We included patients aged ≥18 years with untreated AML who received venetoclax combination therapy as initial treatment at Anjo Kosei Hospital between April 2021 and April 2023. Cytogenetic risk was assessed according to the European LeukemiaNet 2017 criteria.[1] Treatment response was classified as complete remission (CR), CR with incomplete hematologic recovery (CRi), MLFS, residual disease (RD), or morphologic relapse (MR).[1] For cases with multiple treatment assessments between cycles, the evaluation closest to the subsequent cycle was used. Overall survival (OS) was calculated from venetoclax initiation; for response-stratified analyses, OS and treatment-related mortality (TRM) were calculated from the date of bone marrow response assessment after cycle 1 or 2. TRM was defined as death unrelated to AML progression during venetoclax combination therapy.[2] Survival curves were estimated using the Kaplan-Meier method, and the cumulative incidence of relapse and TRM was assessed descriptively. Given the small sample size, single-center design, and limited number of events, the present analysis should be interpreted as exploratory. In particular, early death before response assessment introduces the potential for immortal-time and selection bias, and measurable residual disease (MRD) was not available.
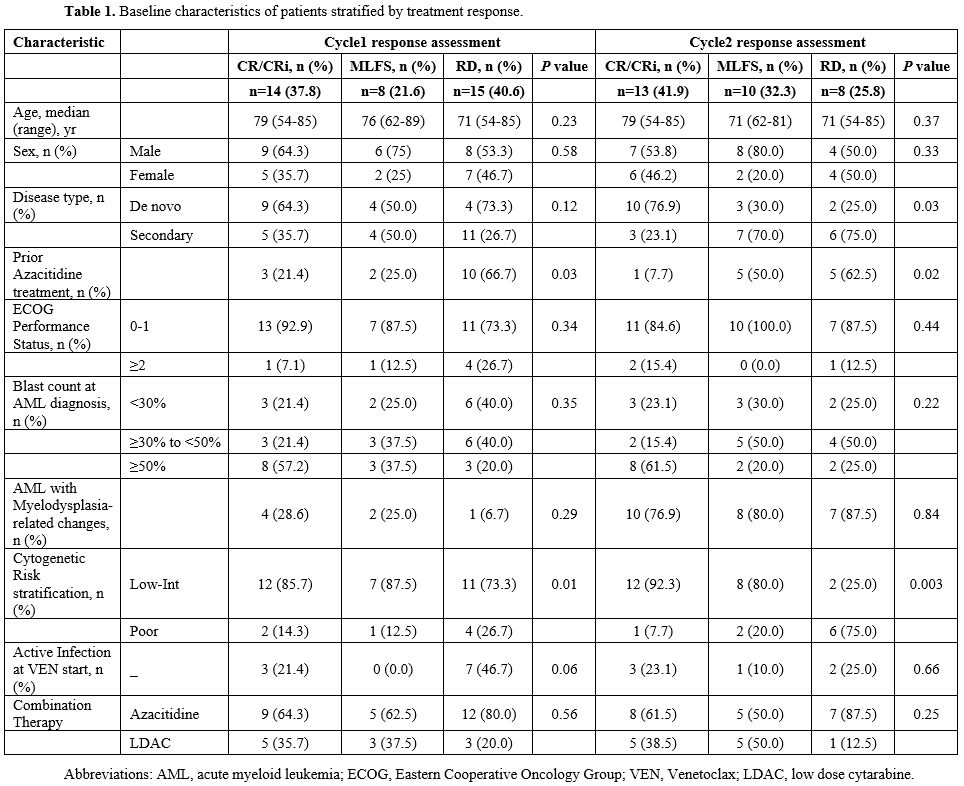
Among 44 treated patients, 7 died during cycle 1, and responses after cycle 1 were therefore evaluable in 37 patients. Of these, 14 (37.8%) achieved CR/CRi, 8 (21.6%) achieved MLFS, and 15 (40.6%) had RD. Six additional patients died during cycle 2, and responses after cycle 2 were evaluable in 31 patients: 13 (41.9%) achieved CR/CRi, 10 (32.3%) achieved MLFS, and 8 (25.8%) had RD/MR. Baseline characteristics and early outcomes are summarized in Table 1. Prior azacitidine exposure was unevenly distributed across response groups, and secondary AML was common among patients with MLFS after cycle 2.
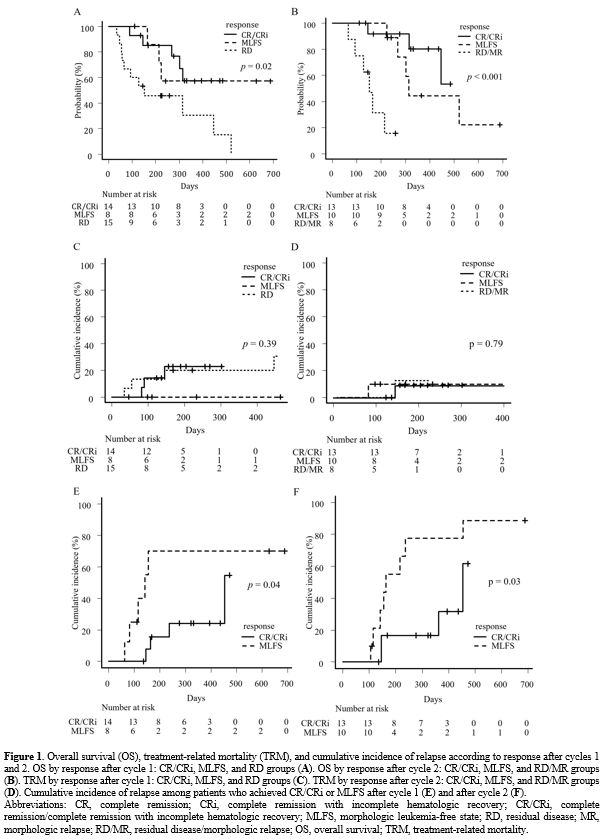
After cycle 1, the 180-day OS rates were 85.1% (95% CI, 52.3-96.1) in the CR/CRi group, 85.7% (95% CI, 33.4-97.9) in the MLFS group, and 45.7% (95% CI, 20.1-68.3) in the RD group (p=0.02) (Figure 1A). After cycle 2, the 180-day OS rates were 91.7% (95% CI, 53.9-98.8) in the CR/CRi group, 100% in the MLFS group, and 31.2% (95% CI, 4.8-64.1) in the RD/MR group (p < 0.001) (Figure 1B). These findings suggest that patients who remained in RD/MR after cycle 2 had poor short-term outcomes, whereas patients with MLFS after cycle 2 had relatively favorable short-term survival despite not achieving remission-equivalent disease control. However, because patients who died before response assessment were excluded from these comparisons and because MRD information was unavailable, these data should not be interpreted as evidence that MLFS is equivalent to CR/CRi.
TRM did not differ clearly across response groups.
After cycle 1, the 180-day TRM rates were 22.9% (95% CI, 4.9-48.5) in the CR/CRi group, 0% in the MLFS group, and 20.0% (95% CI, 4.4-43.7) in the RD group (p=0.39). After cycle 2, the corresponding rates were 9.1% (95% CI, 0.4-34.7), 10.0% (95% CI, 0.5-37.4), and 12.5% (95% CI, 0.2-48.6), respectively (p=0.79) (Figure 1C-D). In contrast, relapse was more frequent among patients with MLFS than among those with CR/CRi. Among patients who achieved CR/CRi or MLFS after cycle 1, the 180-day relapse rates were 15.5% (95% CI, 2.2-40.2) and 70.0% (95% CI, 16.6-93.2), respectively (p=0.04). Among those who achieved CR/CRi or MLFS after cycle 2, the 180-day relapse rates were 16.7% (95% CI, 2.3-42.6) and 55.0% (95% CI, 17.1-81.7), respectively (p=0.03) (Figure 1E-F). Thus, MLFS appeared to be associated with a persistently high risk of relapse even when short-term OS was preserved. Among the 8 patients who achieved MLFS after cycle 2 and subsequently relapsed, post-relapse treatment was best supportive care in 7 patients and venetoclax plus low-dose cytarabine in 1 patient; 4 patients survived for at least 120 days after relapse, suggesting that relapse after MLFS was not uniformly associated with immediate mortality.
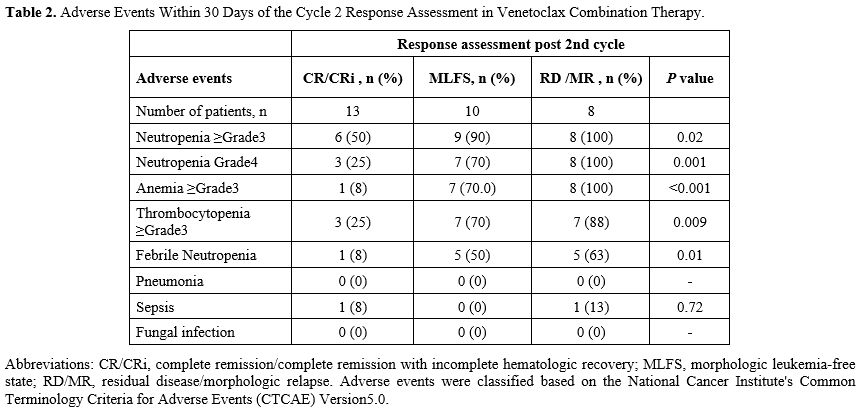
The toxicity burden in the MLFS group was also clinically relevant. Within 30 days of cycle 2 response assessment, grade 3 or higher neutropenia, grade 4 neutropenia, grade 3 or higher anemia or thrombocytopenia, and febrile neutropenia were more common in the MLFS and RD/MR groups than in the CR/CRi group (Table 2). Delays of ≥14 days in initiating cycle 3 due to adverse events occurred in 0 (0%), 5 (50%), and 1 (13%) patients in the CR/CRi, MLFS, and RD/MR groups, respectively. Venetoclax dose reductions due to myelosuppression in cycle 3 were required in 0 (0%), 8 (80%), and 0 (0%) patients, respectively. These observations further suggest that MLFS after cycle 2 represents a clinically burdensome intermediate response state, characterized by persistent cytopenias and a substantial need for treatment modification, consistent with the need for treatment adjustments reported in longer-term follow-up of venetoclax-based therapy.[3]
 |
|
Among patients with MLFS or RD/MR after cycle 2, subsequent CR/CRi was achieved in 4 of 10 (40.0%) patients in the MLFS group and in 1 of 8 (12.5%) patients in the RD/MR group. The median time from cycle 2 response assessment to CR/CRi was 44 days (range, 28-79) in the MLFS group and 60 days in the single RD/MR case. All patients achieved their best response within 4 cycles, and no CR/CRi achievement was observed beyond cycle 5. Two patients who achieved CR/CRi proceeded to allogeneic hematopoietic stem cell transplantation after cycles 2 and 3, respectively. These data are consistent with prior reports showing that early MLFS can function as a transitional response state in a subset of patients treated with venetoclax-based therapy.[4,5]
Discussion
The present study highlights the complex and clinically heterogeneous nature of morphologic leukemia-free state (MLFS) in patients with acute myeloid leukemia (AML) treated with venetoclax-based combination therapy. Although MLFS is formally categorized as a response according to established criteria, our findings suggest that it should not be interpreted as equivalent to CR/CRi or as a surrogate for durable disease control. Rather, MLFS appears to represent an intermediate clinical state characterized by discordance between morphologic blast clearance and incomplete hematopoietic recovery, with important implications for both disease control and treatment tolerance.[6]A central observation of this study is the dissociation between short-term overall survival and relapse risk in patients achieving MLFS. While 180-day survival was comparable to that observed in patients achieving CR/CRi, relapse rates were substantially higher, indicating that morphologic remission alone does not adequately capture residual disease burden in this setting. In the absence of measurable residual disease (MRD) assessment, MLFS likely encompasses biologically heterogeneous states, ranging from near-complete disease eradication with delayed count recovery to persistent subclinical leukemic clones capable of rapid re-expansion. This heterogeneity is consistent with emerging data demonstrating that depth of response, rather than morphologic criteria alone, is an important determinant of durable remission in venetoclax-treated AML.[5,7]
From a biological perspective, MLFS may reflect a convergence of disease-related and treatment-related factors. The high prevalence of secondary AML and prior exposure to hypomethylating agents in the MLFS group may reflect impaired marrow reserve or reduced hematopoietic fitness, which could contribute to delayed count recovery. Venetoclax-based therapy, while highly effective at inducing blast clearance through BCL-2 inhibition, may also contribute to prolonged cytopenias in vulnerable patients. In this context, MLFS may represent not a distinct biological response category, but a transitional phenotype arising from asynchronous leukemic clearance and hematopoietic recovery.[3,4] Because these interpretations were not directly tested in the present study, they should be regarded as hypothesis-generating.
Clinically, MLFS has significant implications. Patients in MLFS experienced a high burden of grade ≥3 cytopenias, frequent treatment delays, and a substantial need for dose modifications, underscoring that MLFS is not a benign or stable disease state. At the same time, a subset of patients subsequently converted to CR/CRi, suggesting that MLFS may, in some cases, be reversible rather than a treatment failure. These observations support a dynamic interpretation of MLFS, in which longitudinal assessment—rather than a single time-point evaluation—is essential for clinical decision-making.[4,8]
Importantly, our data suggests that persistent residual disease or morphologic relapse (RD/MR) after cycle 2 identifies a population with clearly poor short-term outcomes, whereas MLFS occupies an intermediate-risk category. This distinction may have practical implications for therapeutic strategy. While RD/MR may warrant early escalation of treatment or a transition to alternative approaches, the optimal management of MLFS remains undefined. Potential strategies include continued therapy with careful dose adjustment, early consideration of allogeneic hematopoietic stem cell transplantation in eligible patients, or incorporation of MRD-guided decision-making where available.[7,8]
Several limitations must be acknowledged. The retrospective, single-center design and small sample size limit the generalizability of our findings. Most importantly, early deaths prior to response assessment introduce a substantial survivor-selection bias, potentially inflating survival estimates in all response groups. In addition, the absence of MRD assessment precludes definitive conclusions regarding the biological depth of response in MLFS. Finally, the high proportion of patients with secondary AML and prior therapy exposure may have influenced both response kinetics and toxicity profiles, as observed in other real-world venetoclax-treated cohorts.[3,9,10]
Despite these limitations, this study provides clinically relevant insights into an increasingly encountered response category in the era of venetoclax-based therapy. Our findings support the view that MLFS should not be considered remission-equivalent disease control, but rather a clinically meaningful intermediate state that requires careful interpretation and individualized management. Future prospective studies incorporating MRD assessment and molecular profiling will be essential for refining the prognostic and therapeutic implications of MLFS and for guiding response-adapted treatment strategies in AML.[7,8]
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.Author Contributions
K. Motegi and K. Miyao designed the study, organized the project, performed the statistical analysis, and analyzed the data. F.O., H.W., H.S., Y.I., and M.S. contributed to data collection. K. Motegi wrote the first draft of the paper, and all other authors contributed to the final version.References
- Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424-47. https://doi.org/10.1182/blood-2016-08-733196
- Hassan
H, Rompola M, Glaser AW, Kinsey SE, Phillips RS. Validation of a
classification system for treatment-related mortality in children with
cancer. BMJ Paediatr Open. 2017;1:e000082. https://doi.org/10.1136/bmjpo-2017-000082
- Pratz KW, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Döhner H, et al. Long-term follow-up of VIALE-A: Venetoclax and azacitidine in chemotherapy-ineligible untreated acute myeloid leukemia. Am J Hematol. 2024;99:615-624. https://doi.org/10.1002/ajh.27246
- Jonas
BA, Wei AH, Recher C, DiNardo CD, Jang JH, Pratz KW, et al. Timing of
response with venetoclax combination treatment in patients with newly
diagnosed acute myeloid leukemia. Am J Hematol. 2022;97:E299-E303. https://doi.org/10.1002/ajh.26600
- Pollyea
DA, Pratz K, Letai A, Jonas BA, Wei AH, Pullarkat V, et al. Venetoclax
with azacitidine or decitabine in patients with newly diagnosed acute
myeloid leukemia: Long-term follow-up from a phase 1b study. Am J
Hematol. 2021;96:208-17. https://doi.org/10.1002/ajh.26039
- Shallis
RM, Pollyea DA, Zeidan AM. The complete story of less than complete
responses: The evolution and application of acute myeloid leukemia
clinical responses. Blood Rev. 2021;48:100806. https://doi.org/10.1016/j.blre.2021.100806
- DiNardo
CD, Tiong IS, Quaglieri A, MacRaild S, Loghavi S, Brown FC, et al.
Molecular patterns of response and treatment failure after frontline
venetoclax combinations in older patients with AML. Blood.
2020;135:791-803. https://doi.org/10.1182/blood.2019003988
- Jain
AG, Volpe VO, Wang C, Ball S, Tobon K, Chan O, et al. Outcomes by best
response with hypomethylating agent plus venetoclax in adults with
previously untreated acute myeloid leukemia. Ann Hematol.
2025;104:307-315. https://doi.org/10.1007/s00277-024-05976-6
- Garciaz
S, Hospital M-A, Alary A-S, Saillard C, Hicheri Y, Mohty B, et al.
Azacitidine plus venetoclax for the treatment of relapsed and newly
diagnosed acute myeloid leukemia patients. Cancers (Basel).
2022;14:2025. https://doi.org/10.3390/cancers14082025
- Yan
M, Wang G, Wang J, Xu L. Efficacy of venetoclax combined with
azacitidine in elderly patients with relapsed acute myeloid leukemia.
Mediterr J Hematol Infect Dis. 2025;17(1):e2025058. https://doi.org/10.4084/MJHID.2025.058