical approval was obtained from the Institutional Review Board of Ankara Bilkent City Hospital (approval number: TABED 1-26-2314; approval date: 11.03.2026). Given the retrospective, observational design of this study, informed consent was waived.
A total of 30 patients were included. The median age at diagnosis was 63 years (range, 29–84), and the cohort comprised 18 males and 12 females. The ruxolitinib dose ranged from 5 to 50 mg daily. Eleven patients (36.7%) had primary myelofibrosis, twelve (40.0%) secondary myelofibrosis, and seven (23.3%) polycythemia vera. The median duration of therapy was 20.1 months (range, 2.2–82.6).
No patients were receiving additional immunosuppressive therapy other than ruxolitinib. However, seven patients (23.3%) were receiving concomitant cytoreductive therapy (anagrelide and/or hydroxyurea), and one had a history of splenectomy. All patients were alive at the time of analysis.
At last follow-up, the median ANC was 4.2×10⁹/L (range, 1.4–21.0), and no patient had neutropenia. The median ALC was 1.3×10⁹/L (range, 0.49–7.5), with lymphopenia (<1.0×10⁹/L) in 12 patients. No hypogammaglobulinemia was detected, and the median serum IgG level was 11.2 g/L (range, 6.5–29.1 g/L).
Tuberculosis screening and prevention: Latent tuberculosis screening was performed in 29 of 30 patients. IGRA was the preferred screening method when available and was performed in 27 patients. In situations where IGRA was not readily accessible, screening was performed using PPD; two patients were evaluated with PPD alone. In some cases, both IGRA and PPD were available. One patient had no documented screening; the reason for its absence could not be determined from the medical records. A PPD induration >5 mm was considered positive according to institutional criteria. Information regarding prior tuberculosis exposure, detailed radiologic history, or BCG vaccination status was not consistently available in the retrospective records and could not be systematically evaluated.
IGRA was positive in 2 patients and negative in the remaining individuals tested. Among patients evaluated with PPD, indurations of 8 mm, 10 mm, and 11 mm were considered positive. Isoniazid prophylaxis was initiated in five patients: two due to IGRA positivity, one due to suspicious findings on chest radiography in combination with a PPD induration of 8 mm, and two due to PPD indurations of 10 mm and 11 mm, respectively. No tuberculosis reactivation was observed during follow-up.
Herpes virus infections and antiviral strategies: Antiviral prophylaxis was implemented within routine clinical practice and was not guided by a predefined institutional protocol. Overall, 17 patients (56.6%) received antiviral prophylaxis. Oral acyclovir was prescribed to 15 patients (50.0%) at daily doses of 200–400 mg, and oral valacyclovir to 2 patients (6.6%) at doses of 500 mg every other day or once daily. Dose adjustments were made according to creatinine clearance. None of the patients had received recombinant zoster vaccination prior to or during ruxolitinib therapy.
Four patients had a history of herpes zoster before ruxolitinib exposure. During treatment, one patient developed herpes zoster, and two experienced recurrent oral herpes. Treatment response to ruxolitinib could be evaluated in 26 patients. Both patients with a history of recurrent oral herpes responded to ruxolitinib. Among patients without recurrent oral herpes, 19 of 24 (79%) responded to ruxolitinib. No formal statistical comparison was made because of the limited number of cases. The patient who developed herpes zoster — a 63-year-old female — had not received antiviral prophylaxis before infection onset; secondary prophylaxis was initiated thereafter. Potential risk factors for recurrent oral herpes were explored (Table 1). No statistically significant associations were identified. Given the observed variability in prophylaxis use, we explored potential clinical factors associated with the decision to initiate antiviral prophylaxis (Table 2). No statistically significant associations were identified; however, these exploratory analyses are limited by the small sample size and number of events.
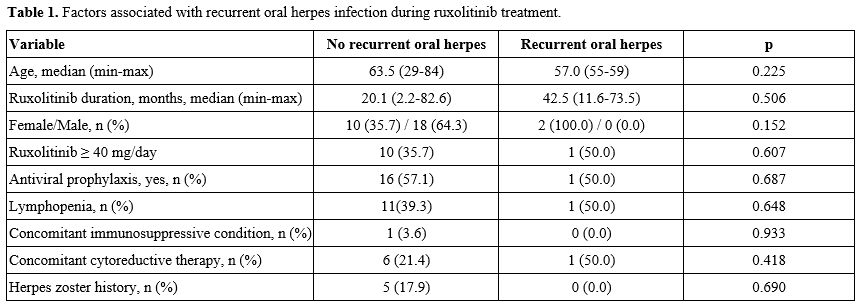 |
Table 1. Factors associated with recurrent oral herpes infection during ruxolitinib treatment. |
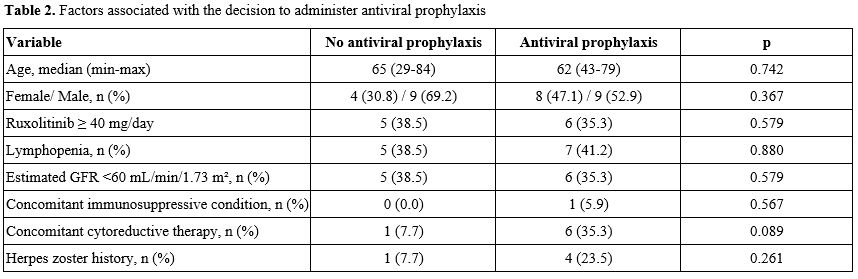 |
Table 2. Factors associated with the decision to administer antiviral prophylaxis. |
This study provides a descriptive real-world account of herpetic and tuberculosis-related infections in patients with Philadelphia chromosome-negative MPNs treated with ruxolitinib. Given the small cohort size and absence of a control group, the present findings are observational and do not permit inference regarding the effectiveness of screening or prophylactic strategies.
In pivotal trials, grade 3–4 neutropenia was reported, whereas lymphopenia and antibody deficiency were not systematically evaluated.[8] However, Ruxolitinib has been used as an immunosuppressor for graft-versus-host disease.[9] In our cohort, lymphopenia was observed, while no cases of hypogammaglobulinemia were detected.
Nearly all patients underwent latent tuberculosis screening with IGRA and/or PPD, and INH prophylaxis was administered according to screening results. At our institution, INH is prescribed for 6–9 months, with ruxolitinib initiation delayed until at least one month of therapy has been completed, following approaches extrapolated from anti–tumor necrosis factor practice.[10] No tuberculosis reactivation was observed during follow-up; however, this finding should be interpreted with caution, given the limited sample size and the absence of a control group. Given variability in screening practices across centers, we report our single-center experience using a uniform protocol.
Herpes zoster was documented in several patients prior to ruxolitinib exposure, and one case occurred during treatment. Real-world studies have reported herpes zoster rates of 12–19% in ruxolitinib-treated patients,[11,12] although prophylactic strategies were not systematically evaluated.[12] In our cohort, antiviral prophylaxis was not uniformly administered, and no clear clinical determinants for its prescription were identified. These findings should be interpreted cautiously, given the limited number of events.
Expert recommendations emphasize educating patients receiving ruxolitinib about the signs and symptoms of herpes zoster to facilitate early medical evaluation.[1,14] We did not identify comparative data to support preferential use of oral acyclovir over valacyclovir for prophylaxis. Recombinant adjuvant zoster vaccination has also been evaluated in ruxolitinib-treated patients with MF and PV, with immunogenic responses reported to be comparable to those of controls.[15]
This study is limited by its retrospective design, small sample size, low number of infectious events, and absence of a comparator group. It therefore does not allow conclusions regarding the effectiveness of screening or prophylactic strategies. Nevertheless, it provides a descriptive real-world account of institutional screening and management practices.
In summary, we report a single-center real-world experience of infectious outcomes and clinical management approaches in ruxolitinib-treated MPN patients. Larger prospective studies are needed to better characterize infectious risks and to evaluate screening and preventive strategies in this setting.
References
- Butler, L.A., C. Forsyth, C. Harrison, and
A.C. Perkins, Real World Management of Cytopenias and Infections in
Patients With Myelofibrosis Treated With Ruxolitinib. E J Haem, 2025.
6(2): p. e70007. https://doi.org/10.1002/jha2.70007 PMid:40123795 PMCid:PMC11927021
- Chen, C.Y. and T.C. Chen, Ruxolitinib associated
psoas muscle tuberculosis abscess in a primary myelofibrosis woman: A
case report and literature review. Medicine (Baltimore), 2024. 103(14):
p. e37653. https://doi.org/10.1097/MD.0000000000037653 PMid:38579059 PMCid:PMC10994542
- Luo, Q., Z. Xiao, and L. Peng, Effects of
ruxolitinib on infection in patients with myeloproliferative neoplasm:
a meta-analysis. Hematology, 2021. 26(1): p. 663-669. https://doi.org/10.1080/16078454.2021.1967256 PMid:34493151
- Lussana, F., M. Cattaneo, A. Rambaldi, and A.
Squizzato, Ruxolitinib-associated infections: A systematic review and
meta-analysis. Am J Hematol, 2018. 93(3): p. 339-347. https://doi.org/10.1002/ajh.24976 PMid:29150886
- Hirai, N., K. Kasahara, S. Yoshihara, T. Nishimura,
K. Omori, Y. Ogawa, T. Ogawa, N. Hishiya, Y. Suzuki, H. Yano, M.
Yoshikawa, and K. Mikasa, Necessity to screen and treat latent
tuberculosis before ruxolitinib treatment-Ruxolitinib-associated
disseminated tuberculosis: A case report and literature review.
IDCases, 2020. 21: p. e00892. https://doi.org/10.1016/j.idcr.2020.e00892 PMid:32642438 PMCid:PMC7332526
- Khalid, F., M. Damlaj, M. AlZahrani, K.A.
Abuelgasim, and G.E. Gmati, Reactivation of tuberculosis following
ruxolitinib therapy for primary myelofibrosis: Case series and
literature review. Hematol Oncol Stem Cell Ther, 2021. 14(3): p.
252-256. https://doi.org/10.1016/j.hemonc.2020.02.003 PMid:32201152
- Peng, Y., L. Meng, X. Hu, Z. Han, and Z. Hong,
Tuberculosis in Patients with Primary Myelofibrosis During Ruxolitinib
Therapy: Case Series and Literature Review. Infect Drug Resist, 2020.
13: p. 3309-3316. https://doi.org/10.2147/IDR.S267997 PMid:33061478 PMCid:PMC7532060
- Öztürk F., Pepeler M.S., Ozhamam E.U., Korkmaz
G.Successful management of steroid-resistant cutaneous acute
graft-versus-host disease,toxic epidermal necrolysis-like,with
ruxolitinib. MediterrJHematolInfectDis2025, 17(1):e2025020 https://doi.org/10.4084/MJHID.2025.020 PMid:40084094 PMCid:PMC11906136
- Manduzio, P., Ruxolitinib in myelofibrosis: to be or not to be an immune disruptor. Ther Clin Risk Manag, 2017. 13: p. 169-177. https://doi.org/10.2147/TCRM.S121683 PMid:28243106 PMCid:PMC5315213
- Duarte, R., S. Campainha, J. Cotter, B. Rosa, P.
Varela, A. Correia, H. Canhão, and J.E. Fonseca, Position paper on
tuberculosis screening in patients with immune mediated inflammatory
diseases who are candidates for biological therapy. GE Jornal Português
de Gastrenterologia, 2012. 19(6): p. 290-299. https://doi.org/10.1016/j.jpg.2012.09.004
- Blanco-Sánchez, A., R. Ayala, G.
Carreño-Tarragona, R. Colmenares, N. López-Muñoz, A. Sáez, M.L.
Palacios-Berraquero, J. Hernández, and J. Martínez-López, Long-Term
Safety Profile of Ruxolitinib in Chronic Myeloproliferative Neoplasms:
A Comprehensive Real-World Analysis. EJHaem, 2025. 6(6): p. e70152. https://doi.org/10.1002/jha2.70152 PMid:41169484 PMCid:PMC12570962
- Coltro, G., E. Sant'Antonio, G.A. Palumbo, F.
Mannelli, V. De Stefano, M. Ruggeri, E.M. Elli, R. Zanotti, O. Borsani,
I. Bertozzi, A. Duminuco, S. Betti, G. Carli, F. Cavalca, I. Tanasi, E.
Rumi, M.L. Randi, B. Garibaldi, G.G. Loscocco, P. Guglielmelli, and
A.M. Vannucchi, Assessment of the efficacy and tolerability of
ruxolitinib for the treatment of myelofibrosis patients in a real-life
setting: An Italian MYNERVA Project. Cancer Med, 2023. 12(7): p.
8166-8171. https://doi.org/10.1002/cam4.5618 PMid:36708083 PMCid:PMC10134270
- Te Linde, E., L.J.E. Boots, L.G.M. Daenen, M.A. de
Witte, and A.H.W. Bruns, High Incidence of Herpes Zoster in Patients
Using Ruxolitinib for Myeloproliferative Neoplasms: Need for
Prophylaxis. Hemasphere, 2022. 6(11): p. e793. https://doi.org/10.1097/HS9.0000000000000793 PMid:36325270 PMCid:PMC9619234
- Sadjadian, P., K. Wille, and M. Griesshammer,
Ruxolitinib-Associated Infections in Polycythemia Vera: Review of the
Literature, Clinical Significance, and Recommendations. Cancers
(Basel), 2020. 12(11). https://doi.org/10.3390/cancers12113132 PMid:33114733 PMCid:PMC7693745
- Astibia-Mahillo, B., V. Leguizamon, E. Mateos, M.
Torres, N. Armenteros, M.C. Luengo-Martínez, A. Fernández-Chávez, N.
Daza, M. Piris-Villaespesa, J. Lopez Jimenez, M. Coiras, and V. García
Gutiérrez, The recombinant adjuvant zoster vaccine induces similar
immune responses in ruxolitinib-treated patients with myelofibrosis and
polycythemia vera. Blood, 2025. 146: p. 7273. https://doi.org/10.1182/blood-2025-7273